14 February 2022: Articles 
Guillain-Barré Syndrome After Ad26.COV2.S Vaccination
Unknown etiology, Unusual clinical course, Challenging differential diagnosis, Educational Purpose (only if useful for a systematic review or synthesis), Rare coexistence of disease or pathology
Hein Linn Thant1ABCDEFG*, Richard Morgan1ABCDEFG, Mario M. Paese1BCDG, Trevor Persaud1CDEG, Jose Diaz1EFG, Lina Hurtado12ABCGDOI: 10.12659/AJCR.935275
Am J Case Rep 2022; 23:e935275
Abstract
BACKGROUND: Since December 2020, multiple vaccines have mobilized mass immunization campaigns capable of mitigating the current SARS-COV-2 pandemic. Ad26.COV2.S (Johnson & Johnson/Janssen) is a recombinant, replication-incompetent vector vaccine encoding the SARS-CoV-2 spike (s) protein and is especially protective against severe-critical disease. It is a single-dose injection; adverse effects after vaccine administration are usually mild and self-limited, including pain at the injection site, headache, fatigue, muscle aches, and nausea. Severe adverse events involving hospitalization and death after Ad26.COV2.S rarely occur. However, not unlike previous viral vector vaccines, ongoing clinical trials may unveil rare complications of Ad26.COV2.S. Guillain-Barré Syndrome (GBS) is an autoimmune demyelinating polyneuropathy that can potentially manifest severe neurological symptoms after vaccination.
CASE REPORT: This report describes a case of classic GBS features that manifested 14 days after a single Ad26.COV2.S vaccine injection. The patient developed flaccid paralysis with treatment-related fluctuations. Our findings warrant further investigation into the potential relationship between SARS-CoV-2 vaccinations and the development of GBS.
CONCLUSIONS: A temporal association between the Ad26.COV2.S (Johnson & Johnson/Janssen) vaccine and the onset of GBS was demonstrated in this case report. A feasible underlying pathogenic mechanism involves the cross-reactivity of antibodies stimulated by adenovirus vaccine components and peripheral nerve glycoproteins. However, there is currently insufficient evidence to support a causal relationship between Ad26.COV2.S and the development of GBS. Further evidence gathered from clinician surveillance and clinical trials are needed to draw these conclusions.
Keywords: COVID-19, SARS-CoV-2, Guillain-Barré syndrome, vaccine, Ad26.COV2.S, Ad26COVS1, COVID-19, COVID-19 Vaccines, Guillain-Barre Syndrome, Humans, Vaccination
Background
Since December 2020, multiple vaccines have mobilized mass immunization campaigns capable of mitigating the current SARS-CoV-2 pandemic. Ad26.COV2.S (Johnson & Johnson/ Janssen) is a recombinant, replication-incompetent vector vaccine encoding the SARS-CoV-2 spike (s) protein and is especially protective against severe-critical disease [1]. It is a single-dose injection; adverse effects after vaccine administration are usually mild and self-limited, including pain at the injection site, headache, fatigue, muscle aches, and nausea [2,3]. Severe adverse events involving hospitalization and death after Ad26.COV2.S rarely occur. However, not unlike previous viral vector vaccines, ongoing clinical trials may unveil rare complications of Ad26.COV2.S. Guillain-Barré syndrome (GBS) is an immune-mediated polyradiculoneuropathy that is possibly a rare manifestation after vaccination. We report a case of GBS after Ad26.COV2.S vaccination refractory to conventional treatment and rehabilitation approaches.
Case Report
A 66-year-old man with diabetes mellitus, hypertension, and prostate cancer in complete remission presented to an acute care center after experiencing 3 days of bilateral lower extremity paresthesia and weakness. During that time, he developed lower limb paresthesia followed by weakness that spread symmetrically in a distal-to-proximal pattern. As a result, he was unable to ambulate despite being physically active prior to symptoms onset. He did not recount having difficulty with chewing, swallowing, breathing, bowel movements, or voiding. Additionally, he denied having facial weakness or paresthesia, vision changes, palpitations, and back or extremity pain. There was no recent history of travel, trauma, or gastrointestinal or respiratory symptoms. Further, he never tested positive for COVID-19. He received a single dose of the Ad26.COV2.S vaccination 14 days prior to hospital admission.
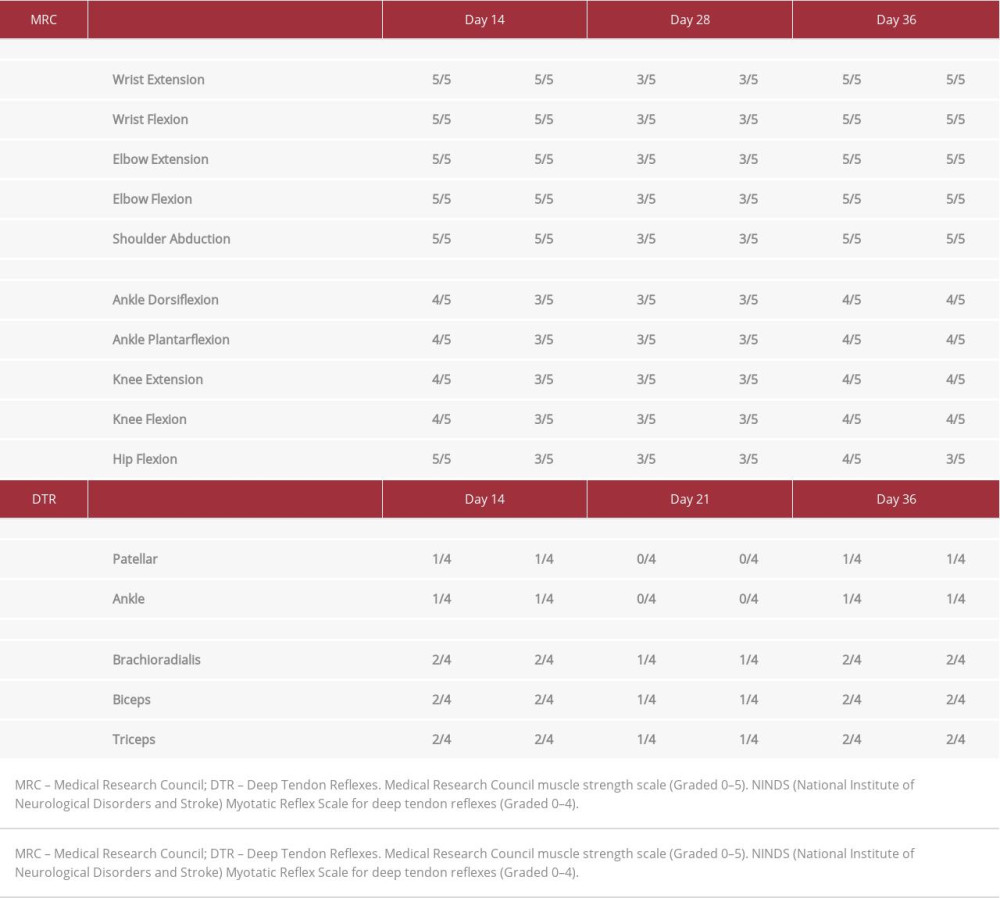
Upon hospital admission, neurological examination revealed decreased upper and lower extremity muscle strength, absent lower extremity deep tendon reflexes, and reduced upper and lower extremity sensory response to light touch (Table 1). He demonstrated flexor planter reflex and negative clonus on exam. Due to his lower extremity weakness, he was unable to participate in gait assessment. He remained conscious, oriented, and any without mental status changes throughout hospitalization. His vitals revealed intermittent hypotension and bradycardia, but he did not report related symptoms. Brain and whole-spine MRI findings were not indicative of acute pathology. Lumbar puncture results showed normal opening pressure, elevated cerebrospinal fluid (CSF) protein 75.0 mg/dL, albumin-cytological dissociation, and no growth of organisms. Chest radiography was unremarkable, and 12-lead EKG demonstrated normal sinus rhythm. The SARS-COV-2 RT-PCR result from the nasopharyngeal swab was negative. Complete blood count and comprehensive metabolic panel were within normal limits. Serologic polyneuropathy screening tests ruled out thiamine, folate and cyanocobalamin deficiency, Lyme disease, HIV, and syphilis. Taken together, these clinical findings pointed toward a diagnosis of GBS.
While in the acute care setting, intravenous immunoglobulins (IVIG) were administered at a dose of 2 mg/kg over the course of 5 days. His lower extremity muscle strength subsequently improved (Table 1), and he was transferred for in-patient rehabilitation. On day 15 of inpatient rehabilitation, he began to experience ascending bilateral upper extremity weakness and paresthesia. His lower extremity weakness simultaneously worsened (Table 1). Due to his acute neurological decline, he was transferred to an acute care hospital, where he underwent plasma exchange for 10 days. Once stabilized, he returned to the inpatient rehabilitation facility for 7 days. Sequential neurological examinations during this time did not indicate changes in motor or sensory function. Owing to his lack of progress in therapy and inability to perform activities of daily living, he is continuing medical management and therapy at a long-term care facility.
Discussion
Guillain-Barré syndrome (GBS) is a progressive immune-mediated polyradiculoneuropathy that classically presents with ascending symmetric flaccid limb weakness and areflexia. The presence of ataxia, dysautonomia, cranial nerve palsy, and paresthesia are also supportive of a diagnosis [4]. In most cases of GBS, an antecedent infection provokes autoantibodies directed against peripheral nerve components. The 3 major subtypes of GBS, classified by target antigen sites, are acute inflammatory demyelinating polyneuropathy (AIDP), acute motor axonal neuropathy (AMAN), and acute motor and sensory axonal neuropathy (AMSAN) [5,6]. Interestingly, an association between various pathogens and specific GBS subtypes has been identified [7]. Whereas pathogen-borne antigens are an established antecedent for GBS, it remains unclear if a causative relationship exists with vaccinations [8]. An increased risk for GBS has been described after influenza A, measles/mumps/ rubella (MMR), human papillomavirus (HPV), tetanus, hepatitis, polio, adenovirus, and quadrivalent meningococcal vacci-nations [9–11]. Similarly, a temporal association between GBS and SARS-CoV-2 vaccinations has been described in the recent literature [12]. To date, there has only been 1 published case of GBS after Ad26.COV2.S vaccine in the United States [13]. We report a case of GBS that occurred 14 days after Ad26. COV2.S vaccine injection without antecedent gastrointestinal or respiratory illness.
As of November 2021, over 16 million people worldwide have received a single injection of Ad26.COV2.S. Among these individuals, there have been 257 provisional reports of GBS registered in the U.S. Department of Health and Human Services (HHS) Vaccine Adverse Events Reporting System (VAERS) [14]. Current research suggests a higher prevalence among middle-aged and elderly males and symptoms onset 11 to 22 days after SARS-CoV-2 vaccination injection [15,16]. In alignment with these findings, the present case of GBS occurred in a 66-year-old male 14 days after vaccination injection. He developed ascending symmetric weakness, loss of reflexes, and paresthesia that progressed through his lower extremities and distal upper extremities. A similar temporal course of atypical GBS with facial diplegia was reported after Ad26.COV2.S vaccination [11]. Besides that published report, an ongoing clinical trial has pinpointed a single case of GBS after Ad26. COV2.S [14]. Immunization with Ad26.COV2.S stimulates robust humoral and cellular immune responses effective against severe and critical disease 14 days after injection [17]. The antibody cross-reactivity of adenovirus vector components of Ad26.COV2.S and peripheral nerve glycoproteins could theoretically lead to GBS [18]. Consequently, demyelination or axonal loss can result in conduction failure. Several other viral vector and mRNA SARS-CoV-2 vaccines have been implicated as causes of GBS [16,19,20] The underlying pathogenic mechanism in vaccine-related GBS remains speculative at this time. Whether the development of GBS after SARS-CoV-2 vaccination is causal or coincidental remains debated [21].
Even though GBS is often a monophasic illness, a remitting and relapsing course occurs in 1–6% of cases [22]. Here, we illustrate a fluctuating course of ascending sensorimotor features. In the present case report, the patient had subtle sensorimotor improvements after IVIG and plasma exchange for a brief duration before recurrence. In Contrast, in the case of atypical GBS after Ad26.COV2.S reported by Jain and colleagues, successful treatment with IVIG and plasma exchange was demonstrated over 14 days [13]. Immunomodulatory therapy is the mainstay of treatment and usually contributes to a favorable prognosis. However, up to 40% of patients lack adequate treatment response 1 month after IVIG or plasma exchange [23]. Further, 10% of patients experience initial symptomatic improvement or stabilization from IVIG and plasma exchange prior to neurological decline [23]. This clinical course, called treatment-related fluctuation, can pose a treatment challenge for physicians [24–26].
The association between SARS-CoV-2 infection and GBS remains controversial. A 2021 review identified a total of 109 published cases of GBS with confirmed SARS-CoV-2 infection. Although causality could not be proven, the authors found that AIDP with facial palsy was the most common presentation [27]. On the other hand, an epidemiological and cohort study by Keddie and colleagues did not find evidence of a causative relationship between SARS-CoV-2 and GBS [28]. A proposed mechanism of AIDP in SARS-CoV-2 involves molecular mimicry between pathogen-borne antigens and human heat shock proteins [5,7,29]. Similarly, most reported cases of GBS after SARS-CoV-2 vaccination are consistent with AIDP [30,31]. In this case report, a history of SARS-CoV-2- like symptoms or laboratory confirmed infection was not identified. However, it is plausible that he developed GBS after an asymptomatic course of SARS-CoV-2 infection.
With the ongoing pandemic and worldwide mass vaccination programs, clinicians should be aware of potential neurological complications that can emerge after administering SARSCoV-2 vaccinations. By the same token, clinicians must demystify vaccination safety profile and efficacy concerns for patients to avert vaccination hesitancy. At present, there is insufficient evidence to suggest a causal relationship between SARS-CoV-2 vaccination and the development of GBS. Therefore, the benefits of SARS-CoV-2 vaccination far outweigh the risks.
Conclusions
A temporal association between the Ad26.COV2.S (Johnson & Johnson/Janssen) vaccine and the onset of GBS was demonstrated in this case report. A feasible underlying pathogenic mechanism involves the cross-reactivity of antibodies stimulated by adenovirus vaccine components and peripheral nerve glycoproteins. However, there is currently insufficient evidence to support a causal relationship between Ad26. COV2.S and the development of GBS. Further evidence gathered from clinician surveillance and clinical trials are needed to draw these conclusions.
References:
1.. Sadoff J, Le Gars M, Shukarev G, Interim results of a phase 1-2a trial of Ad26.COV2.S COVID-19 vaccine: N Engl J Med, 2021; 384(19); 1824-35
2.. Karron RA, Key NS, Sharfstein JM, Assessing a rare and serious adverse event following administration of the Ad26.COV2.S vaccine: JAMA, 2021; 325(24); 2445-47
3.. Razok A, Shams A, Almeer A, Zahid M, Post-COVID-19 vaccine Guillain-Barré syndrome; First reported case from Qatar: Ann Med Surg (Lond), 2021; 67; 102540
4.. Shahrizaila N, Lehmann HC, Kuwabara S, Guillain-Barré syndrome: Lancet, 2021; 397(10280); 1214-28
5.. Koike H, Katsuno M, Macrophages and autoantibodies in demyelinating diseases: Cells, 2021; 10(4); 844
6.. Hadden RD, Cornblath DR, Hughes RA, Electrophysiological classification of Guillain-Barré syndrome: Clinical associations and outcome. Plasma Exchange/Sandoglobulin Guillain-Barré Syndrome Trial Group: Ann Neurol, 1998; 44(5); 780-88
7.. Koike H, Chiba A, Katsuno M, Emerging infection, vaccination, and Guillain-Barré syndrome: A review: Neurol Ther, 2021; 10(2); 523-37
8.. Perez-Vilar S, Hu M, Weintraub E, Guillain-Barré syndrome after high-dose influenza vaccine administration in the United States, 2018-2019 season: J Infect Dis, 2021; 223(3); 416-25
9.. Salmon DA, Proschan M, Forshee R, Association between Guillain-Barré syndrome and influenza A (H1N1) 2009 monovalent inactivated vaccines in the USA: A meta-analysis: Lancet, 2013; 381(9876); 1461-68
10.. Miller ER, McNeil MM, Moro PL, The reporting sensitivity of the Vaccine Adverse Event Reporting System (VAERS) for anaphylaxis and for Guillain-Barré syndrome: Vaccine, 2020; 38(47); 7458-63
11.. Márquez Loza AM, Holroyd KB, Johnson SA, Guillain-Barré syndrome in the placebo and active arms of a COVID-19 vaccine clinical trial: Temporal associations do not imply causality: Neurology, 2021 [Online ahead of print]
12.. Finsterer J, Scorza FA, Scorza CA, Post SARS-CoV-2 vaccination Guillain-Barré syndrome in 19 patients: Clinics (Sao Paulo), 2021; 76; e3286
13.. Jain E, Pandav K, Regmi P, Facial diplegia: A rare, atypical variant of Guillain-Barré syndrome and Ad26.COV2.S vaccine: Cureus, 2021; 13(7); e16612
14.. Shay DK, Gee J, Su JR, Safety monitoring of the Janseen (Johnson & Johnson) COVID-19 vaccine – United States, March-April 2021: MMWR Morb Motral Wkly Rep, 2021; 70; 680-84
15.. Rosenblum HG, Hadler SC, Moulia D, Use of COVID-19 vaccines after reports of adverse events among adult recipients of Janssen (Johnson & Johnson) and mRNA COVID-19 vaccines (Pfizer-BioNTech and Moderna): Update from the Advisory Committee on Immunization Practices – United States, July 2021: MMWR Morb Motral Wkly Rep, 2021; 70; 1094-99
16.. Finsterer J, SARS-CoV-2 vaccinations are unsafe for those experiencing post-vaccination Guillain-Barré syndrome: Ann Med Surg (Lond), 2021; 68; 102584
17.. Corchado-Garcia J, Zemmour D, Hughes T, Analysis of the effectiveness of the Ad26.COV2.S adenoviral vector vaccine for preventing COVID-19: JAMA Netw Open, 2021; 4(11); e2132540
18.. Allen CM, Ramsamy S, Tarr AW, Guillain-Barré syndrome variant occurring after SARS-CoV-2 vaccination: Ann Neurol, 2021; 90(2); 315-18
19.. Waheed S, Bayas A, Hindi F, Neurological complications of COVID-19: Guillain-Barré syndrome following Pfizer COVID-19 vaccine: Cureus, 2021; 13(2); e13426
20.. Patel SU, Khurram R, Lakhani A, Quirk B, Guillain-Barré syndrome following the first dose of the chimpanzee adenovirus-vectored COVID-19 vaccine, ChAdOx1: BMJ Case Rep, 2021; 14(4); e242956
21.. García-Grimshaw M, Michel-Chávez A, Vera-Zertuche JM, Guillain-Barré syndrome is infrequent among recipients of the BNT162b2 mRNA COVID-19 vaccine: Clin Immunol, 2021; 230; 108818
22.. Basta I, Bozovic I, Berisavac I, Recurrent Guillain-Barré syndrome – case series: Neurol India, 2019; 67(6); 1536-38
23.. Hughes RA, Swan AV, Raphaël JC, Immunotherapy for Guillain-Barré syndrome: A systematic review: Brain, 2007; 130(Pt 9); 2245-57
24.. van den Berg B, Walgaard C, Drenthen J, Guillain-Barré syndrome: Pathogenesis, diagnosis, treatment and prognosis: Nat Rev Neurol, 2014; 10(8); 469-82
25.. Ruts L, Drenthen J, Jacobs BC, van Doorn PA, Distinguishing acute-onset CIDP from fluctuating Guillain-Barré syndrome: A prospective study: Neurology, 2010; 74(21); 1680-86
26.. Kleyweg RP, van der Meché FG, Treatment related fluctuations in Guillain-Barré syndrome after high-dose immunoglobulins or plasma-exchange: J Neurol Neurosurg Psychiatry, 1991; 54(11); 957-60
27.. Aladawi M, Elfil M, Abu-Esheh B, Guillain-Barré syndrome as a complication of COVID-19: A systematic review: Can J Neurol Sci, 2021; 49(1); 38-48
28.. Keddie S, Pakpoor J, Mousele C, Epidemiological and cohort study finds no association between COVID-19 and Guillain-Barré syndrome: Brain, 2021; 144(2); 682-93
29.. Diez-Porras L, Vergés E, Gil F, Vidal MJ, Guillain-Barré-Strohl syndrome and COVID-19: Case report and literature review: Neuromuscul Disord, 2020; 30(10); 859-61
30.. Filosto M, Cotti Piccinelli S, Gazzina S, Guillain-Barré syndrome and COVID-19: An observational multicentre study from two Italian hotspot regions: J Neurol Neurosurg Psychiatry, 2021; 92(7); 751-56
31.. Chen Y, Zhang J, Chu X, Xu Y, Ma F, Vaccines and the risk of Guillain-Barré syndrome: Eur J Epidemiol, 2020; 35(4); 363-70
In Press
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943214
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943010
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943687
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943070
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250