20 June 2022: Articles 
A Case of Exacerbation of Haloperidol-Induced Rhabdomyolysis Following the Onset of COVID-19
Unusual clinical course, Challenging differential diagnosis, Adverse events of drug therapy
Keisuke Takada1ABDEF*, Yukiyoshi Sada2ABDEF, Masaru SamuraDOI: 10.12659/AJCR.936589
Am J Case Rep 2022; 23:e936589
Abstract
BACKGROUND: Rhabdomyolysis is a condition in which intracellular components are released into the blood and urine. Rhabdomyolysis can be caused by drug-related complications and COVID-19; however, the underlying mechanism is not clear. In this study, we report a case of rhabdomyolysis complicated by COVID-19, in which we presumed that the cause of rhabdomyolysis was related to prior administration of haloperidol by assessment of the drug history and progression of myopathy.
CASE REPORT: A 52-year-old man with schizophrenia experienced worsening insomnia 10 days before admission. Thus, haloperidol was increased from 1.5 mg to 3 mg once daily, and 2 to 3 days later, he developed hand tremors and weakness. One day prior to admission, the patient suddenly developed severe back pain. Based on the examination, the patient was diagnosed with COVID-19 complicated with rhabdomyolysis. Laboratory findings on admission were as follows: creatine phosphokinase: 41 539 IU/L; urinary myoglobin, 190×10³ ng/mL; and hematuria scale, grade 4. On day 1, he was started on saline infusion; therefore, haloperidol was discontinued. On day 2, the hematuria resolved. On day 5, the tremor, weakness, and back pain had resolved. On day 7, his creatine kinase level was 242 IU/L, and saline was administered.
CONCLUSIONS: It has been suggested that the onset of COVID-19 can exacerbate haloperidol-induced rhabdomyolysis. Therefore, if there is a complication of rhabdomyolysis and COVID-19, it is important to review the drug history, specifically that of haloperidol. We recommend hydration and discontinuation of haloperidol to avoid acute kidney injury, in addition to treating COVID-19.
Keywords: COVID-19, creatine kinase, Haloperidol, rhabdomyolysis, SARS-CoV-2, Acute Kidney Injury, Hematuria, Humans, Male, Middle Aged
Background
Rhabdomyolysis is a condition in which the massive release of intracellular components into the blood and urine by degeneration and necrosis of skeletal muscle cells can be associated with acute renal failure [1]. As it is a life-threatening disease, prompt diagnosis is required to avoid acute complications. Rhabdomyolysis is known to be caused by drug-related complications [2]. It has been suggested that muscle toxicity caused by antipsychotics, including haloperidol, is mediated by serotonin and dopamine blockade [3]. The effect of haloperidol appears to be related to increased blood levels rather than the duration of administration [4].
There have been several case reports of haloperidol-induced rhabdomyolysis without the malignant syndrome [5,6]. There were 309 reports of haloperidol-induced rhabdomyolysis in VigiAccess™ (accessed April 2022) and 164 in the FDA Adverse Event Reporting System (accessed April 2022). In addition, rhabdomyolysis can occur as a complication of viral infections [7]. Furthermore, rhabdomyolysis in COVID-19 has been reported [8], but its mechanism and etiology are not clear.
In cases caused by haloperidol, rhabdomyolysis progresses slowly over several weeks [9]. Hence, there is a report that 78% of rhabdomyolysis cases associated with COVID-19 occurred after the onset of COVID-19 [10]. If the rhabdomyolysis progress did not fit the typical case of haloperidol-only and COVID-19-only involvement, then it may be a combination of these effects. Herein, we report a case of rhabdomyolysis complicated by COVID-19, in which we presumed that the cause of rhabdomyolysis was related to prior administration of haloperidol by assessment of the drug history.
Case Report
A 52-year-old man was diagnosed with schizophrenia several years ago, and pharmacotherapy was continued. One month before admission, his insomnia worsened. The medication was then changed from brexpiprazole 2 mg once daily (QD) to haloperidol 1.5 mg QD. Ten days prior to admission, insomnia persisted. Haloperidol was then increased to 3 mg QD, and 2 to 3 days later, hand tremors and weakness developed. One day prior to admission, he suddenly developed fever and severe back pain with difficulty moving, which prompted a consult at the Emergency Department. As a result of the examination, the patient was diagnosed with COVID-19 complicated by rhabdomyolysis and was admitted to our hospital. His medical history included diabetes mellitus, which was maintained with dapagliflozin 10 mg QD. There was no travel history or history of contact with COVID-19-infected patients. The patient’s social history included no history of smoking.
The physical examination findings on admission were as follows: height, 176 cm; body weight, 85 kg; body mass index, 27.4 kg/m2; blood pressure, 120/87 mm Hg; pulse, 85 beats/ min; body temperature, 36.6°C; respiratory rate, 30 breaths/ min; saturation of percutaneous oxygen, 93% (room air); and consciousness, Glasgow Coma Scale E4V5M6. The psychiatric examination and mental status were normal. A chest examination revealed coarse crackles in the lower right lung. The pain was observed extending from the lumbar spine to the sacrum. Cardiac and abdominal examinations were normal, and no excessive sweating was observed. Neurological examination revealed severe tremor and weakness in both upper limbs, but without muscular rigidity, dysphagia, or akinesia.
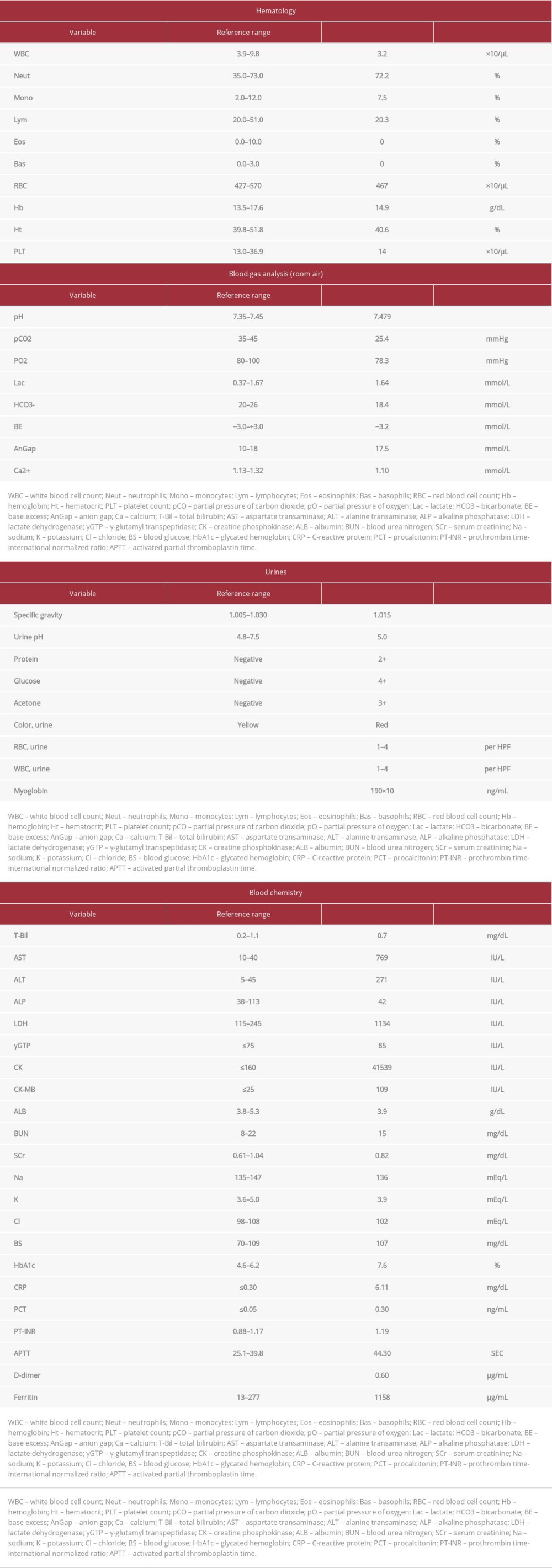
Laboratory examination findings on admission were as follows: white blood cell count, 3.2×103/μL; lymphocyte, 650/μL; C-reactive protein, 6.11 mg/dL; creatinine, 0.82 mg/dL; lactate dehydrogenase, 1134 IU/L; creatine phosphokinase, 41 539 IU/L; ferritin, 1158 μg/mL; D-dimer, 0.8 μg/mL; urinary myoglobin, 190×103 ng/mL; and hematuria scale, grade 4 (Table 1). Loop-mediated isothermal amplification assays of a nasopharyngeal swab sample indicated SARS-CoV-2 infection. Chest computed tomography (CT) revealed bilateral peripheral ground-glass opacities (Figure 1).
The clinical course after hospitalization is shown in Figure 2. On hospital day 1, he was started on saline 1800 mL/day continuous infusion, remdesivir 100 mg intravenous (i.v.) every 24 h (q24h) (200 mg on the first day), dexamethasone 6.6 mg IV q24h, and oxygenation O2 1 L (nasal cannula). In addition, haloperidol and risperidone were discontinued. On hospital day 2, the tremor, back pain, and weakness reduced, and the hematuria disappeared. Additionally, his oxygen saturation level was maintained at >95%. The oxygen administration was discontinued. On hospital day 3, the tremor and weakness resolved, and on hospital day 5, the back pain resolved along with a decrease in creatine kinase (CK) to 2600 IU/L. On hospital day 6, the oxygen saturation decreased to 94% (room air), and oxygen administration of 2 L (nasal cannula) was restarted. On hospital day 7, his CK level was 242 IU/L; thus, he was administered saline. However, his oxygen saturation decreased to 94% (O2 10 L reservoir mask); therefore, he was administered a single dose of tocilizumab 680 mg i.v. and started on methylprednisolone 1000 mg IV q24h. On hospital day 10, the remdesivir was completed, and the methylprednisolone was changed to dexamethasone 6 mg PO QD. Subsequently, the dexamethasone and oxygen were administered, and the patient was discharged on hospital day 15.
Discussion
In this study, we report a case of haloperidol-induced rhabdomyolysis followed by the onset of COVID-19 and exacerbation of rhabdomyolysis at the time of the onset of COVID-19. Immediately after starting the examination, the cause of rhabdomyolysis could not be determined. The patient was thought to have developed COVID-19 a few days before visiting the Emergency Department, and that COVID-19 was the cause of the rhabdomyolysis. Therefore, we reviewed his medical and drug history and revealed that the dose of haloperidol was increased owing to schizophrenia 10 days before the onset of COVID-19. Moreover, as for the musculoskeletal symptoms, tremor and weakness developed immediately after haloperidol was increased, and subsequently, severe back pain suddenly occurred on the first day of COVID-19. In contrast, the most common days of rhabdomyolysis onset associated with COVID-19 are within a week of hospitalization [11], whereas cases of rhabdomyolysis associated with haloperidol occur within 2 months of initiation or 3 to 7 days after a dose increase [4]. Moreover, the timing of the tremors and weakness suggest rhabdomyolysis in this patient was more related to haloperidol intake than to COVID-19. These considerations suggest that the cause of this rhabdomyolysis was strongly related to haloperidol, as the musculoskeletal symptoms developed before the onset of COVID-19. In this regard, it is recommended that a careful review of the medical and drug history should be completed to determine the cause of rhabdomyolysis in such patients.
Generally, CK elevation caused by haloperidol is a differential diagnosis of the malignant syndrome and rhabdomyolysis [12]. A previous report suggested that there is a risk of developing malignant syndrome when prior haloperidol decanoate administration complicates COVID-19 [13]. However, in our patient, no physical or neurological examination suggested a malignant syndrome; therefore, CK elevation was strongly suspected to be caused by rhabdomyolysis. Further, the differential diagnoses leading to rhabdomyolysis, other than haloperidol, were trauma, exertion, and other factors such as electrolyte and endocrine system abnormalities; however, these were considered unlikely after examination upon admission. To the best of our knowledge, there is no report of exacerbation of haloperidol-induced rhabdomyolysis at the onset of COVID-19, which is a rare case.
The related mechanisms of rhabdomyolysis, haloperidol, and COVID-19 demonstrate that they have similar effects on the dopaminergic systems. COVID-19 appears to cause disorders of the dopaminergic system [14]. Haloperidol-induced rhabdomyolysis was described as a consequence of dopamine blockade [3]. The common effect of these is depletion of the dopaminergic system. We then hypothesized that the occurrence of comorbidities exacerbates rhabdomyolysis. However, this hypothesis needs to be established in an experimental animal model.
By December 31, 2021, several cases of rhabdomyolysis and COVID-19 had been reported [8,15–20]. However, our patient was not screened for serum phosphorus, which is necessary to assess rhabdomyolysis and its complications. Therefore, the McMahon score [21] can estimate the severity score of rhabdomyolysis without measuring serum phosphorus, and this score ranges from 8.5 to 11.5 points to predict the patient’s prognosis. This value indicates that the risk of death and conversion to renal replacement therapy was 30% (phosphorus <4.0 mg/dL) to 65% (phosphorus >5.4 mg/dL). Thus, the risk of prognostic failure in our patient was moderately high. In addition, there are a few reports of death in cases of rhabdomyolysis complicated by COVID-19 [8,19,22]. Most of the cases of death had initiated treatment on days 3 to 5 after the onset of COVID-19 symptoms. The reason for the avoidance of acute kidney injury and the survival of this patient was that he was diagnosed on the second day after the onset of COVID-19 symptoms. Furthermore, it was considered effective to immediately discontinue haloperidol and initiate the hydration and treatment of COVID-19.
The limitations of this study are as follows: First, this case was not analyzed for the SARS-CoV-2 variant; therefore, the results may not be identical for all cases of SARS-CoV-2. Second, this study is based on a single case, and therefore, it may not apply to all rhabdomyolysis complicated by COVID-19.
Conclusions
The association between rhabdomyolysis, haloperidol, and COVID-19 is hypothesized to be caused by a combined disorder of the dopaminergic system. Moreover, the onset of COVID-19 can exacerbate haloperidol-induced rhabdomyolysis. Therefore, if there is a complication of rhabdomyolysis and COVID-19, it is important to review the drug history, including that of haloperidol, before the onset of the disease to determine its etiology.
The recommended management strategies to avoid AKI and save lives are as follows. Early discontinuation of haloperidol and hydration should be achieved in the management of rhabdomyolysis.
Figures
References:
1.. Khan FY, Rhabdomyolysis: A review of the literature: Neth J Med, 2009; 67; 272-83
2.. Prendergast BD, George CF, Drug-induced rhabdomyolysis – mechanisms and management: Postgrad Med J, 1993; 69; 333-36
3.. Packard K, Price P, Hanson A, Antipsychotic use and the risk of rhabdomyolysis: J Pharm Pract, 2014; 27; 501-12
4.. Star K, Iessa N, Almandil NB, Rhabdomyolysis reported for children and adolescents treated with antipsychotic medicines: A case series analysis: J Child Adolesc Psychopharmacol, 2012; 22; 440-51
5.. Karre PR, Gujral J, Rodriguez E, Rhabdomyolysis with erratic serum creatinin phosphokinase levels: Intramuscular injection of haloperidol decanoate: BMJ Case Rep, 2009; 2009; bcr0620091931
6.. Marsh SJ, Dolson GM, Rhabdomyolysis and acute renal failure during high-dose haloperidol therapy: Ren Fail, 1995; 17; 475-78
7.. Ayala E, Kagawa FT, Wehner JH, Rhabdomyolysis associated with 2009 influenza A(H1N1): JAMA, 2009; 302; 1863-64
8.. Khosla SG, Nylen ES, Khosla R, Rhabdomyolysis in patients hospitalized with COVID-19 infection: Five case series: J Investig Med High Impact Case Rep, 2020; 8; 2324709620984603
9.. Yoshikawa H, Watanabe T, Abe T, Oda Y, Ozawa K, Haloperidol-induced rhabdomyolysis without neuroleptic malignant syndrome in a handicapped child: Brain Dev, 2000; 22; 256-58
10.. Finsterer J, Scorza F, SARS-CoV-2 associated rhabdomyolysis in 32 patients: Turk J Med Sci, 2021; 51; 1598-601
11.. Haroun MW, Dieiev V, Kang J, Rhabdomyolysis in COVID-19 patients: A retrospective observational study: Cureus, 2021; 13; e12552
12.. Meltzer HY, Cola PA, Parsa M, Marked elevations of serum creatine kinase activity associated with antipsychotic drug treatment: Neuropsychopharmacology, 1996; 15; 395-405
13.. Kajani R, Apramian A, Vega A, Neuroleptic malignant syndrome in a COVID-19 patient: Brain Behav Immun, 2020; 88; 28-29
14.. Rethinavel HS, Ravichandran S, Radhakrishnan RK, Kandasamy M, COVID-19 and Parkinson’s disease: Defects in neurogenesis as the potential cause of olfactory system impairments and anosmia: J Chem Neuroanat, 2021; 115; 101965
15.. Byler J, Harrison R, Fell LL, Rhabdomyolysis following recovery from severe COVID-19: A case report: Am J Case Rep, 2021; 22; e931616
16.. Li APZ, Thomas S, Gokmen R, Kariyawasam D, Rhabdomyolysis and severe biphasic disturbance of calcium homeostasis secondary to COVID-19 infection: BMJ Case Rep, 2021; 14; e23961
17.. Samies NL, Pinninti S, James SH, Rhabdomyolysis and acute renal failure in an adolescent with coronavirus disease 2019: J Pediatr Infect Dis Soc, 2020; 9; 507-9
18.. Chedid NR, Udit S, Solhjou Z, COVID-19 and rhabdomyolysis: J Gen Intern Med, 2020; 35; 3087-90
19.. Solís JG, Esquivel Pineda A, Alberti Minutti P, Albarrán Sánchez A, Case report: Rhabdomyolysis in a patient with COVID-19: A proposed diagnostic-therapeutic algorithm: Am J Trop Med Hyg, 2020; 103; 1158-61
20.. Taxbro K, Kahlow H, Wulcan H, Fornarve A, Rhabdomyolysis and acute kidney injury in severe COVID-19 infection: BMJ Case Rep, 2020; 13; e237616
21.. McMahon GM, Zeng X, Waikar SS, A risk prediction score for kidney failure or mortality in rhabdomyolysis: JAMA Intern Med, 2013; 173; 1821-28
22.. Anwar H, Al Lawati A, Adolescent COVID-19 – associated fatal rhabdomyolysis: J Family Med Prim Care, 2020; 11; 2150132720985641
Figures
In Press
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943687
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943070
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943370
18 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943803
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250