21 September 2022: Articles 
Splenic Hamartoma: A Case Report and Literature Review
Challenging differential diagnosis, Rare disease
Khaled A. Obeidat1AEF*, Mohammed W. Afaneh1BEF, Hamzeh Mohammad Al-Domaidat1BEF, Hamzeh Ibrahim Al-Qazakzeh1BF, Fatima J. AlQaisi2BFDOI: 10.12659/AJCR.937195
Am J Case Rep 2022; 23:e937195
Abstract
BACKGROUND: Splenic hamartoma (SH) is a benign vascular lesion, usually found incidentally on abdominal images or at autopsy. Only around 200 cases have been reported since 1861, when SH was first described by Rokitansky. Although it is very rare, it is important to be familiar with it, as it may be a diagnostic challenge to distinguish SH from other mass lesions of the spleen based solely on preoperative investigations.
CASE REPORT: We describe a case of symptomatic, isolated, single splenic hamartoma in a 19-year-old, otherwise healthy young man who presented with upper abdominal pain, nausea, and vomiting for a few months. The examination was unremarkable. The patient has been previously evaluated with abdominal ultrasonography, which found a suspicious splenic hyperechoic lesion. Computed tomography revealed a heterogeneous 5×7 cm enhancing lesion in the spleen, concerning for splenic hamartoma. The patient underwent laparoscopic splenectomy and recovered well. The histopathology examination confirmed the diagnosis of splenic hamartoma.
CONCLUSIONS: Splenic hamartoma is a rare benign vascular lesion of debated etiology. Most cases are asymptomatic and are found incidentally on images, in splenectomies performed for other reasons, or at autopsy. Radiologic findings may suggest the diagnosis and new modalities have shown accuracy in distinguishing splenic hamartomas. However, resection with formal or partial splenectomy is usually still needed since the differential diagnosis is wide, from benign to aggressive lesions, and histopathology remains the criterion standard for diagnosis. Given its benign nature, we found no cases of recurrence or metastasis in the literature.
Keywords: case reports, Hamartoma, Splenectomy, Adult, Humans, Male, Splenic Diseases, Ultrasonography, Vascular Diseases, young adult
Background
Splenic hamartoma (SH) is a benign vascular lesion, usually found incidentally on abdominal images, at surgery, or at autopsy [1]. Only around 200 cases have been reported since 1861, when the first case was described by Rokitansky [2]. It has since been reported with different names, including “splenoma”, “spleen in a spleen”, and “posttraumatic splenic scars”. An incidence rate of 0.024% to 0.13% was detected on postmortem studies, but a higher rate, reaching 2.7%, was found based on a series of splenectomies [3]. SH can occur in any age group, with a clear predilection for the elderly and with no gender preference [2,3]. Although it is very rare, it is important to be familiar with SH, as it may be a diagnostic challenge to distinguish SH from other mass lesions of the spleen based solely on preoperative investigations. Here, we describe a case of symptomatic, isolated, single splenic hamartoma in a 19-year-old man. After that, we discuss several reported cases of splenic hamartomas, along with a detailed review of the characteristic features of splenic hamartomas, including proposed etiology, general behavior, imaging, and histopathology.
Case Report
A 19-year-old Middle-Eastern man was referred to the outpatient clinic due to upper abdominal discomfort for a few months, associated with nausea and a few episodes of non-bilious vomiting. He had no change in bowel habit or fecal characteristics. During that period, he described a normal level of activity and had no chills or fever. He reported no change in weight or appetite. He had no known medical illnesses, with a negative past surgical history. The systematic review and family history were unre-markable, but he had concerns about having a serious disease.
On physical examination, he was slightly overweight, with a body mass index of 27 kg/m2. His vital signs were within normal limits except for a persistent sinus tachycardia at around 110 bpm. Cardiologic evaluation was unrevealing, and an idiopathic sinus tachycardia was reported. He had a normal chest and abdominal exams with no palpable masses or lymphadenopathy. There were no skin changes and no evidence of anemia or bleeding diathesis.
His basic laboratory test results and metabolic panel were within the normal ranges. Prior to referral to our clinic, he was evaluated with esophagogastroscopy, which showed no significant findings except for a small sliding hiatal hernia and an abdominal ultrasound that described a hyperechoic, ill-defined splenic lesion. A computed tomography (CT) scan of the abdomen and pelvis with contrast was requested and revealed a bulky, slightly enlarged spleen containing a heterogeneous hyper-enhancing area measuring about 5.1×5.5×6.9 cm, concerning for a large splenic hamartoma (Figure 1). Our differential diagnosis also included splenic hemangioma, lymphangioma, infectious and granulomatous lesions, and other splenic neoplasms, both benign and malignant. Additionally, radiologists reported a wedge-shaped splenic infarct and a prominent splenic vein. No other masses, cysts, or lymphadenopathy were noted.
After appropriate pre-splenectomy vaccinations, the patient consented to a laparoscopic splenectomy, without complications.
The spleen was extracted intact through a low transverse abdominal incision. Perioperative thromboprophylaxis with enoxaparin was administered and the postoperative period was unremarkable.
Grossly, the resected spleen weighed 253 g and measured 14×10×5 cm, with a smooth surface and unremarkable hilum (Figure 2). Sectioning showed an ill-defined red-tan lesion with a heterogenous cut-surface measuring 8×8×5 cm and a central tan-white scar (Figure 3).
Histopathologic examination revealed a solid vascular tumor with haphazardly arranged small slit-like vascular spaces of varying sizes lined by plump endothelial cells. Interspersed small lymphocytic aggregates were found but no well-defined lymphoid follicles were identified. Immunohistochemical stains were used to identify and differentiate between the suspected SH and the other lesions mentioned above. Our patient’s lesion was positive for CD8 and CD31 and negative for CD34 (Figure 4). Immunostaining for CD3, CD20, and CD45 (Leukocytes Common Antigen) was also demonstrated, and Ki-67 proliferative index was low, indicating a low mitotic rate (Figure 5).
Discussion
We describe the case of a patient with an uncommon, symptomatic splenic hamartoma in a 19-year-old man. Although most reported cases are asymptomatic, about 20% manifest clinically [1]. Females and children are more likely to present with symptoms [2]. Females usually have larger hamartomas, which may reflect a hormonal influence [2]. Presentations vary between primary mass-related symptoms and systemic manifestations [1,3]. The former depends on size and includes abdominal pain, early satiety, and spontaneous rupture [4,5]. Some splenic hamartomas have rapid growth [6]. Systemic manifestations are mostly related to hypersplenism resulting in anemia, thrombocytopenia, or pancytopenia. Less commonly, fever, anorexia, malaise, and weight loss occur [2,7]. The literature shows a strong association between splenic hamartoma and hematologic and solid malignancies, especially squamous cell carcinoma, renal cell carcinoma, and thymoma [1]. Hematologic disorders are particularly prevalent in children [3]. When a splenic mass is detected in a patient with known malignancy, splenic hamartoma should be high on the differential [3]. It may also occur in association with other hamartoma-forming diseases, such as tuberous sclerosis and Wiskott-Aldrich-like syndrome [5,7]. In one series, splenic hamartoma was associated with accessory spleens in one-third of cases [7].
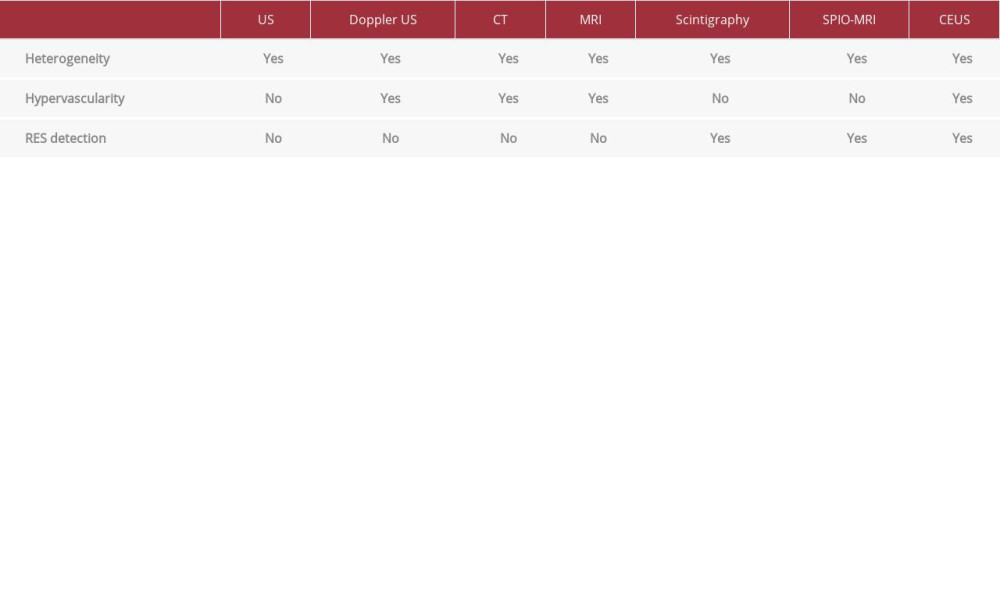
Radiologically, splenic hamartomas may have suggestive features as elaborated by Wang et al [8]. On ultrasound, most hamartomas appear as hyperechoic solid lesions that show hypervascularity on colored Doppler studies. On computed tomography scans, hamartomas appear as well-defined, isodense or hypodense, solid, space-occupying lesions compared to the surrounding normal splenic tissue. They usually enhance heterogeneously with contrast. Cystic changes and calcifications due to ischemia or hemorrhage can sometimes be seen, and when present, can support the diagnosis [4]. Magnetic resonance imaging findings are variable based on the composition of the lesion: fibrous vs non-fibrous. Most often they are isoin-tense on T1-weighted images and heterogeneously hyperin-tense on T2-weighted images. Positron emission tomography has been used, but was unable to clearly distinguish splenic hamartoma from other splenic lesions [9].
Based on the cardinal features of splenic hamartomas – hypervascularity, heterogeneity on imaging, and the presence of reticuloendothelial system (RES) cells – efforts have been devoted recently to use of technetium-99m-labeled colloid scintigraphy, superparamagnetic iron oxide magnetic resonance imaging (SPIO-MRI), and contrast-enhanced ultrasonography (CEUS) to obtain an accurate preoperative diagnosis [10]. Compared to all other benign and malignant splenic lesions that contain no RES cells, hamartomas appear hot on scintigraphy as colloid is being phagocytosed by RES cells found only within the solid part of hamartomas [10]. SPIO-MRI uses nanoparticles that are also taken up by the RES cells and can reveal RES-containing solid parts of hamartomas as being hypoin-tense on T2-weighted sequences [10,11]. CEUS is a promising, cost-effective modality that uses different contrast agents and can show the 3 characteristic features of splenic hamartomas. Sonazoid contrast microbubbles are sequestered within the RES cells; therefore, solid parts of hamartomas appear hyper-echoic on the post-vascular phase in contrast to other splenic lesions that appear hypoechoic [10]. Yang et al defined the CEUS features of benign and malignant splenic lesions including splenic hamartomas [12]. In our case, we relied on information from the CT scan due to the lack of these 3 modalities. Radiologic tools used in the diagnosis of splenic hamartomas are summarized in Table 1.
Splenic hamartomas most often present as a solitary mass, but multiple lesions have been reported [13,14]. They form round or oval-shaped, well-demarcated, unencapsulated, expansive lesions compressing the surrounding normal splenic parenchyma without invasion or infiltration [5]. Size can be as small as few millimeters or can reach multiple centimeters. The median diameter is 5 cm [15]. Histologically, they are classified into a red pulp type composed of bizarre sinusoids and a white pulp type containing lymphoid tissue. Most lesions are a mixture of both, with a dominant component [5,16]. Symptomatic cases are invariably of the red pulp type due to its sinusoidal nature, which can sequester cells and result in hypersplenism and expansion [13]. Typical microscopic features are of disorganized narrow vascular channels lined by CD8-positive, rounded endothelial cells without atypia, showing a low mitotic rate with a low Ki-67 index, in a background of a red pulp stroma, with or without white pulp [17–19]. However, lymphoid follicles are characteristically not present [20]. CD8-positivity (a T cell marker), which entails a splenic sinusoidal origin, helps distinguish this entity from the more common hemangioma, which only shows positivity for endothelial markers such as CD 34 and factor VIII-related antigen [7,21,22].
Whether splenic hamartomas are congenital malformations or are an acquired vascular proliferation is still debated [17,20]. Some scholars consider them to be neoplastic, while others classify them as reactive lesions following trauma, ischemic insult, or an infectious or inflammatory process [1,20]. Others also regard them as active proliferative lesions when associated with hematologic malignancies [1].
Differential diagnosis usually remains wide even after imaging characterization, and splenectomy remains a crucial step for differentiation from other entities. The later includes vascular and non-vascular solid lesions of the spleen [17]. It may be difficult to differentiate a splenic hamartoma from littoral cell angioma, hemangioma, hemangioendothelioma, lymphangioma, sclerosing angiomatoid nodular transformation, and angiosarcoma. Radiologically, a similar picture may be seen with lymphoma, metastatic neoplasms, inflammatory myofibroblastic tumor, and disseminated granulomatous diseases [1,2,4,17,22]. Nonetheless, maintaining a broad perspective helps in identifying those at risk of having a more serious illness.
Although splenic hamartoma may be suggested by images, tissue is required for definitive diagnosis and is usually obtained via splenectomy or partial splenectomy, which can also relieve symptoms and exclude malignancies [2,15,23]. When associated with hematologic disorders, splenectomy can sometimes result in cure [1]. Extraction of the intact spleen has important diagnostic and therapeutic rules. A fragmented spleen may add difficulty to the diagnosis and result in peritoneal dissemination in case a malignancy is diagnosed. In addition, hemato-logic disturbances may continue to occur if a splenic fragment is retained within the peritoneal cavity [3].
It is of paramount importance to mention that fine-needle aspiration, although it can provide evidence for diagnosis, carries the risk of hemorrhage and peritoneal seeding. Hence, surgery becomes mandatory [4]. In children, given the important immune function of the spleen, every effort should be made to save normal splenic tissue with partial splenectomy for single small lesions. Otherwise, total splenectomy may be preferred for multiple, large, or centrally-located lesions or when malignancy cannot be excluded [4,9,15,23].
Conclusions
Splenic hamartoma is a rare, benign, vascular lesion of debated etiology. Most cases are asymptomatic and are found incidentally on images, in splenectomies performed for other reasons, or at autopsy. Most cases were managed in a similar general approach with some differences in imaging modalities used for diagnosis based on preference and availability. Radiologic findings may suggest the diagnosis and new modalities have shown some accuracy in diagnosing splenic hamartomas. However, resection with formal or partial splenectomy is usually still needed since the differential diagnosis is wide, from benign to aggressive lesions, and histopathology remains the criterion standard for diagnosis. Given its benign nature, no cases of recurrence or metastasis were found in the literature.
Figures
References:
1.. Sim J, Ahn HI, Han H, Splenic hamartoma: A case report and review of the literature: World J Clin Cases, 2013; 1(7); 217-19
2.. Lee H, Maeda K, Hamartoma of the spleen: Arch Pathol Lab Med, 2009; 133(1); 147-51
3.. Yazici P, Aydin U, Ersin S, Kaplan H, Hamartoma – a rare benign tumor of the spleen: A report of four cases: Eurasian J Med, 2008; 40(1); 48-51
4.. Wang RT, Xu XS, Hou HL, Symptomatic multinodular splenic hamartoma preoperatively suspected as metastatic tumor: A case report: World J Gastroenterol, 2014; 20(30); 10637-41
5.. Tsitouridis I, Michaelides M, Tsitouridis K, Symptomatic splenoma (hamartoma) of the spleen. A case report: Hippokratia, 2010; 14(1); 54-56
6.. Basso SM, Sulfaro S, Marzano B, Incidentally discovered asymptomatic splenic hamartoma with rapidly expansive growth: A case report: In Vivo, 2012; 26(6); 1049-52
7.. Hayes TC, Britton HA, Mewborne EB, Symptomatic splenic hamartoma: Case report and literature review: Pediatrics, 1998; 101(5); E10
8.. Wang JH, Ma XL, Ren FY, Multi-modality imaging findings of splenic hamartoma: A report of nine cases and review of the literature: Abdom Imaging, 2013; 38(1); 154-62
9.. Avila L, Sivaprakasam P, Viero S, Splenic hamartoma in a child in the era of PET-CT: Pediatr Blood Cancer, 2009; 53(1); 114-16
10.. Sugihara T, Koda M, Kato J, Contrast-enhanced sonography with Sonazoid as a new diagnostic tool for splenic hamartoma: A single case report: J Med Ultrason (2001), 2016; 43(1); 113-18
11.. Wang YX, Superparamagnetic iron oxide-based MRI contrast agents: Current status of clinical application: Quant Imaging Med Surg, 2011; 1(1); 35-40
12.. Yang R, Lu Q, Xu J, Value of contrast-enhanced ultrasound in the differential diagnosis of focal splenic lesions: Cancer Manag Res, 2021; 13; 2947-58
13.. Pisani Ceretti A, Bislenghi G, Laparoscopic splenectomy for splenic hamartoma: A case report: Case Rep Gastrointest Med, 2012; 2012; 435802
14.. Elenga N, Labbé S, Leduc N, Asymptomatic multinodular splenoma (splenic hamartoma) in a child with sickle cell anemia: Int Med Case Rep J, 2017; 10; 233-36
15.. Eker T, Kocaay AF, Sevim Y, Çakmak A, Splenic hamartoma is a rare cause of abdominal pain: Case report and literature review: Turk J Surg, 2015; 33(4); 294-95
16.. Gonzalez Urquijo M, Rodarte-Shade M, A giant splenic hamartoma associated with hematologic disorders:: Ann Med Surg (Lond), 2018; 36; 199-202
17.. Vlachou P, Fagkrezos D, Tzivelopoulou A, A rare case of a splenic hamartoma in a patient with a huge palpable abdominal mass: A case report: J Med Case Rep, 2015; 9; 4
18.. Cheng N, Chen J, Pan Y, Splenic hamartoma with bizarre stromal cells: A case report and literature review: Diagn Pathol, 2018; 13(1); 8
19.. Zhang LF, Tou JF, Wang X, Splenic hamartomas in two children: World J Surg Oncol, 2014; 12; 180
20.. Abramowsky C, Alvarado C, Wyly JB, Ricketts R, “Hamartoma” of the spleen (splenoma) in children: Pediatr Dev Pathol”, 2004; 7(3); 231-36
21.. Sabra TA, Maher A, AlRashidi R, Ibrahim H, A splenic hamartoma: Adding a new case to the literature: A case report: Int J Surg Case Rep, 2022; 90; 106647
22.. Conlon S, Royston D, Murphy P, Splenic hamartoma: Cytopathology, 2007; 18(3); 200-2
23.. Sankar S, Thanka J, Jagdishchandrabose S, Rajendran S, Splenic hamartoma: A rare vascular space occupying lesion of the spleen: Indian J Pathol Microbiol, 2011; 54(1); 223-25
Figures
In Press
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943010
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943687
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943070
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943370
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250