19 July 2021: Articles 
A Rare Cause of Chronic Liver Disease Diagnosed by Endoscopic Ultrasound-Guided Liver Biopsy
Challenging differential diagnosis, Rare disease
Nourah A. Alajeel1AEF, Mohammad ShehabDOI: 10.12659/AJCR.933002
Am J Case Rep 2021; 22:e933002
Abstract
BACKGROUND: Elevated liver enzymes is a common clinical problem with many possible etiologies, yet some are rare and can be missed. Patients with sickle cell disease (SCD) may be at risk of liver disease due to recurrent blood transfusion predisposing to viral hepatitis. Furthermore, recurrent transfusions can increase the risk of iron overload, which can create deposits in the liver, eventually resulting in chronic liver disease. Liver biopsy is an essential tool to establish a diagnosis of liver disease in many patients with unexplained elevation of liver enzymes. Recently, endosocpic ultrasound (EUS)-guided liver biopsy has been shown to be safe and effective in obtaining adequate liver tissue. However, the safety and efficacy has not been established in patients with SCD.
CASE REPORT: A 59-year-old man with SCD and beta-thalassemia minor was evaluated for persistently elevated liver enzymes (mainly cholestatic). He had a background history of treated hepatitis C virus infection. He had multiple blood transfusions in the past for sickle cell crisis. A diagnostic work-up revealed negative viral and autoimmiune serology and no evidence of biliary obstruction on abdominal imaging. The iron profile was elevated, consistent with iron overload. An EUS-guided liver biopsy confirmed a diagnosis hepatic hemosiderosis secondary to long-term blood transfusions.
CONCLUSIONS: This report emphasizes the importance of careful monitoring of iron levels in patients with hematological conditions requiring long-term blood transfusions. In addition, it highlights the emerging role of EUS-guided liver biopsy as a safe and accurate alternative to percutaneous liver biopsy.
Keywords: Anemia, Sickle Cell, Endoscopic Ultrasound-Guided Fine Needle Aspiration, Endosonography, Hemosiderosis, Image-Guided Biopsy, Iron Overload, Liver, Liver Diseases
Background
Patients with hemoglobinopathies frequently require multiple blood transfusions during their lifespan. This places them at increased risk of a number of blood transfusion-related hepatic complications, including viral hepatitis and iron overload syndromes. In addition, some hemoglobinopathies such as sickle cell disease (SCD) are associated with certain liver diseases, including sickle cell hepatopathy, SCD cholangiopathy, cholelithiasis, and choledocholithiasis [1,2]. Liver biopsy is considered the criterion standard test to diagnose the etiology of liver disease. This has traditionally been performed via the percutaneous or trans-jugular routes. However, percutaneous liver biopsy has been associated with increased risk of adverse events in patients with SCD, including bleeding and death [3,4]. Recently, endoscopic ultrasound (EUS)-guided liver biopsy has been introduced and was shown to have equal diagnostic accuracy as the percutaneous approach [5]. However, the efficacy and safety of EUS-guided liver biopsy has not been tested in patients with SCD. This case report describes a patient with SCD who presented with unexplained elevated liver enzymes, with the diagnosis being established with the help of EUS-guided liver biopsy.
Case Report
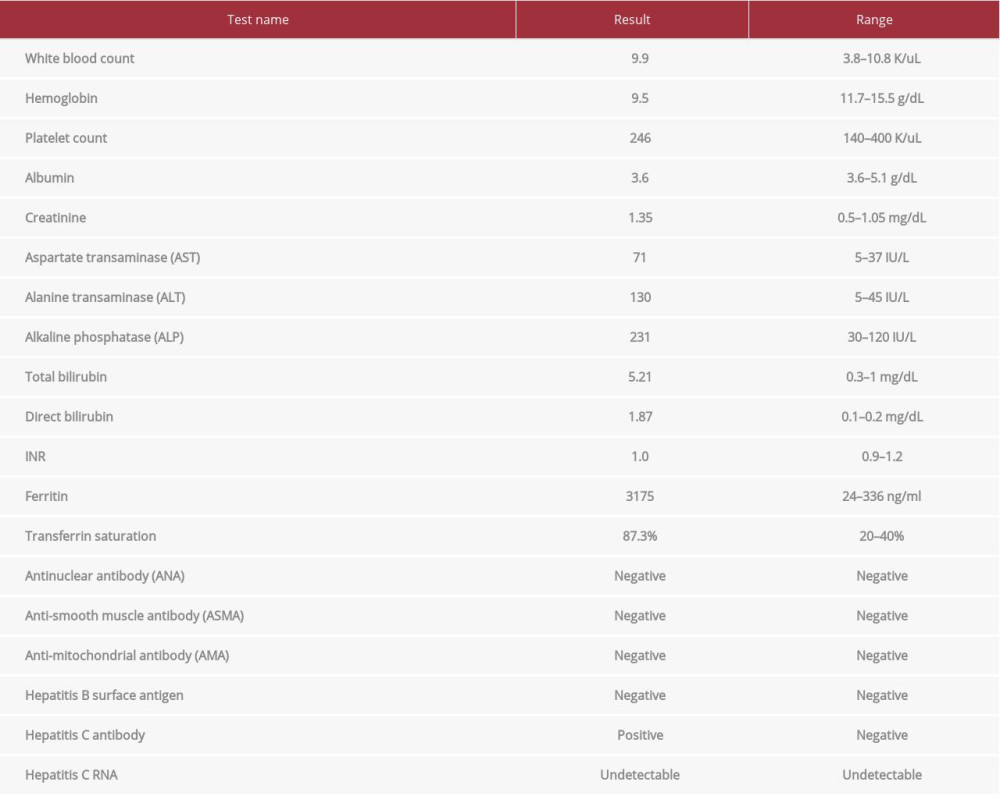
A 59-year-old man was referred to the liver clinic for evaluation of elevated liver enzymes. His past medical history was significant for sickle cell disease, beta-thalassemia minor (HbS/beta), glucose-6-phosphate dehydrogenase (G6PD) deficiency, hyper-tension, subclinical hypothyroidism, and laparoscopic cholecystectomy in 2000 for pigment gallbladder stones. In addition, he had a history of hepatitis C virus (HCV) infection that was treated in the past with interferon-based therapy with successful viral eradication. He did not consume tobacco or alcohol. The patient had a long history of multiple hospital admissions for acute hemolytic crises for which he was transfused massive amounts of packed red blood cells (PRBC), and he had received around 10–15 units of PRBC yearly for the past 15 years. During his hospital stays and during clinic follow-up, he was noted to have persistently elevated liver enzymes. His most recent blood test during routine follow-up showed a predominately cholestatic picture with an R-factor of 1.5 (Table 1). An abdominal ultrasound revealed an enlarged liver with nodular contour, with no focal lesions or biliary tree abnormalities. He underwent further investigations to diagnose his liver disease. Serological work-ups for hepatitis B, hepatitis C (including HCV RNA), and autoimmune hepatitis were unremarkable. His iron profile showed elevated transferrin saturation (87.3%) and ferritin (3175 ng/mL). Genetic testing for hereditary hemochromatosis was negative. Magnetic resonance cholangiopancreatography
The patient was suspected to have iron overload syndrome (based on elevated ferritin and transferrin saturation); however, given the non-conclusive imaging results and the persistent cholestatic picture of elevation in liver enzyme that is atypical for iron overload syndrome, a liver biopsy was scheduled. An endoscopic ultrasound (EUS)-guided liver biopsy was performed using 19G fine-needle aspiration (FNA) targeting the left liver lobe from the stomach (Figure 1). The procedure was done under conscious sedation and the patient had no post-procedure adverse events. Endoscopic assessment did not show any esophageal varices.
The liver biopsy showed evidence of extensive vaculation and cytoplasmic cleaning associated with extensive iron deposition, predominantly in Kupffer cells. There was mild chronic inflammation in the portal tracts without associated lobular inflammation, and mild steatosis was seen in the hepatocytes. Reticulin and trichrome stains showed pericellular and bridging fibrosis with occasional regenerative nodules (Figure 2). The overall picture was consistent with grade 4 hemosiderosis (modified Scheuer’s system). The patient was diagnosed with hepatic hemosiderosis secondary to long-term blood transfusions and iron overload, and was started on iron chelation therapy (Deferasirox tablets).
Discussion
Sickle cell disease (SCD) poses many risks to the liver, and the mechanism of injury can be multifactorial. Patients with SCD often have a higher chance of receiving multiple blood transfusions throughout their lifetime, which can result in the possibility of transfusional iron overload [1,2]. Iron accumulation in the liver can ultimately result in hepatic hemosidrosis. This is characterized by iron deposition in Kupffer cells, which helps to distinguish it from hereditary hemochromatosis, in which iron tends to deposit in the hepatocytes [2].
The diagnosis of hepatic hemosiderosis is typically established in a patient with risk factors who is found to have elevated liver enzymes with evidence of increased iron stores biochemically and by imaging using magnetic resonance imaging (MRI)-based estimate of liver iron content. In situations where MRI is not available, percutaneous liver biopsy is usually required to confirm the diagnosis and quantify iron stores. However, percutaneous liver biopsy has been associated with serious complications in patients with SCD. In a retrospective study in patients with SCD who underwent percutaneous liver biopsy, the risk of bleeding was high (36%) among patients with acute sickle cell hepatopathy [3]. Therefore, patients with SCD are considered to be at high risk for complications after percutaneous liver biopsy, and a safer option is required in this population [4].
Recently, EUS-guided liver biopsy has been introduced and shown to provide high diagnostic yield coupled with an excellent safety profile. EUS-guided liver biopsy has been mostly used in patients with unexplained elevation of liver enzymes in whom histological assessment of the liver is warranted. Specifically, the EUS approach has been preferred when endoscopic assessment is needed to rule out portal hypertension-related complications (eg, esophageal and gastric varices, portal hypertensive gastropathy). Furthermore, EUS offers the advantage of ruling out obstructive biliary lesion (eg, stones or malignancy) before performing EUS-guided liver biopsy, since these lesions can occasionally be missed by other imaging modalities [5,6]. The EUS approach has been increasing in popularity since it has been shown to have a high rate of successful histological diagnosis (93.9%) coupled with a low risk of overall adverse events (2.3%) [5]. When compared to the percutaneous and trans-jugular approaches, EUS-guided biopsy has been shown to have equivalent diagnostic yield [7]. EUS-guided liver biopsy provides further advantages over the other approaches by allowing endoscopic evaluation of esophageal and gastric varices in patients with suspected cirrhosis, and it allows access to both liver lobes for biopsy. In addition, EUS-guided biopsy is usually well-tolerated using conscious sedation, has shorter recovery time, and is associated with lower post-procedure pain [8]. In our patient, we elected to proceed with liver biopsy given the uncertainty with the diagnosis since the elevation of his liver enzymes was mostly cholestatic yet he had no evidence of biliary pathology on imaging and had biochemical evidence of iron overload. The EUS approach was chosen since it provided the opportunity to screen for esophageal varices endoscopically as the patient had evidence of advance fibrosis shown by transient elastography, and it avoided the percutaneous approach, which had been shown have higher risk in patients with SCD.
To the best of our knowledge, this is the first case report to demonstrate the safety and efficacy of EUS-guided liver biopsy in establishing the diagnosis of hepatic hemosiderosis in a patient with SCD. The liver biopsy was able to establish a definite diagnosis and allowed staging of the disease histologically.
Conclusions
Patients with SCD have increased risk of multiple liver disease, including hepatic iron overload, and accurate diagnosis is essential. EUS-guided liver biopsy offers a safe and effective alternative to percutaneous liver biopsy to establish the diagnosis and should be strongly considered as the first-line technique for biopsy in patients with SCD given the increased risk associated with percutaneous liver biopsy in this population.
Figures
References:
1.. Koullapis N, Kouroupi IG, Dourakis SP, Hepatobiliary manifestations of sickle cell disease: Haema, 2005; 8; 393-404
2.. Batts KP, Iron overload syndromes and the liver: Mod Pathol, 2007; 20(Suppl.1); S31-39
3.. Zakaria N, Knisely A, Portmann B, Acute sickle cell hepatopathy represents a potential contraindication for percutaneous liver biopsy: Blood, 2003; 101; 101-3
4.. Machado NO: Complications of liver biopsy – risk factors, management and recommendations, liver , 2011, InTech 978-953-307-644-7
5.. Mohan BP, Shakhatreh M, Garg R, Efficacy and safety of EUS-guided liver biopsy: A systematic review and meta-analysis: Gastrointest Endosc, 2019; 89; 238-46
6.. Johnson KD, Laoveeravat P, Yee EU, Endoscopic ultrasound guided liver biopsy: Recent evidence: World J Gastrointest Endosc, 2020; 12(3); 83-97
7.. Pineda JJ, Diehl DL, Miao CL, EUS-guided liver biopsy provides diagnostic samples comparable with those via the percutaneous or transjugular route: Gastrointest Endosc, 2016; 83; 360-65
8.. Diehl DL, Endoscopic ultrasound-guided liver biopsy: Gastrointest Endosc Clin N Am, 2019; 29; 173-86
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133