05 July 2021: Articles 
New Horizons for Diagnostic Pitfalls of Cerebral Venous Thrombosis: Clinical Utility of a Newly Developed Cerebral Venous Thrombosis Diagnostic Score: A Case Report and Literature Review
Mistake in diagnosis
Faisal Khan1ABCDEF*, Muhannad SeyamDOI: 10.12659/AJCR.932123
Am J Case Rep 2021; 22:e932123
Abstract
BACKGROUND: Diagnosing cerebral venous thrombosis (CVT) poses significant challenges owing to a nonspecific clinical presentation, poorly correlated laboratory biomarkers, and low sensitivity of non-contrast head computed tomography (CT). We describe a case of missed CVT diagnosis, due to low clinical suspicion and nonrecognition of anemia as a prothrombotic factor, especially during an ulcerative colitis (UC) flare. A recently proposed CVT clinical probability score can guide clinicians in pursuing further neurovascular imaging.
CASE REPORT: A 35-year-old man, with treatment-naive UC, presented to the Emergency Department (ED) with new-onset diffuse headache, 4 weeks of bloody diarrhea, and weight loss. Initial ED laboratory studies revealed severe anemia and unremarkable non-contrast head CT. Two days later, the patient returned to the ED for worsening headache. Non-contrast head CT revealed a left temporal hypodensity. This was later confirmed as acute ischemia on magnetic resonance imaging (MRI). MR venogram revealed thrombosis of the left transverse and sigmoid sinuses, leading to initiation of therapeutic subcutaneous anticoagulation. Repeat MRI, secondary to worsening headache, revealed the development of petechial hemorrhages within the core of venous ischemia in the left temporal lobe. Therapeutic anticoagulation, along with symptomatic management of UC, led to clinical stabilization.
CONCLUSIONS: CVT should be suspected in patients with UC, especially in the context of anemia, presenting with new-onset or worsening headaches. Recognizing anemia as a thrombogenic factor is crucial. Diagnosis of CVT is challenging due to non-focal symptoms and poorly correlating diagnostic tests. We endorse implementing the CVT clinical probability score into AHA/ASA CVT guidelines to enhance diagnostic accuracy.
Keywords: Anemia, fibrin fragment D, Inflammatory Bowel Diseases, Stroke, Venous Thrombosis, Headache, Intracranial Thrombosis, Neuroimaging, Phlebography
Background
Cerebral venous thrombosis (CVT) poses a significant diagnostic challenge because of its nonspecific clinical presentation. Holocranial headache, in 90% of cases, is the most common acute presenting symptom [1], followed by seizures (generalized or focal) in about 40% of cases [1,2] and focal neurological deficits in approximately 37% of cases [3]. Diagnosis of CVT has proven to be cumbersome because there are no specific laboratory biomarkers and non-contrast head computed tomography (CT) has a low sensitivity [4,5]. Magnetic resonance imaging (MRI), CT venography, and MR venography are the criterion standard diagnostic tests for diagnosis of CVT [1]. Multiple risk factors have been associated with the pathogenesis of CVT. The correlation between inflammatory bowel disease (IBD) and CVT has been well established, and physicians should recognize IBD as a potential contributor to CVT. Misdiagnosis of CVT in patients with IBD can result in fatal consequences, with a 25% mortality rate [6].
We describe a patient with treatment-naive ulcerative colitis (UC) who presented to the Emergency Department (ED) with a new-onset headache and bloody diarrhea. Laboratory results revealed severe anemia, and after transfusion, the patient was discharged home with a diagnosis of tension headache. Two days later, he presented to the ED with worsening headache and hypersomnolence, and eventually was diagnosed with CVT. The diagnosis was initially missed due to low clinical suspicion and an unremarkable non-contrast head CT scan. This delay in diagnosis led to the development of venous ischemia, with associated petechial hemorrhage and a protracted hospitalization course. Diagnosis of CVT should be considered in patients with underlying inflammatory bowel disease (IBD) with a new-onset or atypical headache, especially in the context of severe anemia. The thrombogenic basis of anemia has been well established in the literature [6–11], but its clinical relevance has not yet been fully recognized.
A recently proposed CVT clinical probability score by Heldner et al can be implemented as a guide in the pursuance of diagnostic neurovascular imaging. We anticipate that the utilization of this score will improve diagnostic accuracy and management.
Case Report
A 35-year-old man with a history of treatment-naive UC presented to the ED with persistent new-onset holocranial headache of a throbbing quality and moderate severity. Additionally, he reported a 4-week history of bloody diarrhea and significant weight loss, for which he did not seek any medical attention. A mental status examination revealed that the patient was alert and oriented to person, place, time, and situation, with no speech impairment. A cranial nerve and sensorimotor examination was normal, with no meningeal signs. The laboratory findings from the patient’s initial ED visit are listed in Table 1. Of note, the complete blood count (CBC) revealed severe anemia with a hemoglobin (Hb) level of 5.5 g/dL. No D-dimer level was obtained during this ED visit. A non-contrast head CT scan showed no acute intracranial abnormalities. Four units of packed red blood cells were given to the patient. His headache responded to symptomatic treatment and the administration of intravenous morphine. The patient refused further workup and was discharged home with a diagnosis of episodic tension headache.
The patient returned to the ED 2 days later with a worsening headache and hypersomnolence. The laboratory findings are listed in Table 2. Of note, the CBC revealed an Hb level of 8.4 g/dL. A hypercoagulable panel (protein S, protein C, anti-thrombin III, factor VIII, antinuclear antibody, cardiolipin antibody, prothrombin G20210A, and factor V Leiden) showed no abnormalities. A non-contrast head CT scan revealed a left temporal hypodensity (Figure 1). Head and neck CT angiography showed no evidence of thrombosis in the intra-cranial or extra-cranial arterial circulation.
The patient was admitted for further evaluation owing to persistent symptoms and abnormal neuroimaging findings. A diffusion-weighted imaging (DWI) sequence revealed hyperintensity in the left temporal lobe, consistent with acute left temporal lobe ischemia (Figure 2), with no corresponding signal alteration on apparent diffusion coefficient (Figure 3). Fluid-attenuated inversion recovery (FLAIR) sequences revealed hyperintensity in the left temporal and occipital lobes (Figure 4). There was no evidence of hemorrhage on gradient echo images. MR venography of the head revealed acute venous thrombosis involving the left transverse and sigmoid sinuses extending into the proximal jugular bulb, with no evidence of any involvement of the deep venous system (Figure 5). This finding was corroborated by the CT venography of the brain revealing an absence of flow at the level of the left transverse sinus, while showing preserved flow on the right transverse sinus (Figure 6). A diagnosis of CVT, secondary to the procoagulant effect of UC potentiated by severe anemia, was made. Consequently, therapeutic anticoagulation with 60 mg/0.6 mL subcutaneous low-molecular-weight heparin (enoxaparin) twice daily was initiated. The patient was also started on 40 mg prednisone and 1600 mg mesalamine orally 3 times per day by the gastroenterologist. Worsening headache on day 6 prompted a repeat brain MRI. The DWI and FLAIR sequences show stability of venous ischemia in the left temporal and occipital lobes (Figure 7). Development of petechial hemorrhages within the core of the venous ischemic region of the left temporal lobe was seen on gradient echo sequence (Figure 8). An interdisciplinary discussion was initiated because the patient exhibited clinical and radiological deterioration. The patient and his family were consulted regarding transferring him to a higher level of care, with the option of endovascular intervention; however, the patient refused. Therapeutic doses of subcutaneous enoxaparin were continued with frequent neurological check-ups to monitor for any clinical deterioration. His headaches and hypersomnolence gradually improved, and on day 13, the patient was discharged home on warfarin 7.5 mg (target international normalized ratio [INR] 2.0–3.0), prednisone (40 mg, tapered over 3 months), mesalamine 400 mg 4 times daily, and ferrous sulfate 325 mg once daily.
On outpatient follow-up 3 months later with his primary care physician, the patient was found to be in remission, with significant weight gain and resolution of the anemia. The primary care physician ordered repeat neuroimaging; however, the patient refused additional testing. No headaches or focal neurological symptoms were reported at this visit, and the patient was lost to follow-up.
Discussion
Inflammatory bowel disease has been associated with a variety of neurological complications [12–15] (Table 3). One of the most serious and overlooked cerebrovascular complications of IBD is CVT [16]. IBD has a higher association with thrombosis, as compared to other systemic autoimmune disorders, such as rheumatoid arthritis and celiac disease [17–20]. Chronic IBD increases the risk of thromboembolism 3-fold, which increases to 15-fold during an active disease state [19]. Thromboembolic events can occur in up to 60% of patients during disease exacerbation or in the presence of intestinal complications, such as abscesses, fistulas, and pancolitis [6,19,20].
Harrison and Truelove (1967) were the first to report CVT in patients with UC [21]. Since then, multiple published cases have established the thrombotic complications of IBD [4,14,18,20,22]. CVT in particular is more commonly reported as a complication of IBD in the medical literature [1,5,12,13,15,16,19]. Young adults (average age, 29 years) with IBD are more prone to CVT than are young adults without IBD [6,19].
The pathogenesis of thromboembolism in IBD is not completely understood but has been hypothesized to be multifactorial [17]. IBD induces a prothrombotic state by altering levels of the coagulation factors, decreasing fibrinolytic activity, and inducing endothelial dysfunction during the active disease state, while also promoting platelet hyperactivity, independently of the disease state [10]. Alterations to components of the coagulation cascade include increased levels of fibrinogen and factors V, VII, VIII, and XI, as well as decreased levels of factor XIII. Additionally, fibrinolysis is altered by decreased levels of tissue-type plasminogen activator, and increased production of fibrinolytic inhibitors, including plasminogen activator inhibitor and thrombin-activatable fibrinolysis inhibitor. IBD has also shown an inhibitory effect on the production of anticoagulants, such as antithrombin III, protein C, and protein S [16]. Further, chronic inflammation in IBD is presumed to induce vascular endothelial surfaces into a procoagulant state via cytokines (interleukin [IL] 1, tumor necrosis factor [TNF] α) and endotoxins [10]. Finally, pathogenic mechanisms unrelated to disease activity status include thrombocytosis, hyperactivity of platelets secondary to chronic inflammatory response, and increased CD40 ligand production by platelets [16]. The interaction between CD40-CD40L and cytokines (IL-1 and TNF-α) is known to increase the production of various cell adhesion molecules such as intercellular adhesion molecule-1, vascular cell adhesion molecule-1, and platelet endothelial cell adhesion molecule [10].
We believe that the untreated severe anemia in our patient is a major potentiating factor for thrombosis. Anemia has long been considered a thrombogenic factor [7] and a frequent complication of IBD [23]. We performed a literature review search using PubMed and Google Scholar for English-language articles from 2006 to 2020, using the key terms “cerebral venous thrombosis”, “anemia”, and “iron deficiency anemia”. The main exclusion criteria used were (1) lack of anemia, (2) lack of CVT, (3) lack of CVT on neuroimaging, and (4) articles lacking sufficient data. The patient demographic data, clinical features, location of the CVT, diagnostic neuroimaging, and types of anemia are outlined in Table 4, which summarizes 21 case reports and 1 case series discussing 22 patients. Additionally, there are 5 case-control studies including a total of 222 patients and 1 cohort study of 51 patients. We also included 1 review of literature with 30 patients (with overlap of 1 case report in our table) that primarily focuses on abnormal prothrombotic laboratory findings [24].
The International Study on Cerebral Vein and Dural Sinus Thrombosis found the correlation of anemia with CVT to be 9.2% [6], which was confirmed by Stolz et al for severe anemia (Hb <9 g/dL; 11.7% of cases, odds ratio [OR] 1.10) [8]. A stronger association between CVT and anemia (27% of cases, OR 4.4%) was later established by Coutinho et al in 2015 [11]. This pivotal relationship has recently been proven again in a large retrospective study of 6 million patients that identified 36,327 patients with iron deficiency anemia [25]. The risk of thrombosis in patients with iron deficiency anemia without thrombocytosis was 7.8%, which increased to 15.8% in patients with iron deficiency anemia and thrombocytosis [25].
Both microcytic and macrocytic anemias have been implicated as a cause of CVT in patients with IBD [11]. In patients with UC, there is a higher prevalence of iron deficiency anemia than of macrocytic anemia, both of which occur secondary to hematochezia, malabsorption, and chronic disease activity status [26]. The pathogenesis for CVT in microcytic anemia comprises 4 pathways: (1) reactive thrombocytosis due to molecular mimicry, (2) alteration in erythrocyte viscosity causing turbulent blood flow, (3) anemic hypoxia resulting in increased production of cytokines, and (4) low serum ferritin leading to increase production of factor VIII [9,27] (Figure 9). Macrocytic anemia, because of vitamin B12 and folate malabsorption in IBD, leads to homocysteinemia, an established risk factor for CVT [11,22]. Coutinho et al reported that 1.4% of patients (OR 8.9) with CVT had macrocytic anemia [11]. Homocysteine has been shown to increase endothelial inflammation by increasing production of monocyte chemoattractant protein-1 and vascular cell adhesion molecule-1 as well as phosphorylation of p38. These changes cause an additional number of T cells and monocytes to adhere to the endothelium [16] (Figure 9). We encourage physicians to be aware of the fact that anemia has been strongly advocated as a prothrombotic factor, as it is seemingly overlooked in the clinical setting. Additionally, the interplay between the thrombogenic nature of IBD and anemia in the pathogenesis of CVT is important to acknowledge.
An average delay of 7 days in diagnosis of CVT can occur owing to a myriad of nonspecific clinical symptoms, although establishing the diagnosis may take up to several weeks [6]. The most common presentation is headache (90%) [1], which can progress over time and characteristically presents as a diffuse, severe pressure-type headache or a thunderclap headache [28–30]. The headache can be accompanied by other neurological features such as vomiting (29.2%), altered mental status (21.5%), papilledema (7.1%) [19], or diplopia following sixth nerve palsy [1], secondary to an increase in intracranial pressure (false localizing sign). Focal neurological deficits as a consequence of venous infarcts were seen in approximately 53% of patients [3,6,8,11,29], and focal or generalized seizures can occur in up to 40% of patients [1,2].
The aforementioned diverse presentation makes the diagnosis of CVT challenging, particularly when headache is the only presenting symptom. A diagnosis of CVT should be considered in young adults with IBD who present with a new-onset headache [18,31,32]. Initial workup including a complete blood count, basic metabolic panel, coagulation screen (bleeding time, prothrombin time, activated partial thromboplastin time), liver function tests, and inflammatory markers (erythrocyte sedimentation rate and C-reactive protein) should be followed by a D-dimer level, a hypercoagulability panel, and neurovascular imaging.
The use of D-dimer as a predictor of CVT was initially proposed by the European Stroke Organization in 2017 and has been endorsed by the European Academy of Neurology [32]. This is supported by the fact that D-dimer has a sensitivity of 97.8%, specificity of 84.9%, and negative predictive value of 99.8% for CVT [33]. A limitation to this suggestion, as raised by the European Stroke Organization, is that the reliability of D-dimer varies with time (symptoms lasting >1 week), thrombus load (number of sinuses involved), and clinical presentation [32]. D-dimer sensitivity decreases to 87.1%, especially in the presence of isolated headache [32–34]. Heldner et al recently introduced a new clinical scoring system (Tables 5, 6), based on D-dimer levels and risk stratification, for the pursuance of neurovascular imaging in CVT diagnosis [29].
Estimating the probability of CVT using this scoring system is based on individually weighted criteria (Table 5). The calculated score, in conjunction with D-dimer levels, yields different probabilities of CVT (low, moderate, high) (Table 6). D-dimer showed an effective exclusion of CVT when patients presented with low clinical probability (0–2 points) and D-dimer <500 ug/L. Moderate probability (3–5 points) and high probability (6–12 points) scores are associated with a lower reliability of D-dimer, and clinicians should pursue neurovascular imaging to diagnose CVT, irrespective of D-dimer levels.
Additionally, in this clinical score, thrombophilia has been defined as any hematological disorder related to coagulopathy [29]. Although the association between anemia and thrombosis is well established [6,8,11,25], it is underrecognized in clinical practice. Hence, taking anemia (thrombophilia) and the duration of symptoms (> 6 days) into account, our patient had a CVT clinical score of 6, indicating high probability. Thus, in our patient, this should have prompted further neuroimaging on the first ED visit, potentially avoiding the missed diagnosis and progression to further complications.
A diagnosis of CVT is best confirmed by CT venography or MR venography [1,32]. Non-contrast head CT has a low sensitivity in diagnosing CVT (30%), but can be utilized to recognize complications of CVT [1]. Infrequent signs seen on non-contrast head CT include the dense triangle/dense clot sign and the cord sign [2,35,36]. The classic finding of empty delta sign on contrast-enhanced head CT has low sensitivity [1]. Contrast-enhanced and 2-dimensional time-of-flight MR venography are helpful in detecting well-established criterion standard noninvasive diagnostic tests for detecting CVT. They have high sensitivity in detecting CVT during the acute, sub-acute, and chronic phases. CT venography has a 95% sensitivity [37]. When CT venography or MR venography findings are inconclusive, cerebral angiography is needed to confirm the diagnosis of CVT [35,38,39]. Digital subtraction angiography can indicate intraluminal-filling defects, signs of venous congestion, and absence of cortical veins or dural sinuses [39].
The acute treatment of CVT involves anticoagulation with therapeutic doses of low-molecular-weight heparin or unfractionated heparin [1,22,40]. The risks and benefits of either option should be weighed and applied individually. The criterion standard, long-term treatment of CVT is warfarin, with a target INR of 2.0–3.0 [1,40]. In patients with IBD-induced CVT, transient hypercoagulability requires 3 months of anticoagulation treatment. In patients with an isolated episode of idiopathic CVT, 6 to 12 months of treatment is required. Recurrent episodes of CVT (2.2% with treatment) [41] or anticoagulant deficiency necessitate indefinite treatment [19]. Direct oral anticoagulants have been increasingly used as an alternative to warfarin for the chronic management of CVT because they show a comparable efficacy and an improved safety profile [42].
The indications for endovascular intervention include clinical deterioration despite anticoagulation, progressive/severe neurological deficits, seizures, stupor, posterior fossa/deep-cerebral venous system involvement, and coma and patients who have contraindications to anticoagulation therapy [39]. A combination of interventional techniques, including local thrombolysis, direct aspiration thrombectomy, stent retriever thrombectomy, balloon thrombectomy, balloon angioplasty, stenting, and rheolytic thrombectomy, may be required for revascularization [39]. The Thrombolysis or Anticoagulation for Cerebral Venous Thrombosis randomized control trial further suggests that neurointervention may not improve functional outcomes of patients with CVT, as compared with standard clinical care [43]. There are currently no consensus guidelines for these interventional procedures [39]. Invasive interventions, including decompressive craniectomy and hematoma evacuation, should be reserved for patients with intractable intracranial hypertension or large parenchymal lesions leading to herniation [39].
Early neurological complications of CVT include hemorrhagic strokes (40%), intracranial hypertension (40%), communicating hydrocephalus (6.6%), and status epilepticus [1]. Late complications include chronic headaches, which should be evaluated for recurring CVT, seizures (5–32%), vision loss (2–4%), and dural arteriovenous fistula formation (1–3%), secondary to persistent occlusion [1]. Saposnik et al showed that with appropriate treatment, 79% of patients have complete recovery, while 10.4% experience persistent mild to moderate disability (modified Rankin Scale [mRS] score 2–3), and 2.2% have severe disability (mRS score 4–5) [1]. It is imperative to note that the overall mortality rate for CVT alone is 5.6% to 9.4% [36]; however, CVT in conjunction with UC, has a mortality rate as high as 25% [6].
Conclusions
Cerebral venous thrombosis should be investigated in patients with IBD who report new-onset or worsening headaches, especially in the context of underlying anemia. Diagnosing CVT can be challenging owing to a nonspecific clinical presentation and lack of diagnostic laboratory markers. Establishing a timely diagnosis of CVT is critical in patients with IBD, given the high mortality rate. The current American Heart Association/ American Stroke Association (AHA/ASA) update regarding the diagnosis and management of CVT was published in 2011 [1]. Heldner et al [29] recently developed a clinical probability score to guide in the pursuance of diagnostic neurovascular imaging. This score can better assist in the risk stratification of patients presenting to a clinician, especially in the acute setting, analogous to the Wells’ criteria for deep vein thrombosis and pulmonary embolism. We endorse implementing the CVT clinical probability score in future AHA/ASA recommendations for CVT diagnosis. Additionally, clinicians should be aware that anemia is a major risk factor for CVT in patients with UC.
Figures
Tables
Table 1.. Patient laboratory findings on first Emergency Department visit.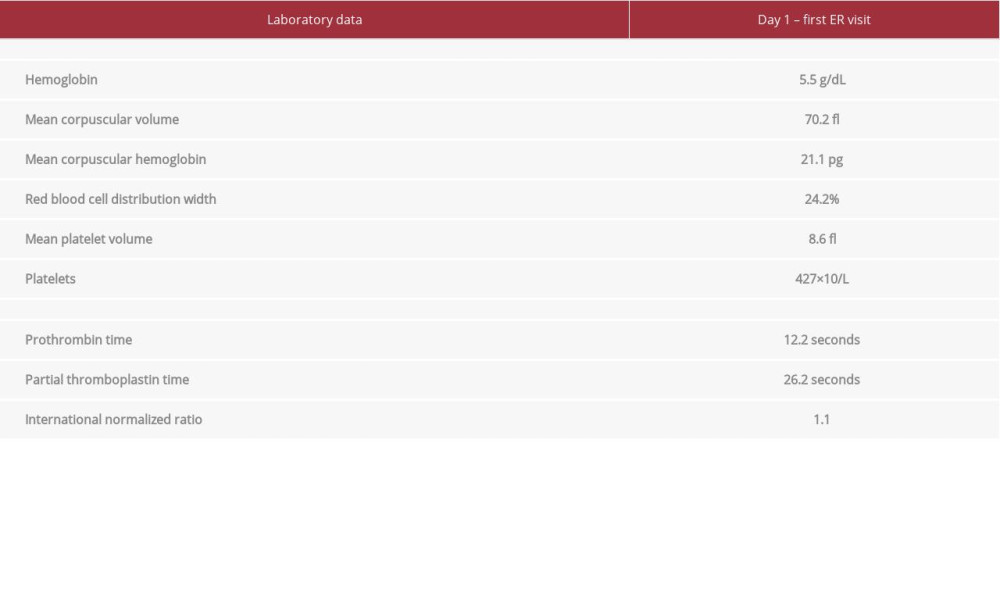 Table 2.. Patient laboratory findings on second Emergency Department visit and admission.
Table 2.. Patient laboratory findings on second Emergency Department visit and admission.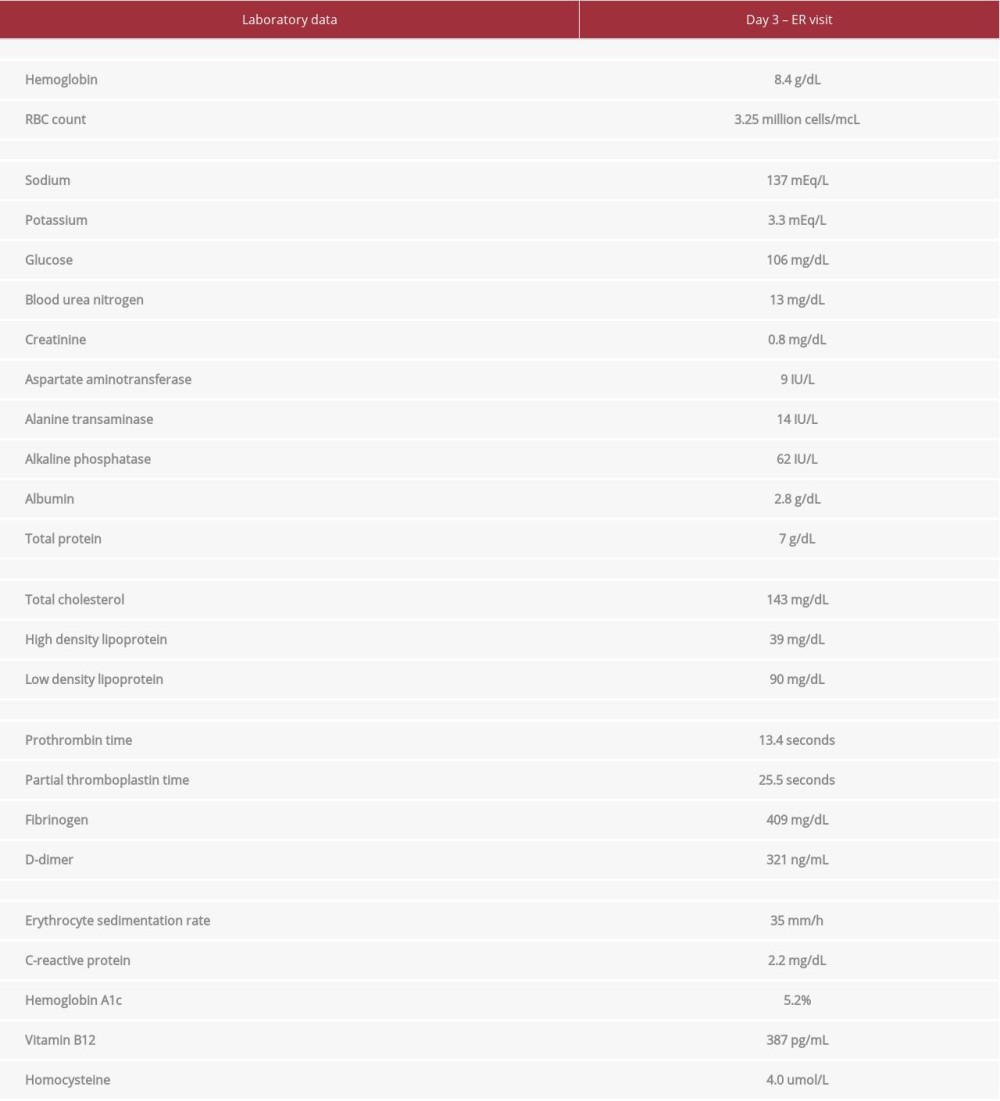 Table 3.. Neurological complications of inflammatory bowel disease.
Table 3.. Neurological complications of inflammatory bowel disease.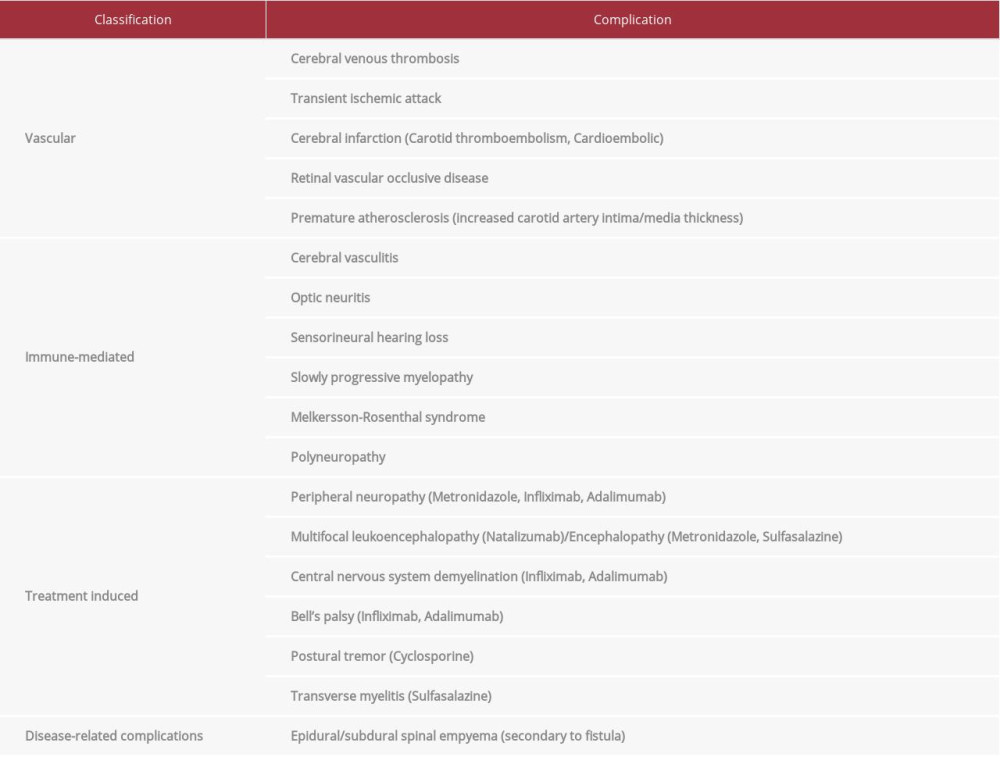 Table 4.. Review of literature: Cases with cerebral venous thrombosis and anemia.
Table 4.. Review of literature: Cases with cerebral venous thrombosis and anemia.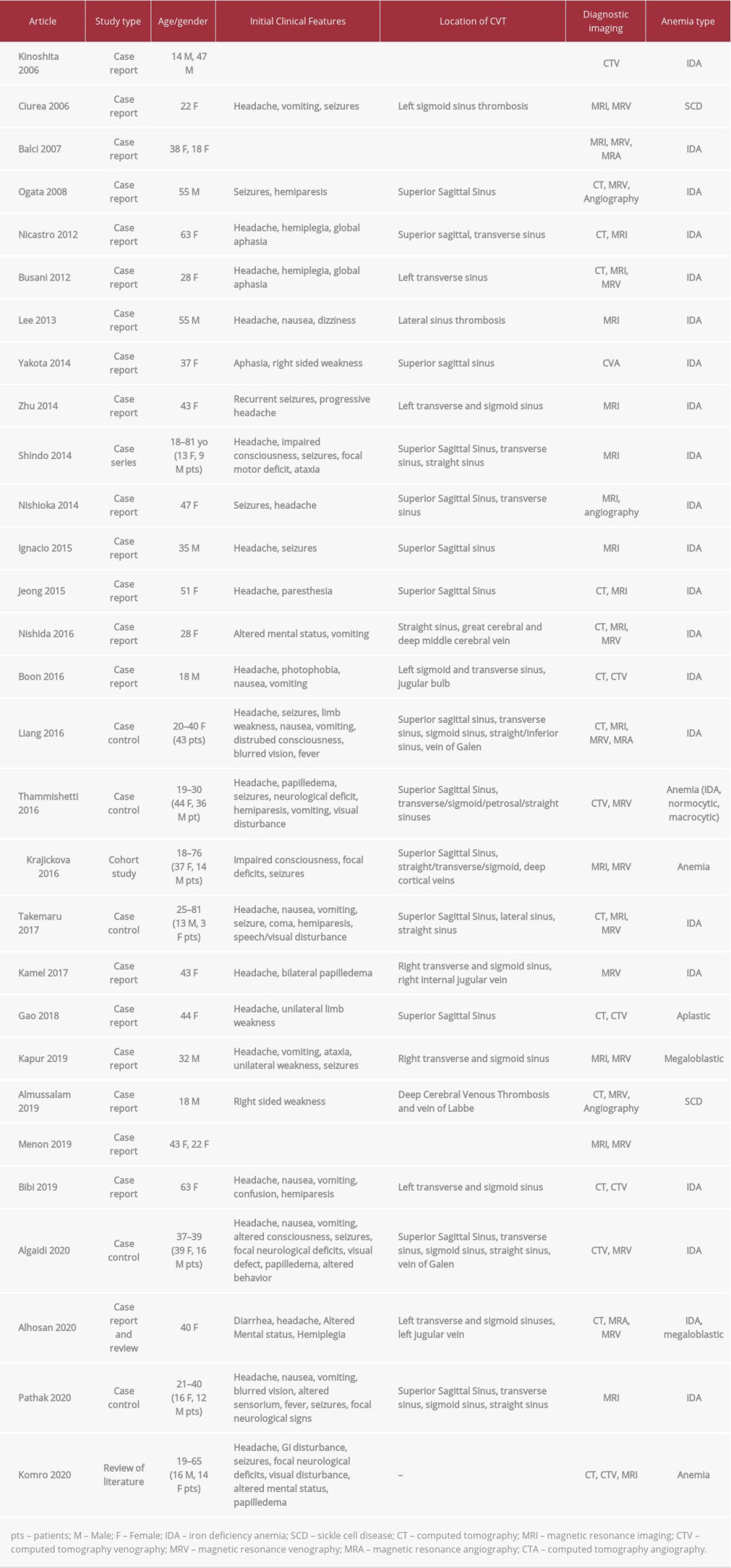 Table 5.. Clinical scoring based on presentation [modified with permission from Heldner, 2020].
Table 5.. Clinical scoring based on presentation [modified with permission from Heldner, 2020].![Clinical scoring based on presentation [modified with permission from Heldner, 2020].](https://jours.isi-science.com/imageXml.php?i=t5-amjcaserep-22-e932123.jpg&idArt=932123&w=1000) Table 6.. Probability groups and D-dimer levels [modified with permission from Heldner, 2020].
Table 6.. Probability groups and D-dimer levels [modified with permission from Heldner, 2020].![Probability groups and D-dimer levels [modified with permission from Heldner, 2020].](https://jours.isi-science.com/imageXml.php?i=t6-amjcaserep-22-e932123.jpg&idArt=932123&w=1000)
References:
1.. Saposnik G, Barinagarrementeria F, Brown RD, Diagnosis and management of cerebral venous thrombosis: A statement for healthcare professionals from the American Heart Association/American Stroke Association: Stroke, 2011; 42(4); 1158-92
2.. Alvis-Miranda HR, Milena Castellar-Leones S, Alcala-Cerra G, Rafael Moscote-Salazar L, Cerebral sinus venous thrombosis: J Neurosci Rural Pract, 2013; 4(4); 427-38
3.. Guenther G, Arauz A, Cerebral venous thrombosis: A diagnostic and treatment update: Neurologia, 2011; 26(8); 488-98
4.. Abdalla AO, Alluri D, Hassaballa M, A case of cerebral venous sinus thrombosis presenting during relapse of ulcerative colitis: Am J Case Rep, 2019; 20; 419-22
5.. Patel P, Hasan A, Al Shathir M, ischemic stroke at the time of diagnosis of ulcerative colitis: Tenn Med E-J, 2015; 1(4) Article 11
6.. Ferro JM, Canhão P, Stam J, Prognosis of cerebral vein and dural sinus thrombosis: Results of the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT): Stroke, 2004; 35(3); 664-70
7.. Richardson FL, Effect of severe hemorrhage on the number of blood plates in blood from the peripheral circulation of rabbits: J Med Res, 1904; 13(1); 99-103
8.. Stolz E, Valdueza JM, Grebe M, Anemia as a risk factor for cerebral venous thrombosis? An old hypothesis revisited. Results of a prospective study: J Neurol, 2007; 254(6); 729-34
9.. Franchini M, Targher G, Montagnana M, Lippi G, Iron and thrombosis: Ann Hematol, 2008; 87(3); 167-73
10.. Zezos P, Kouklakis G, Saibil F, Inflammatory bowel disease and thromboembolism: World J Gastroenterol, 2014; 20(38); 13863-78
11.. Coutinho JM, Zuurbier SM, Gaartman AE, association between anemia and cerebral venous thrombosis: Case-control study: Stroke, 2015; 46(10); 2735-40
12.. White H, Gastrointestinal disorders and the nervous system: Continuum (Minneap Minn), 2020; 26(3); 577-90
13.. Ferro JM, Neurologic manifestations of inflammatory bowel disease: Gastroenterol Hepatol (N Y), 2014; 10(9); 599-600
14.. Morís G, Inflammatory bowel disease: An increased risk factor for neurologic complications: World J Gastroenterol, 2014; 20(5); 1228-37
15.. Plata-Bello J, Acosta-Lopez S, Neurological manifestations of inflammatory bowel disease. new concepts in inflammatory bowel disease, Batool Mutar Mahdi: IntechOpen, 2018 https://www.intechopen.com/books/new-concepts-in-inflammatory-bowel-disease/neurological-manifestations-of-inflammatory-bowel-disease
16.. Owczarek D, Cibor D, Głowacki MK, Inflammatory bowel disease: Epidemiology, pathology and risk factors for hypercoagulability: World J Gastroenterol, 2014; 20(1); 53-63
17.. Cheng K, Faye AS, Venous thromboembolism in inflammatory bowel disease: World J Gastroenterol, 2020; 26(12); 1231-41
18.. Grainge MJ, West J, Card TR, Venous thromboembolism during active disease and remission in inflammatory bowel disease: A cohort study: Lancet, 2010; 375(9715); 657-63
19.. Katsanos AH, Katsanos KH, Kosmidou M, Cerebral sinus venous thrombosis in inflammatory bowel diseases: QJM, 2013; 106(5); 401-13
20.. Miehsler W, Reinisch W, Valic E, Is inflammatory bowel disease an independent and disease specific risk factor for thromboembolism?: Gut, 2004; 53(4); 542-48
21.. Harrison MJ, Truelove SC, Cerebral venous thrombosis as a complication of ulcerative colitis: Am J Dig Dis, 1967; 12(10); 1025-82
22.. Casella G, Cortelezzi CC, Marialuisa D, Cerebral arterial thrombosis in ulcerative colitis: Case Rep Gastrointest Med, 2013; 2013; 679147
23.. Crohn BB, Ginzburg L, Oppenheimer GD, Regional ileitis: A pathologic and clinical entity. 1932: Mt Sinai J Med, 2000; 67(3); 263-68
24.. Komro J, Findakly D, Cerebral venous sinus thrombosis in adults with prothrombotic conditions: A systematic review and a case from our institution: Cureus, 2020; 12(4); e7654
25.. Song AB, Kuter DJ, Al-Samkari H, Characterization of the rate, predictors, and thrombotic complications of thrombocytosis in iron deficiency anemia: Am J Hematol; 2020 [Online ahead of print]
26.. Kaitha S, Bashir M, Ali T, Iron deficiency anemia in inflammatory bowel disease: World J Gastrointest Pathophysiol, 2015; 6(3); 62-72
27.. Bibi A, Liyanapthirana C, Khan S, Rare presentation of iron deficiency anaemia with cerebral venous sinus thrombosis in a middle-aged woman: BMJ Case Rep, 2019; 12(1); bcr-2018225851
28.. Rizelio V, Sérgio Faro Santos P, Suspicion of cerebral venous thrombosis based on the patient’s headache characteristics: Neurology, 2018; 15(Suppl.); 1.207
29.. Heldner MR, Zuurbier SM, Li B, Prediction of cerebral venous thrombosis with a new clinical score and D-dimer levels: Neurology, 2020; 95(7); e898-909
30.. de Bruijn SF, Stam J, Kappelle LJ, Thunderclap headache as first symptom of cerebral venous sinus thrombosis. CVST Study Group: Lancet, 1996; 348(9042); 1623-25
31.. Bargen JA, Barker NW, Extensive arterial and venous thrombosis complicating chronic ulcerative colitis: Arch Intern Med, 1936; 58(1); 17-31
32.. Ferro JM, Bousser MG, Canhão P, European Stroke Organization guideline for the diagnosis and treatment of cerebral venous thrombosis – Endorsed by the European Academy of Neurology: Eur Stroke J, 2017; 2(3); 195-221
33.. Alons IM, Jellema K, Wermer MJ, Algra A, D-dimer for the exclusion of cerebral venous thrombosis: A meta-analysis of low risk patients with isolated headache: BMC Neurol, 2015; 15; 118
34.. Kosinski CM, Mull M, Schwarz M, Do normal D-dimer levels reliably exclude cerebral sinus thrombosis?: Stroke, 2004; 35(12); 2820-25
35.. Dmytriw AA, Song JSA, Yu E, Poon CS, Cerebral venous thrombosis: state of the art diagnosis and management: Neuroradiology, 2018; 60(7); 669-85
36.. Idiculla PS, Gurala D, Palanisamy M, Cerebral venous thrombosis: A comprehensive review: Eur Neurol, 2020; 83(4); 369-79
37.. Chiewvit P, Piyapittayanan S, Poungvarin N, Cerebral venous thrombosis: Diagnosis dilemma: Neurol Int, 2011; 3(3); e13
38.. Stam J, Thrombosis of the cerebral veins and sinuses: N Engl J Med, 2005; 352(17); 1791-98
39.. Lee SK, Mokin M, Hetts SW, Current endovascular strategies for cerebral venous thrombosis: Report of the SNIS Standards and Guidelines Committee: J Neurointerv Surg, 2018; 10(8); 803-10
40.. Ferro JM, Coutinho JM, Dentali F, Safety and efficacy of dabigatran etexilate vs dose-adjusted warfarin in patients with cerebral venous thrombosis: A randomized clinical trial: JAMA Neurol, 2019; 76(12); 1457-65
41.. Leach JL, Wolujewicz M, Strub WM, Partially recanalized chronic dural sinus thrombosis: Findings on MR imaging, time-of-flight MR venography, and contrast-enhanced MR venography: AJNR Am J Neuroradiol, 2007; 28(4); 782-89
42.. Lee GKH, Chen VH, Tan CH, Comparing the efficacy and safety of direct oral anticoagulants with vitamin K antagonist in cerebral venous thrombosis: J Thromb Thrombolysis, 2020; 50(3); 724-31
43.. Coutinho JM, Ferro JM, Zuurbier SM, Thrombolysis or anticoagulation for cerebral venous thrombosis: rationale and design of the TO-ACT trial: Int J Stroke, 2013; 8(2); 135-40
Figures
Tables
 Table 1.. Patient laboratory findings on first Emergency Department visit.
Table 1.. Patient laboratory findings on first Emergency Department visit. Table 2.. Patient laboratory findings on second Emergency Department visit and admission.
Table 2.. Patient laboratory findings on second Emergency Department visit and admission. Table 3.. Neurological complications of inflammatory bowel disease.
Table 3.. Neurological complications of inflammatory bowel disease. Table 4.. Review of literature: Cases with cerebral venous thrombosis and anemia.
Table 4.. Review of literature: Cases with cerebral venous thrombosis and anemia.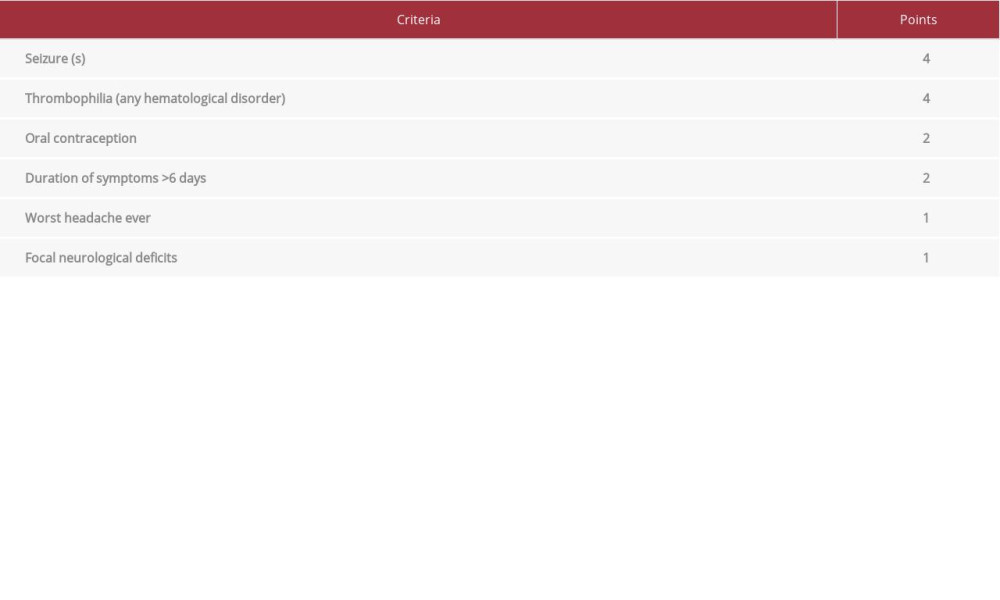 Table 5.. Clinical scoring based on presentation [modified with permission from Heldner, 2020].
Table 5.. Clinical scoring based on presentation [modified with permission from Heldner, 2020].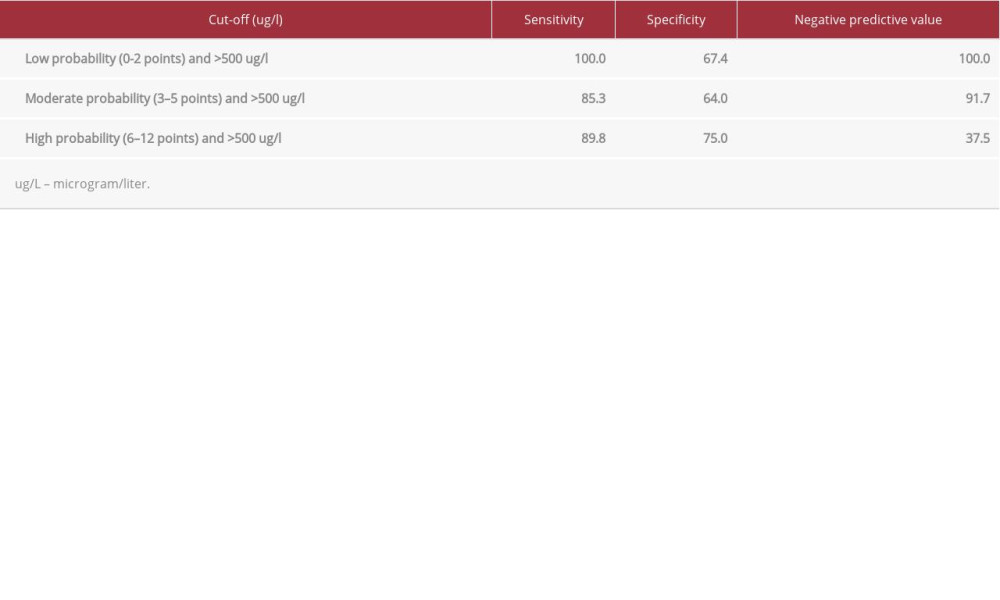 Table 6.. Probability groups and D-dimer levels [modified with permission from Heldner, 2020].
Table 6.. Probability groups and D-dimer levels [modified with permission from Heldner, 2020]. Table 1.. Patient laboratory findings on first Emergency Department visit.
Table 1.. Patient laboratory findings on first Emergency Department visit. Table 2.. Patient laboratory findings on second Emergency Department visit and admission.
Table 2.. Patient laboratory findings on second Emergency Department visit and admission. Table 3.. Neurological complications of inflammatory bowel disease.
Table 3.. Neurological complications of inflammatory bowel disease. Table 4.. Review of literature: Cases with cerebral venous thrombosis and anemia.
Table 4.. Review of literature: Cases with cerebral venous thrombosis and anemia. Table 5.. Clinical scoring based on presentation [modified with permission from Heldner, 2020].
Table 5.. Clinical scoring based on presentation [modified with permission from Heldner, 2020]. Table 6.. Probability groups and D-dimer levels [modified with permission from Heldner, 2020].
Table 6.. Probability groups and D-dimer levels [modified with permission from Heldner, 2020]. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








