12 July 2022: Articles 
Clinical Presentation of Serogroup 1-Associated Pneumonia and Diffuse Alveolar Hemorrhage: A Case Report and Literature Review
Challenging differential diagnosis, Diagnostic / therapeutic accidents, Management of emergency care, Rare disease, Educational Purpose (only if useful for a systematic review or synthesis)
Kazuhiro Ishikawa1ABCDEF*, Tomoaki NakamuraDOI: 10.12659/AJCR.936309
Am J Case Rep 2022; 23:e936309
Abstract
BACKGROUND: We report a case of diffuse alveolar hemorrhage (DAH) caused by Legionella pneumophila serogroup (SG) 1 and review the existing literature to identify risk factors and determine the prognosis of patients with Legionella pneumonia-associated DAH.
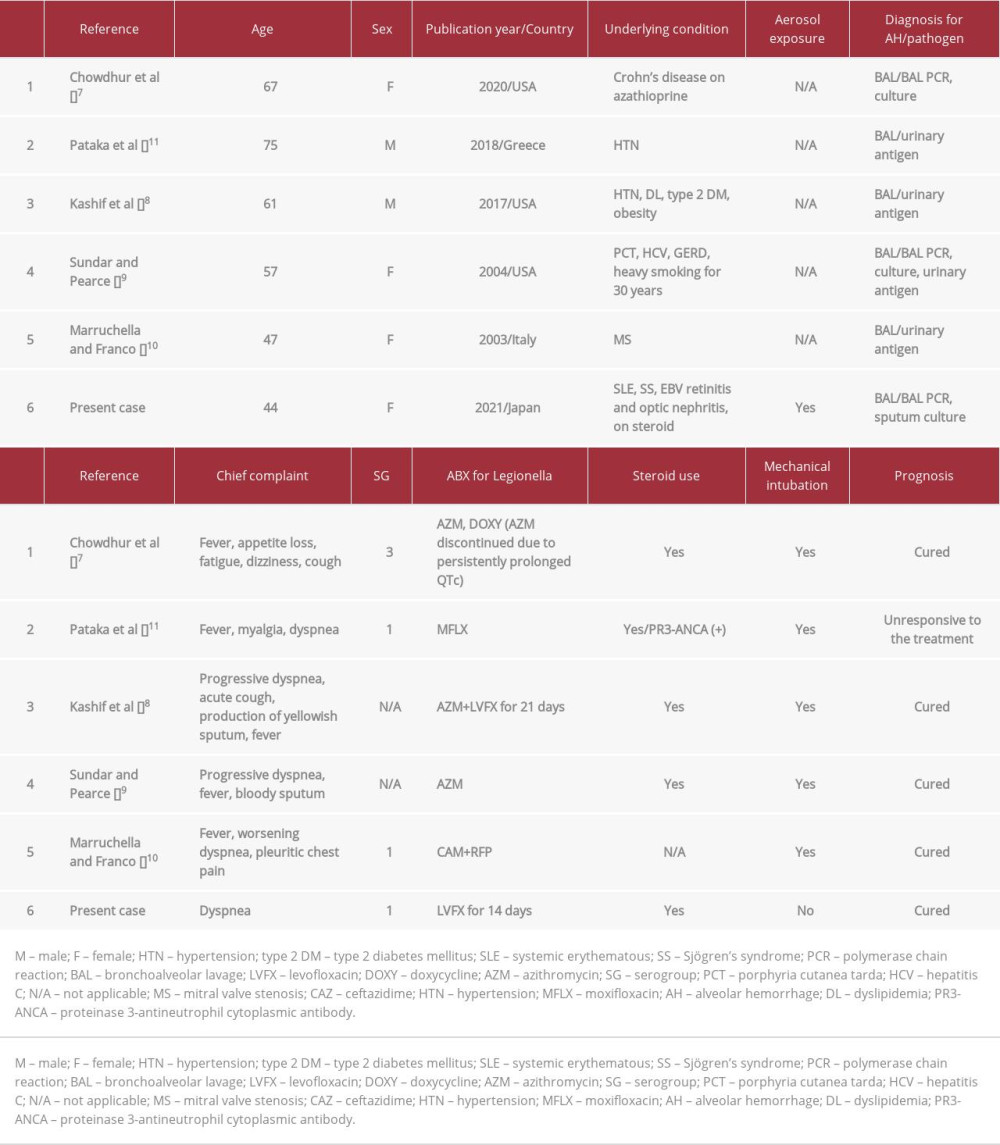
CASE REPORT: A 44-year-old woman was admitted to our hospital following the presentation of dyspnea for a few days. Chest computed tomography (CT) findings revealed “crazy-paving” pattern in the right upper lobe implicating DAH and consolidation in the lower lobe. Analysis of the bronchoalveolar lavage (BAL) fluid revealed DAH, with further analyses identifying L. pneumophila SG 1 as the causative agent. The patient was successfully treated with levofloxacin and a red blood cell transfusion and discharged on the 32nd day of hospitalization. A literature review of 6 reported cases (including our case) of Legionella pneumonia-associated DAH revealed that the median age of patients with DAH was 59 years (range, 44-75 years), involving female patients in 4 cases (67%) and the use of immunosuppressive drugs in 2 cases (33%). Three cases were BAL Legionella polymerase chain reaction (PCR)-positive and 4 cases were diagnosed using a urinary Legionella antigen test (one case was simultaneously PCR-positive). These infections were caused by L. pneumophila SG 1 in three cases and SG 3 in one case. Mechanical ventilation was used in 5 cases (83%) and one patient had an unfavorable prognosis. Steroids for DAH were used in 5 cases (83%), and 2 cases responded to this treatment.
CONCLUSIONS: Our case highlights that clinicians should be aware of Legionella spp. as a cause of DAH in an immunocompetent host with “crazy-paving” pattern on chest CT, and perform a urinary antigen test and BAL PCR for diagnosis.
Keywords: Legionella pneumophila, Adult, Aged, Female, Hemorrhage, Humans, Legionnaires' Disease, Middle Aged, Serogroup
Background
Diffuse alveolar hemorrhage (DAH) represents a syndrome that can complicate many clinical conditions and may be life-threatening, requiring prompt treatment. The typical presentations of DAH are anemia, hemoptysis, and pulmonary infiltrates on chest X-ray. A variety of diseases are associated with the development of DAH. Most cases of DAH are caused by the disruption of pulmonary capillarity and closely associated with systemic vasculitis and findings such as anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis, anti-glomerular basement membrane (GBM) disease, systemic lupus erythematosus (SLE), and connective tissue diseases [6]. DAH also co-occurs with several other conditions, including the use of certain drugs, infection, and transplantation. Here, we report our experience with a case of DAH caused by
Case Report
A 44-year-old woman with a history of SLE and Sjögren’s syndrome, who had been prescribed valganciclovir and methylprednisolone for retinitis and optic neuritis caused by Epstein–Barr virus for 3 months, presented with dyspnea, which had been occurring for a couple of days prior to admission. She prescribed regular drug and did not started new drug these days. The patient visited our outpatient clinic and denied the experience of any bloody sputum or orthopnea. She stated that she replaces the water in her bathtub once every 3 days and has been using a humidifier at her workplace. She also had an allergy to sulfamethoxazole/trimethoprim that was identified as a skin rash in a previous round of treatment. She had no history of smoking or alcohol consumption and was conscious with an oxygen saturation of 86% on room air upon admission. Her respiratory rate was 22 breaths/min, blood pressure was 124/54 mmHg, and pulse rate was 92 beats/min. Physical examination revealed respiratory effort while breathing and bilateral coarse crackles, but there was no swelling in the lower legs or jugular vein distention. Laboratory data revealed the following: white blood cell count, 13,400 cells/µL (neutrophils 97.5%); hemoglobin, 6.0 g/dL; platelet count, 15.1×103/µL; blood urea nitrogen, 85.8 mg/dL; creatinine, 2.09 mg/dL; lactate dehydrogenase, 1,164 U/L; aspartate aminotransferase, 40 U/L; alanine aminotransferase, 70 U/L; creatine kinase, 325 U/L; sodium, 144 mEq/L; C-reactive protein, 23.1 mg/dL; β-d-glucan, 47.0 pg/mL; C3 protein, 124 mg/dL; and C4 protein, 34 mg/dL. The anti-GBM antibody, PR3-ANCA, and MPO-ANCA evaluations were all negative. The anti-DNA antibody was <2 IU/mL, anti-IgG antibody was 745 mg/dL, and natriuretic test-pro-B-type natriuretic peptide was 2,056 pg/mL. Urine antigen tests for
Discussion
We first identified our target papers with a literature search of the relevant databases including PubMed, Embase, and Ichushi (up to June 2021) (process described in Supplementary Table 1). We then reviewed the titles and abstracts of the database records and retrieved the full text for studies deemed appropriate for this evaluation before extracting the relevant case data. These evaluations returned 5 articles describing 5 cases of DAH due to
Evaluation of the case data revealed that the median age of patients with DAH was 59 years (range, 44–75 years) and was reported in 4 females (67%) and 2 males, with 3 from the United States, 2 from Europe, and one from Japan. The use of immunosuppressive drugs such as steroids and azathioprine was found in only 2 cases. Non-immunosuppressed patients also had DAH due to
DAH is used to describe a condition in which the alveolar space is filled with blood, which is usually caused by hemorrhages within the pulmonary microcirculation resulting from collagen diseases such as SLE, ANCA-related vasculitis syndrome, Goodpasture syndrome, and conditions caused by certain drugs, infections, cardiac diseases, and allergies [12,13]. DAH is a life-threatening condition that requires prompt diagnosis and treatment as it progresses rapidly, presenting as dyspnea, bloody sputum, and cough [12]. The mortality rate is reportedly 25–50% [14]. For the evaluation of the causative agent of DAH in our patient, we first considered connective tissue diseases such as SLE, ANCA-associated vasculitis, and Goodpasture syndrome, and examined the echocardio-gram for valvular disease or congestive heart failure. The patient tested negative for all these diseases/conditions, which left the possibility of DAH caused by infection. The most common DAH-causing infections are influenza A, dengue hemorrhagic fever, leptospirosis, malaria, and
In general, CT finding of DAH is the “crazy-paving” pattern consisting of scattered or diffuse ground-glass attenuation with superimposed interlobular septal thickening and intralobular lines [21]. In contrast, CT findings of
In total, 4 patients were treated with macrolides (replaced with tetracyclines during treatment due to side effects), and 2 with fluoroquinolones. Of these, 5 patients were intubated, and one patient, who had a particularly poor prognosis, was treated with moxifloxacin. A previous study showed that the efficacy of azithromycin and fluoroquinolone for the treatment of
In animal models, the administration of
Despite these observations, there are some limitations to our study. DAH is generally diagnosed using BAL samples (50 mL×3 times), and the color of the BAL fluid becomes more intense between washes due to increased bleeding. In our case, BAL evaluation was performed only once due to concerns about the possibility of BAL worsening the respiratory condition of the patient. However, a chest CT revealed a pattern of DAH. The patient initially needed blood transfusions, but after treatment for
Conclusions
Our case study suggests that immunosuppressed patients presenting with DAH, who are known to be exposed to contaminated aerosols, should be evaluated for infectious DAH from pathogens such as
References:
1.. Edelstein PH, Roy CR, Legionnaires’ disease and Pontiac fever: Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases, 2014; 2633-44, Philadelphia, Elsevier
2.. Kassha K, Abuanza I, Hadi SA, Hilton R, Severe Legionnaires’ disease complicated by multi-organ dysfunction in a previously healthy patient: A case report: Cases J, 2009; 2; 9151
3.. , Legionella (Legionnaires’ Disease Pontiac Fever) [accessed 31 October 2021].https://www.cdc.gov/legionella/clinicians/disease-specifics.html
4.. Amemura-Maekawa J, Kura F, Helbig JH: J Med Microbiol, 2010; 59; 653-59
5.. Palusińska-Szysz M, Cendrowska-Pinkosz M, Occurrence and pathogenicity of the family of Legionellaceae: Postepy Hig Med Dosw, 2008; 62; 337-53
6.. Lara AR, Schwarz MI, Diffuse alveolar hemorrhage: Chest, 2010; 137; 1164-71
7.. Chowdhury A, Syed AK, Susan H: J Community Hosp Intern Med Perspect, 2020; 10(3); 262-64
8.. Kashif M, Patel R, Bajantri B, Diaz-Fuentes G, Legionella pneumonia associated with severe acute respiratory distress syndrome and diffuse alveolar hemorrhage – a rare association: Respir Med Case Rep, 2017; 21; 7-11
9.. Sundar KM, Pearce MJ, Diffuse alveolar hemorrhage due to Legionella pneumonia: Sarcoidosis Vasc Diffuse Lung Dis, 2004; 21; 158-59
10.. Marruchella A, Franco C, Severe alveolar haemorrhage in Legionella pneumonia: Sarcoidosis Vasc Diffuse Lung Dis, 2003; 20; 77
11.. Pataka A, Kotoulas SC, Sourla E, Is it just an infection?: Breathe (Sheff), 2018; 14; e100-4
12.. Collard HR, Schwarz MI, Diffuse alveolar hemorrhage: Clin Chest Med, 2004; 25; 583-92
13.. Colby TV, Fukuoka J, Ewaskow SP, Pathologic approach to pulmonary hemorrhage: Ann Diagn Pathol, 2001; 5; 309-19
14.. Franks TJ, Koss MN, Pulmonary capillaritis: Curr Opin Pulm Med, 2000; 6; 430-35
15.. von Ranke FM, Zanetti G, Hochhegger B, Marchiori E, Infectious diseases causing diffuse alveolar hemorrhage in immunocompetent patients: A state-of-the-art review: Lung, 2013; 191; 9-18
16.. de Prost N, Parrot A, Cuquemelle E, Diffuse alveolar hemorrhage in immunocompetent patients: etiologies and prognosis revisited: Respir Med, 2012; 106; 1021-32
17.. Ito A, Yamamoto Y, Ishii Y, Evaluation of a novel urinary antigen test kit for diagnosing Legionella pneumonia: Int J Infect Dis, 2021; 103; 42-47
18.. Avni T, Bieber A, Green H, Diagnostic accuracy of PCR alone and compared to urinary antigen testing for detection of Legionella spp.: A systematic review: J Clin Microbiol, 2016; 54; 401-11
19.. Gershengorn HB, Keene A, Dzierba AL, Wunsch H, The association of antibiotic treatment regimen and hospital mortality in patients hospitalized with Legionella pneumonia: Clin Infect Dis, 2015; 60; e66-79
20.. Baskerville A, Conlan JW, Ashworth LA, Dowsett AB: Br J Exp Pathol, 1986; 67; 527-36
21.. Santiago ER, Jeremy JE, Mariano V, Tomas F, “Crazy-paving” pattern at thin-section CT of the lungs: Radiologic-pathologic overview: Radiographics, 2003; 23(6); 1509-19
22.. Fumikazu S, Hitoshi T, Hajime G, Kazuhiro T: J Comput Assist Tomogr, 2007; 31(1); 125-31
In Press
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942826
14 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942770
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943214
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943010
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250