21 April 2023: Articles 
Double Fascicular Nerve Transfer Restored Nearly Normal Functional Movements in a Completely Paralyzed Upper Extremity Resulting from an ACDF Surgery: A Case Report and Review of Recent Literature
Unusual clinical course, Educational Purpose (only if useful for a systematic review or synthesis)
Rahul K. Nath1ABCD*, Chandra Somasundaram1CDEFDOI: 10.12659/AJCR.938650
Am J Case Rep 2023; 24:e938650
Abstract
BACKGROUND: Cervical spine deformities can occur because of genetic, congenital, inflammatory, degenerative, or iatrogenic causes.
CASE REPORT: We report a 45-year-old woman who presented to our clinic with complete paralysis of the left upper extremity 5 months after C4-C6 discectomy and fusion surgery. The electrodiagnostic and EMG reports 3 months after her previous surgery revealed left C5-C7 polyradiculopathy involving the upper trunk, lateral and posterior cords, and atrophy of the left deltoids, triceps, and biceps muscles. She underwent the following nerve transfer procedures with the senior author (RKN): The median nerve fascicles were transferred to the biceps and brachialis branches of the musculocutaneous nerve. Radial nerve triceps branches were transferred to the deltoid and teres minor branches of the axillary nerve. The patient could fully abduct her left shoulder to 170°, and the LUE functions were restored to nearly normal 17 months after the surgery.
CONCLUSIONS: Neurolysis combined with nerve transfer might be the most effective treatment for cervical spinal root injuries. Advances in peripheral nerve rewiring, transcranial magnetic stimulation, brain-computer interface robotic technologies, and emerging rehabilitation will undoubtedly increase the possibility of reviving the extremities in patients with central pathology by restoring the descending motor signals through the residual neural network connections.
Keywords: Cervical Spine Fusion Anomalies, Brachial Plexus Neuropathies, Nerve Transfer, Range of Motion, Articular, Female, Humans, Middle Aged, Brachial Plexus, Muscle, Skeletal, Paralysis, Upper Extremity
Background
Cervical spine deformities can occur because of genetic, congenital, inflammatory, degenerative, or iatrogenic causes. The most common single cause reported is postsurgical (iatrogenic) cervical spine injury [1]. ACDF is an effective procedure to treat cervical spine diseases [2,3]. However, spinal cord injury (SCI), nerve root impairment, and other complications have been reported following ACDF [4,5]. Even the most experienced cervical spine surgeon with great care can inadvertently cause an iatrogenic cervical spine injury. Patient positioning, distractor placement, amount or extent of decompression, type of instrumentation, selection or placement of the bone graft, and use of postoperative immobilization are all suggested risks that may result in postoperative cervical spine deformity [6].
The reported incidence of iatrogenic neurological injury ranges from 3.2% to 0.3% after cervical spinal surgery [7,8]. The C5 root is at a higher risk of iatrogenic injury because of its short course as it exits the spinal cord [9]. C6 injuries can occur simultaneously due to the proximity to the C5 root. C5–C6 palsies are reported in 5–10% of patients after cervical spine surgery [10–12].
Injury to C7–T1 or C8 and T1 roots is a rare occurrence in spine surgery, in which shoulder and elbow movements remain normal, but weakness of the hand can occur [13]. Despite its low incidence, iatrogenic neurological deterioration can cause severe functional limitations, such as complete loss of functional movements in this patient’s left upper extremity (LUE).
Case Report
We report the case of a 45-year-old woman who presented to us with complete paralysis of the LUE and 0° in shoulder abduction 5 months after an anterior cervical discectomy and fusion (ACDF) surgery, specifically a C4–C6 discectomy and fusion.
Three days before her ACDF surgery, the patient felt a sudden onset of excruciating pain from the neck radiating into her left arm, and she came to the emergency room (ER) at 4 AM the next day. An X-ray was taken and found no pathology; she was discharged with pain medication. The patient later received an MRI, revealing C4–C6 compression and nerve impingement. The patient underwent an ACDF procedure 3 days after her ER visit. According to her previous medical records, and her statement, she could not lift her left arm after waking up from the surgery. She was in the acute rehabilitation unit (ARU) for 1 week and received physical therapy for 3 months. She had left shoulder abduction of 1/5 MRC and left elbow flexion of 0/5 MRC strength during her 3-month follow-up evaluation with the previous surgeon. However, her left wrist extension and flexion were 5/5 and her left-hand grip was 5/5. The surgeon referred her to a peripheral nerve specialist for axillary nerve transfer.
The patient contacted our clinic 3 months after her prior ACDF surgery, as her previous surgeon referred her to a peripheral nerve specialist. However, she visited our clinic 5 months after her previous surgery. She stated she had difficulties in activities of daily living. Her shoulder became dislocated regularly, and she was able to put it back in. She had multiple imaging (X-ray, MRI, and EMG) analyses before and 3 months after the ACDF and before nerve transfer surgeries. The electrodiagnostic report 3 months after the ACDF surgery revealed left C5–C7 polyradiculopathy involving the upper trunk and lateral and posterior cords. The EMG results 3 months after the previous surgery showed atrophy of the left deltoids, triceps, and biceps muscles, and she was unable to activate these muscles volitionally. She reported having arm pain (7/10) but no neck pain pre-operatively.
The patient has undergone the following surgeries with the senior author (RKN) 5 months after she had ACDF surgery at another hospital. The first nerve transfer was done with the transposition of the 2 major fascicle groups of the median nerve to the biceps and brachialis branches of the musculocutaneous nerve. The second nerve transfer was done with the transposition of radial nerve triceps branches (1 and 2) to the deltoid and teres minor branches of the axillary nerve. The appropriate nerves in the surgery (median, radial, and axillary nerves) were neurolyzed. Additionally, latissimus dorsi and teres major muscle contractures were released.
The patient was brought to the operating room, where she underwent general anesthesia. The left arm and chest were prepped and draped in the usual sterile fashion. An incision was created immediately parallel to the scapular border in the lateral chest. Contractures of the latissimus and teres major were released. The superomedial border of the triceps was dissected and found the axillary nerve.
The neurological changes during the surgery were monitored. A free-running EMG was performed to test the motor system’s integrity and nerve/root irritability. A continuous electroencephalogram (EEG) was monitored via a modified 10–20 montage throughout the procedure to monitor the cortical function and depth of anesthesia. Stimulated EMG was used to identify neural structures, and CMAP responses were communicated to the surgeon (RKN). There were no CMAP responses in the axillary nerve to the deltoid, and in the musculocutaneous to the biceps and brachialis at 5mA. The radial nerve was dissected in the roof of the axilla and identified as a triceps branch. Through external and internal neurolysis, the branch was found to have 2 terminal ends, and each was separately dissected for use as axillary nerve donors.
The axillary nerve underwent external and internal neurolysis to separate its 2 motor branches. One terminal end of the radial triceps branch was sutured to the deltoid branch and the other to the teres minor branch using a 9-0 nylon microsuture. Through a new proximal medial arm incision, the branches of the brachial plexus were able to be accessed. The left ulnar F-waves were absent. The median nerve had good electrical continuity and underwent external and internal neurolysis. The musculocutaneous nerve was identified within the substance of the biceps muscle, and it had zero electrical conductivity in the brachialis or biceps branch. At this time, 2 group fascicles of the median nerve were dissected free with a Jacobsen hemostat and then transected. The distal end of one group fascicle was transposed and sutured to the musculocutaneous branch of the brachialis. The second group fascicle was then sutured to the branch of the biceps muscle.
All procedures were done with meticulous microsurgical techniques and under high magnification. All wounds were closed, and Dermabond was applied. The patient was stable and awake without problems after surgery.
She was instructed to use a sling for 3 weeks, followed by regular therapy. Physical therapy was prescribed 5 times weekly for 3 months and included teaching home exercises. Protocols for managing complex regional pain syndrome (CRPS) were also provided.
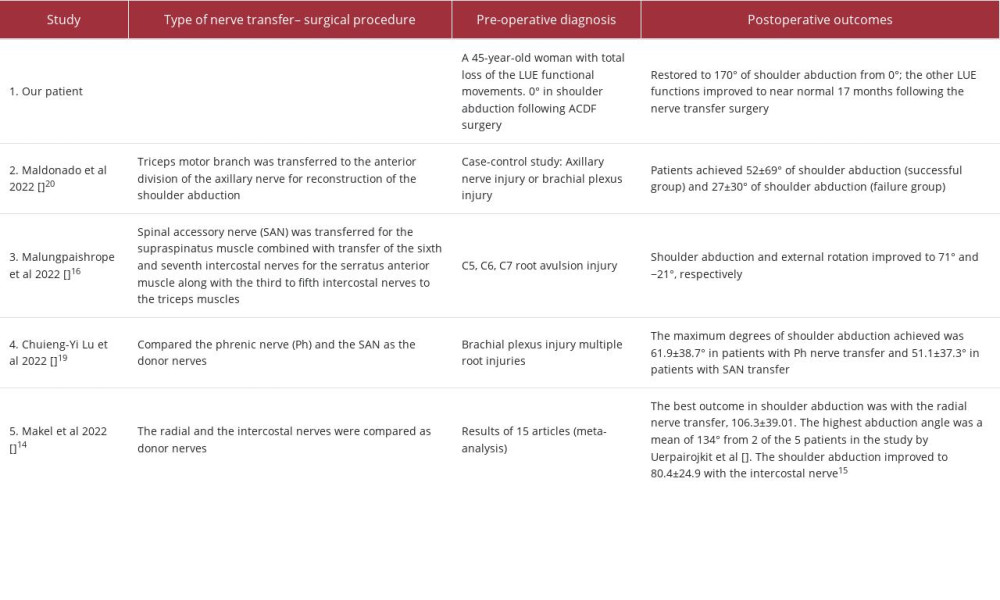
Figure 1A–1C show the patient’s completely paralyzed LUE preoperatively (zero-degree of shoulder abduction). Figure 2A–2C show the patient’s left upper limb active range of motion (AROM) 17 months after the nerve transfer surgery. Our patient’s results are compared with other recently reported triple or multiple nerve transfer procedures in Table 1.
Discussion
Distal nerve transfers are a powerful treatment option for proximal nerve injuries. Addressing nerve root injuries in the context of severe cervical spinal cord injuries resulting in complete paralysis of the extremities has been challenging. Distal-targeted nerve transfers have been successfully performed as a preferred alternative to the site of a proximal injury [14–29] since the regeneration distance will be much shorter and, therefore, the recovery will be faster. Additionally, in cases of iatrogenic surgical injuries at the proximal cervical spinal level (ACDF), as in the present case report, the distal operative area will have no scarring from previous surgeries, allowing for technical precision in reconstructive efforts.
The meta-analysis article by Makel et al [14] summarized the results of 15 articles on distal nerve transfer for brachial plexus injury. They compared the effectiveness of the radial nerve transfer to the intercostal nerve. The best outcome reported was a mean ROM of 106° with the radial nerve vs 80° with the intercostal nerve. Of the 15 included study reports, the best outcome with an average of 134° ROM was from 2 of the 5 patients in the study by Uerpairojkit et al [15]. Uerpairojkit et al [15] used the spinal accessory and the radial nerve to the long head of the triceps. The intercostal nerve transfer is performed in cases of total brachial plexus injury or involvement of the C7 root [14,16]. The disadvantages of intercostal nerve transfers are the need for extensive preparation for harvesting them and the risk of iatrogenic pneumothorax [17]. The contralateral C7 nerve root transfer is an alternative for pan-plexus injuries. Despite higher axon content for use, the reported outcomes have been inconsistent, with common complications [18].
Malungpaishrope et al [16] showed that shoulder abduction improved to 71° after the simultaneous transfer of the spinal accessory nerve (SAN) and intercostal nerves to supraspinatus and triceps muscles, respectively. Chuieng-Yi Lu et al [19] reported that the maximum degrees of shoulder abduction achieved was 61.9±38.7° in patients with phrenic and 51.1±37.3° with SAN nerve transfers. The phrenic nerve is an alternative for shoulder or elbow reanimation in patients with limited donor options. In a case-control study, Maldonado et al [20] compared 2 groups of patients (axillary nerve injury or brachial plexus injury). These 2 groups achieved 52±69° of shoulder abduction (successful group) and 27±30° (failure group) after the triceps motor branch transfer to the anterior division of the axillary nerve.
Song et al [21] compared the results of ipsilateral C7 (IC7) plus SAN transfers with those of triple nerve transfers (TNT), which involved transferring the long head of the triceps nerve branch to the anterior branch of the axillary nerve and transferring one fascicle of the ulnar nerve to the biceps motor branch [21]. Their patients achieved an average of 105° (IC7 plus SAN) and 89° (TNT) in shoulder abduction [21]. Fasce et al [22] showed a mean of 135° in shoulder abduction with triple nerve transfers. In comparison to recently reported double nerve transfer procedures to reanimate shoulder abduction [14,22] or elbow flexion [23], as well as triple or multiple nerve transfers [21,22], our surgical outcome (double nerve transfers with neurolysis and muscle contracture release) was more effective, as it restored all UE functional movements (shoulder abduction and elbow flexion) to nearly normal (Figure 2A–2C).
Better outcomes have been shown when surgeries are performed at less than 6 months (before developing significant muscular atrophy and fibrosis), as the reinnervation possibilities are greater. Fasce et al [22] reported that patients under age 25 years have better outcomes due to their higher regenerative capability. Solla et al [24] reported that older patients had worse outcomes. However, our patient was older (45 years) but still achieved all UE functions 17 months after the nerve transfer surgery, as she had external and internal neurolysis of the involved nerve fascicles and release of muscle contractures. Additionally, she had surgeries within 5 months after her iatrogenic injury. Neurolysis can provide excellent to good outcomes, as shown recently in 82% of the 73 Serratus anterior palsy patients [30]. Neurolysis during nerve transfer surgery remains the best treatment for spinal root avulsion injuries [25].
Solla et al [24] reported that their patients shoulder abduction improvements were excellent (>60°) and good (>40°) after SAN to SSN, whether they were associated with axillary nerve reconstruction strategies or not, respectively. Four patients who had SAN to SSN combined with radial to axillary nerve transfer demonstrated a statistically significant increase in the range of abduction (median, 90° vs 42.5°, respectively) compared with those who had SAN to SSN transfer alone. Improvements in shoulder abduction after SAN to SSN transfer are comparatively smaller [24,26,27]. The supraspinatus is not recruited earlier, as commonly believed, but was found to be recruited consistently with many other shoulder muscles, including the deltoid, and work synergistically in abducting the shoulder [31]. The SAN to SSN transfer also failed to provide useful recovery of external rotation [28]. Moreover, the transfer of SAN to SSN via the traditional supraclavicular anterior approach often leads to partial denervation of the trapezius muscle [29].
Conclusions
Neurolysis combined with nerve transfer might be the most effective treatment for cervical spinal root injuries. Advances in peripheral nerve rewiring, transcranial magnetic stimulation, brain-computer interface robotic technologies, and emerging rehabilitation will undoubtedly increase the possibility of reviving the extremities in patients with central pathology such as hemiplegia or stroke by restoring the descending motor signals through the residual neural network connections.
Figures
References:
1.. Chi JH, Tay B, Stahl D, Lee R, Complex deformities of the cervical spine: Neurosurg Clin N Am, 2007; 18(2); 295-304
2.. Lee HC, Chen CH, Wu CY, Comparison of radiological outcomes and complications between single-level and multilevel anterior cervical discectomy and fusion (ACDF) by using a polyetheretherketone (PEEK) cage-plate fusion system: Medicine (Baltimore), 2019; 98(5); e14277
3.. Fang H, Liu PF, Ge C, Anterior cervical corpectomy decompression and fusion for cervical kyphosis in a girl with Ehlers-Danlos syndrome: A case report: World J Clin Cases, 2019; 7(4); 532-37
4.. Grob D, Luca A, Surgery for cervical stenosis: Anterior cervical decompression, corpectomy, and fusion: Eur Spine J, 2010; 19(10); 1801-2
5.. Daniels AH, Riew KD, Yoo JU, Adverse events associated with anterior cervical spine surgery: J Am Acad Orthop Surg, 2008; 16(12); 729-38
6.. Lehman RA, Angevine P, Rhim SC, Riew KD, Iatrogenic cervical deformity: Neurosurg Clin N Am, 2006; 17(3); 247-61
7.. Oh HS, Hwang BW, Park SJ, Percutaneous endoscopic cervical discectomy (PECD): An analysis of outcome, causes of reoperation: World Neurosurg, 2017; 102; 583-92
8.. Flynn TB, Neurologic complications of anterior cervical discectomy in Louisiana: J La State Med Soc, 1984; 136(7); 6-8
9.. Uematsu Y, Tokuhashi Y, Matsuzaki H, Radiculopathy after laminoplasty of the cervical spine: Spine, 1998; 23(19); 2057-62
10.. Bydon M, Macki M, Kaloostian P, Incidence and prognostic factors of C5 palsy: A clinical study of 1001 cases and review of the literature: Neurosurgery, 2014; 74(6); 595-605
11.. Nassr A, Eck JC, Ponnappan RK, The incidence of C5 palsy after multilevel cervical decompression procedures: A review of 750 consecutive cases: Spine (Phila Pa 1976), 2012; 37(3); 174-78
12.. Shou F, Li Z, Wang H, Prevalence of C5 nerve root palsy after cervical decompressive surgery: A meta-analysis: Eur Spine J, 2015; 24(12); 2724-34
13.. Goubier J-N, Teboul F, Management of hand palsies in isolated C7 to T1 or C8, T1 root avulsions: Tech Hand Up Extrem Surg, 2008; 12(3); 156-60
14.. Makeľ M, Sukop A, Kachlík D, Possible donor nerves for axillary nerve reconstruction in dual neurotization for restoring shoulder abduction in brachial plexus injuries: A systematic review and meta-analysis: Neurosurg Rev, 2022; 45(2); 1303-12
15.. Uerpairojkit C, Leechavengvongs S, Witoonchart K, Nerve transfer to serratus anterior muscle using the thoracodorsal nerve for winged scapula in C5 and C6 brachial plexus root avulsions: J Hand Surg Am, 2009; 34(1); 74-78
16.. Malungpaishrope K, Leechavengvongs S, Uerpairojkit C, Outcomes of spinal accessory nerve and intercostal nerve transfers for shoulder stabilisation and elbow extension in patients with c5,6,7 root avulsion injury: J Hand Surg Asian Pac, 2022; 27(3); 447-52
17.. Kovachevich R, Kircher MF, Wood CM, Complications of intercostal nerve transfer for brachial plexus reconstruction: J Hand Surg Am, 2010; 35(12); 1995-2000
18.. Sammer DM, Kircher MF, Bishop AT, Hemi-contralateral C7 transfer in traumatic brachial plexus injuries: Outcomes and complications: J Bone Joint Surg Am, 2012; 94(2); 131-37
19.. Chuieng-Yi Lu J, An-Jou Lin J, Lee CS, Phrenic nerve as an alternative donor for nerve transfer to restore shoulder abduction in severe multiple root injuries of the adult brachial plexus: J Hand Surg Am, 2022 [Online ahead of print]
20.. Maldonado AA, Dittman LE, Loosbrock MF, Factors associated with poorer outcomes from triceps motor branch to anterior axillary nerve transfer: A case-control study: World Neurosurg, 2022; 167; e1115-e21
21.. Song J, Qiu WJ, Chen L, Transfers of the ipsilateral C7 plus the spinal accessory nerve versus triple nerve transfers for treatment of C5–C6 avulsion of the brachial plexus: J Hand Surg Am, 2020; 45(4); 363.e1-.e6
22.. Fasce I, Fiaschi P, Bianconi A, Long-term functional recovery in C5–C6 avulsions treated with distal nerve transfers: Neurol Res, 2021; 30; 1-7
23.. Srampickal GM, Mathew A, Raveendran S, Restoration of elbow flexion in adult traumatic brachial plexus injury – a quantitative analysis of results of single versus double nerve transfer: Injury, 2021; 52(3); 511-15
24.. Solla DJF, de Oliveira AJM, Riechelmann RS, Functional outcome predictors after spinal accessory nerve to suprascapular nerve transfer for restoration of shoulder abduction in traumatic brachial plexus injuries in adults: The effect of time from injury to surgery: Eur J Trauma Emerg Surg, 2022; 48(2); 1217-23
25.. Agarwal P, Mittal S, Sharma D, Diagnosis, and management of adult BPI: Results of first 50 cases: J Clin Orthop Trauma, 2021; 12(1); 166-71
26.. Heise CO, de Oliveira AJ, Bhering T, Nerve transfers for acute flaccid myelitis: A case series: Childs Nerv Syst, 2021; 37(1); 211-15
27.. Texakalidis P, Tora MS, Lamanna JJ, Combined radial to axillary and spinal accessory nerve (SAN) to suprascapular nerve (SSN) transfers may confer superior shoulder abduction compared with single SA to SSN transfer: World Neurosurg, 2019; 126; e1251-e56
28.. Baltzer HL, Wagner ER, Kircher MF, Evaluation of infraspinatus reinnervation and function following spinal accessory nerve to suprascapular nerve transfer in adult traumatic brachial plexus injuries: Microsurgery, 2017; 37(5); 365-70
29.. Guan SB, Hou CL, Chen DS, Gu YD, Restoration of shoulder abduction by transfer of the spinal accessory nerve to suprascapular nerve through dorsal approach: A clinical study: Chin Med J (Engl), 2006; 119(9); 707-12
30.. Roulet S, Bernier D, Le Nail LR, Neurolysis of the distal segment of the long thoracic nerve for the treatment of scapular winging due to serratus anterior palsy: A continuous series of 73 cases: J Shoulder Elbow Surg, 2022; 31(10); 2140-46
31.. Reed D, Cathers I, Halaki M, Ginn K, Does supraspinatus initiate shoulder abduction?: J Electromyogr Kinesiol, 2013; 23(2); 425-29
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133