12 October 2023: Articles 
Successfully Managing Severe Anemia in a Trauma Patient Who Refused Blood Transfusion: A Case Report
Management of emergency care
Irene F. SanchezDOI: 10.12659/AJCR.940326
Am J Case Rep 2023; 24:e940326
Abstract
BACKGROUND: Surgical management in patients who undergo traumatic blood loss but who refuse blood transfusion can be challenging, but physicians and surgeons must comply with the wishes and beliefs of their patients. This report describes the management of severe anemia, with hemoglobin level of 2.5 g/dL, in a 71-year-old male Korean trauma patient who declined blood transfusion.
CASE REPORT: A 71-year-old man was admitted to hospital with severe blood loss following trauma. He declined blood transfusion due to his religious belief as a Jehovah’s Witness. On day 4, the patient’s hemoglobin level dropped from 7.7 to 3.9 g/dL. Despite the need for blood transfusion, the patient refused. Hence, therapeutic strategies, including crystalloid fluid resuscitation, bleeding control, vasopressor support, erythropoietin administration, supplementation with iron, folic acid, and vitamin B12, coagulopathy correction, oxygen consumption reduction, and mechanical ventilation were implemented. Following 16 days of supportive management, the hemoglobin reached 7.4 g/dL. However, it suddenly decreased on day 41 (2.5 g/dL) due to episodes of melena secondary to an actively bleeding gastric ulcer, which was successfully managed with endoscopic hemostasis. Despite increased vasopressor dosage and addition of vasopressin and hydrocortisone, the patient became unresponsive with persistent hypotension. Methylene blue was used as the final therapeutic agent. The patient responded well and subsequently recovered without blood transfusion.
CONCLUSIONS: This report has presented the clinical challenges of managing the case of a patient who requires but declines blood transfusion and has highlighted the approach to clinical care while respecting the wishes of the patient.
Keywords: Anemia, Republic of Korea, Jehovah's Witnesses, Multiple Trauma, Transfusion Refusal, Male, Humans, Aged, Blood Transfusion, Hemoglobins, Vitamin B 12
Background
Hemorrhage resulting in shock is one of the most common preventable causes of death in patients with trauma [1]. Prompt blood volume resuscitation is vital to control and reverse the effects of shock. Volume resuscitation using blood products restores the oxygen-carrying capacity of the intravascular mi-lieu [2]. However, blood transfusions cannot be administered to patients with religious restrictions.
In the United States, 7 in 10 Americans (70%) identify as Christians, and of these, approximately 1% identify as Jehovah’s Witnesses [3]. As of October 22, 2022, the total population of the Republic of Korea was 51 368 779, with approximately 100 000 Jehovah’s Witnesses [4]. One restriction that these devotees uphold is their refusal to undergo blood transfusions. They believe that receiving a blood transfusion would result in eternal damnation [5]. This restriction makes the management of critically ill trauma patients more challenging. Patient autonomy and freedom of religion must be considered while clinicians aim to provide appropriate and timely care.
Bloodless management strategies have been described in the literature for the care of Jehovah’s Witness patients in need of transfusion. These strategies include fluid resuscitation, minimization of blood loss, control of bleeding sources, augmentation of erythropoiesis, and vitamin supplementation [6–10]. Current bloodless approaches included methods relevant to both medical and surgical patients [7]. Methods applicable to both medical and surgical patients include minimizing laboratory testing, low-volume microtainers for phlebotomy, inline blood-return devices for indwelling arterial and central venous catheters, tolerating lower hemoglobin levels, discontinuing herbal supplements, and diagnosing and treating anemia [7]. Methods relevant only to surgical patients include early diagnosis and treatment of preoperative anemia; intraoperative autologous blood salvage; meticulous surgical techniques; perioperative antifibrinolytics; new methods of cautery, topical sealants and hemostatic agents; avoiding hypothermia; intentional hypotension; point-of-care coagulation testing; and judicious decision-making for when to operate [7].
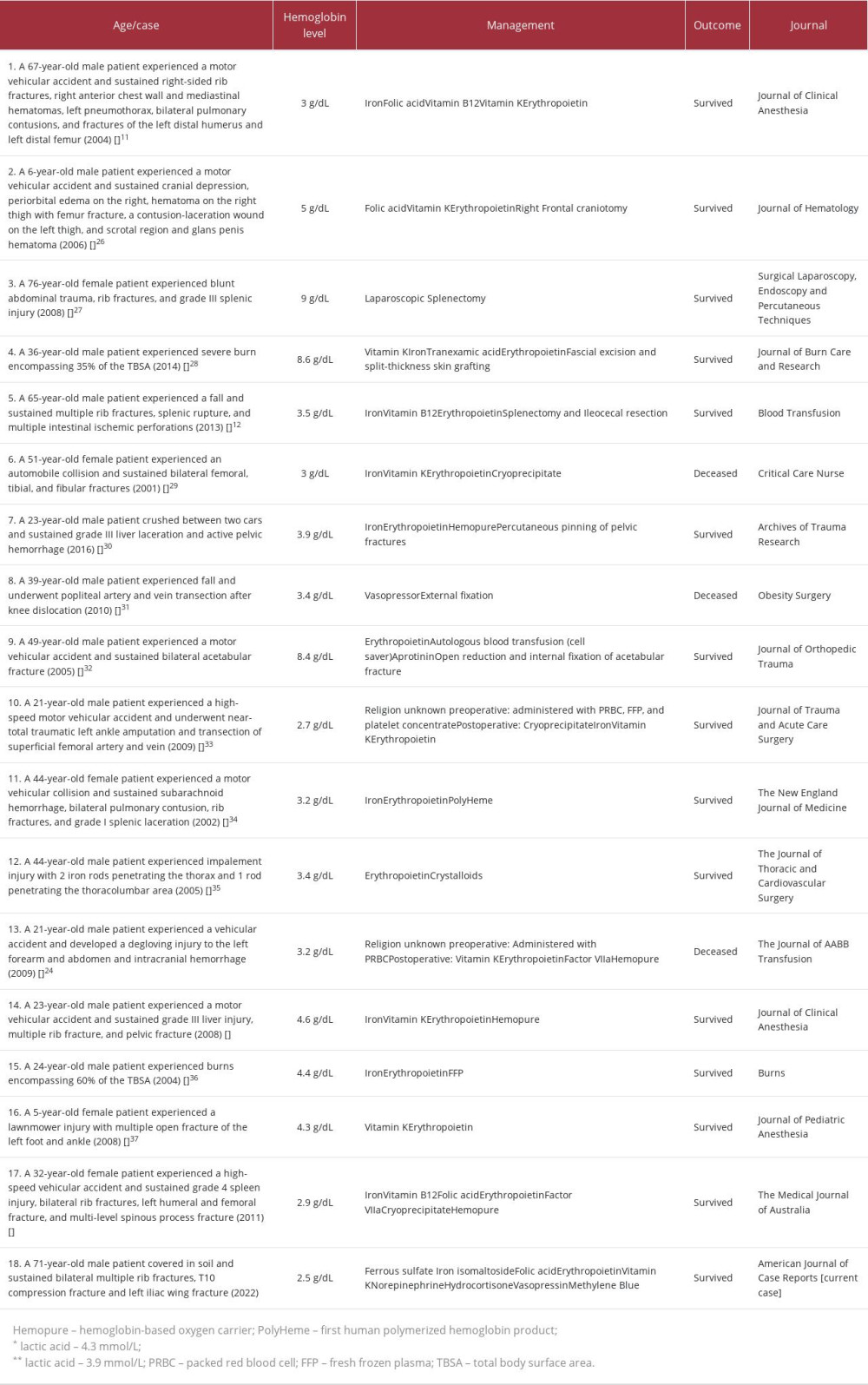
These strategies have been used by numerous published case reports concerning trauma patients who refused blood transfusion. One case involved a 67-year-old male patient with an initial hemoglobin of 9.7 g/dL who sustained right-sided rib fractures, right anterior chest wall and mediastinal hematomas, left pneumothorax, bilateral pulmonary contusions, and fracture of the left distal humerus and left distal femur. Fluid resuscitation using crystalloids and hetastarch was given with supplementation of iron, folic acid, vitamin B12, erythropoietin, and vitamin K. The patient survived, with a discharged hemoglobin level of 7.9 g/dL [11] (Table 1). Another case was of a 65-year-old male patient who fell and sustained multiple rib fractures, splenic rupture, and multiple intestinal ischemic perforations. He had an initial hemoglobin level of 6.4 g/dL. Colloids and crystalloids were given, as well as iron, vitamin B12, and erythropoietin supplementation. He also underwent splenectomy with ileocecal resection. The patient survived, with a discharge hemoglobin level of 11.2 g/dL [12] (Table 1).
This report describes the management of the case of a 71-year-old Korean male trauma patient with severe anemia and a hemoglobin level of 2.5 g/dL who declined blood transfusion.
Case Report
A 71-year-old male Jehovah’s Witness patient presented to the Emergency Department (ED) after being covered in soil for 1 h, due to a landslide. The patient’s medical and surgical histories were unremarkable. Upon arrival at the ED, a primary survey was conducted. The patient’s airways were clear, but breathing was laborious. The patient reported dyspnea and chest pain. He was afebrile, with an initial blood pressure of 125/87 mmHg, a heart rate of 99 beats/min, a respiratory rate of 25 cycles/min, and an oxygen saturation of 96% in room air. A few minutes after arrival, the patient showed signs of shock, such as hypotension (60/30 mmHg), tachycardia (119 beats/min), and oxygen desaturation (90% at 15 L/min). Intravenous (i.v.) access was established, together with radial artery arterial line and subclavian vein central line catheterization. Volume resuscitation was initiated with 0.9% sodium chloride. Simultaneously, a quick assessment of the possible causes of shock was performed. Chest radiography showed multiple fractures in the 1st–2nd and 7th–12th right ribs and the 1st–9th and 11th left ribs. A computed tomography scan from the head to the thigh with i.v. injection of a contrast agent was performed, which showed bilateral pneumohemothorax, T10 compression fracture, and a closed, nondisplaced, stable left iliac wing fracture. Intra-abdominal findings were unremarkable.
The patient was admitted to the surgical Intensive Care Unit (ICU) with hemorrhagic shock due to multiple rib fractures. The average blood loss per fractured rib was 100–150 mL [13]. With 8 fractured ribs on the right and 10 fractured ribs on the left, class III–IV hemorrhagic shock (total estimated blood loss of 1800–2700 mL) was expected to occur [2]. Massive thoracic bleeding caused hypotension (60/30 mmHg), tachycardia (119 beats/min), and oxygen desaturation (90% at 15 L/min). Bilateral closed-tube thoracostomy using a 24 Fr chest tube was performed, which drained 420 mL of bloody fluid on the right side and 430 mL of bloody fluid on the left side. Owing to the development of acute hypoxemic respiratory failure due to atelectasis and hemothorax from multiple rib fractures, the patient was intubated to provide ventilatory support. To promote comfort, reduce anxiety, and facilitate patient-ventilator synchrony, sedation was initiated using propofol 1000 mg (4 mL/h) and dexmedetomidate 200 µg (4 mL/h), as well as analgesia using sufentanil citrate 250 µg (4 mL/h). A neuromuscular blocker such as rocuronium bromide, was administered. To support circulation, fluid resuscitation with crystalloids was initiated, and the patient was responsive. However, crystalloid therapy alone cannot replenish lost blood cell components. This was the management dilemma encountered in this case. The patient refused blood transfusion because of his religious beliefs. Hemoglobin levels were expected to decrease in the presence of multiple bilateral rib fractures. On admission, the patient had an initial hemoglobin level of 7.7 g/dL. A further decrease in hemoglobin (3.9 g/dL) was evident on hospital days 4 to 6. This was due to an increase in the sanguineous output from the left chest tube (1680 mL) for 3 days. This prompted embolization of the inter-costal vessels to control the sources of bleeding and prevent further anemia. Supportive therapy was initiated to increase the hemoglobin levels. Ferrous sulfate was administered (256 mg tablet once a day) for 59 days, folic acid (1 mg tablet once a day) for 59 days, iron isomaltoside (200 mg via i.v. infusion once a day) for 27 days, and erythropoietin (10 000 IU via subcutaneous injection once a day, 3 times a week). Finally, a hemoglobin level of 7.4 g/dL was achieved after 16 days.
During the second week of admission, the patient required vasopressor treatment. Prolonged episodes of hypotension and unresponsiveness to fluid therapy were observed. Norepinephrine (0.05–0.1 µg/kg/min) was administered and titrated accordingly to support blood circulation and organ perfusion. Although the patient’s blood pressure measurements fluctuated during his hospital stay, his blood pressure was maintained within an acceptable range using norepinephrine.
On day 41 of hospitalization, the hemoglobin level severely decreased to 2.5 g/dL. Despite ongoing acid suppression medications, the patient experienced episodes of melena. Endoscopic investigation confirmed the presence of active bleeding from a gastric ulcer, which was successfully controlled using endoscopic hemostasis. Eventually, the patient developed refractory hypotension on hospital day 41, even after successful control of gastric ulcer bleeding. The norepinephrine dose was increased from 0.2 µg/kg/min to 1 µg/kg/min. However, the patient continued to experience hypotension. The patient was started on vasopressin 20 IU (3–12 µgtt/min) and hydrocortisone 100 mg (4 mL/h). Despite that, the patient still experienced episodes of hypotension. Owing to the absence of a clinical response despite increasing the dose of norepinephrine and the addition of vasopressin and hydrocortisone, methylene blue was used as the last therapeutic option. On day 42, an i.v. bolus of 100 mg methylene blue was administered 4 times to achieve an initial response. However, the episodes of hypotension persisted. On day 43, an i.v. bolus of methylene blue 100 mg was again administered 4 times. The patient showed a good response, with the blood pressure gradually returning to normal. Methylene blue was then discontinued. Also, on hospital day 43, the patient experienced atrial fibrillation due to a severe reduction in hemoglobin level. Amiodarone hydrochloride was administered; however, no response was observed. Hence, defibrillation was performed, which eventually stabilized the patient’s heart rate. These events were attributed to the cardiac effects of anemia and hypoxia. Thereafter, no other episodes of bleeding or cardiac events were noted, and the hemoglobin levels gradually increased. Vasopressin was titrated down from 12 µgtt/min to 0.3 µgtt/min on day 45. Vasopressin and hydrocortisone were discontinued on day 46. The norepinephrine dose was decreased to 0.4 µg/kg/min on day 44. It was further titrated down to 0.05 µg/kg/min on day 52, and was terminated on day 56.
The presence of infection complicated the overall condition of the patient. During the course of the illness, the patient developed fever, which prompted clinicians to investigate the source. After the patient was intubated, a sputum culture examination was conducted, which revealed multiple isolates during the entire ICU stay. The sputum isolates included
On day 56 of hospitalization, the patient’s clinical condition dramatically improved, with the absence of hypotension. Hemoglobin levels showed an increasing trend. The patient was weaned off the ventilator on day 58. The patient was then transferred to the general ward and referred for rehabilitation. The patient was eventually discharged, with a hemoglobin level of 14.1 g/dL.
Discussion
This clinical case of a 71-year-old critically ill trauma patient was managed without blood transfusion. We describe our approach for handling this unique case scenario successfully.
Acute hemorrhage causes loss of blood volume leading to hypovolemia, hypoperfusion, and hypoxemia with hypoxia. Class I to II hemorrhagic shock (15–20%) can be sufficiently balanced by compensatory physiologic responses, but class III to IV (>30) is critical, and compensatory mechanisms are inadequate [14].
The Advanced Trauma and Life Support protocol indicates that early administration of blood products as part of trauma management is vital to avoid coagulopathy and thrombocytopenia [2]. However, blood transfusion is not an option for some religious devotees. Hence, the management of bleeding and anemia became challenging in this case and involved controlling blood loss, enhancing erythropoiesis, optimizing oxygen delivery, reducing oxygen consumption, and preventing hemodilution and iatrogenic anemia [8].
Hughes et al [6] presented contemporary strategies in clinical management of Jehovah’s witness patients and subdivided it into ensuring hemostasis, minimizing iatrogenic blood loss, enhancing red blood cell production, maintaining blood volume, and minimizing intraoperative red blood cell loss. We used these strategies in the management of the present case.
The primary priority in any hemorrhagic patients is control of bleeding [6]. Efforts must be made to ensure hemostasis from all possible sources. DeLoughery et al [10] explained a comprehensive transfusion replacement strategy in patients who decline blood products. Focus must be directed to achieve hemostasis and the use of tranexamic acid, fibrinogen concentrate, or prothrombin complex concentrate if the patient consents to receive them. Damage control surgery is suggested to address actively bleeding areas. In the present case, multiple bilateral rib fractures were identified as the initial cause of blood loss. To address the source of bleeding, the patient underwent embolization, which was controlled successfully. However, on day 41 of admission, clinicians were challenged again by an actively bleeding gastric ulcer, which caused the hemoglobin level to drop further (2.5 g/dL). Hence, endoscopic intervention was done and successfully controlled the bleeding.
Acute anemia commonly results from acute hemorrhage. Anemia can be classified as mild, moderate, severe, or life-threatening. Some studies have classified severe anemia as having a hemoglobin level of 6.5–7.9 g/dL, whereas life-threatening anemia presents a hemoglobin level of <6.5 g/dL [15]. Based on case reports, case series, and retrospective analyses, severe anemia was defined as a hemoglobin level of less than 5 or 5–7 g/dL [8]. A mortality rate of 34.4% was observed in patients with a hemoglobin level of 4–5 g/dL who refused to receive blood transfusion [9]. When hemoglobin levels drop below 5–6 g/dL, serious adverse effects, including myocardial infarction, arrhythmia, and cardiac failure, can occur [16,17]. In the present case, the patient had a hemoglobin level of 2.5 g/dL. At this hemoglobin level, serious adverse effects were expected, one of which was evident when the patient had an episode of atrial fibrillation. Initially, the patient did not respond to the amiodarone HCl treatment. Therefore, defibrillation was performed.
Since crystalloids cannot replace the lost cellular blood components, enhancement of erythropoiesis is vital. Posluszny et al [8] initiated the Anemia Protocol in Jehovah’s Witness patients with severe anemia (hemoglobin <7 g/dL). He suggested giving erythropoietin (40 000 IU i.v. until hemoglobin >7 g/dL, then weekly thereafter), iron (100 mg i.v. daily for 10 days, then shift to oral supplementation), vitamin C (500 mg 3 times daily, except in renal failure), folate (1 mg orally or i.v. per day) and vitamin B12 (i.v. or oral supplement). Administration of recombinant human erythropoietin promotes hematopoiesis. Aggressive supplementation with iron and erythropoietin (40 000 U daily until hemoglobin >7g/dL) is advised in trauma patients [10]. Koestner et al [18] reported the case of a 36-year-old female Jehovah’s Witness who had experienced multiple traumas. Erythropoietin at an initial dose of 300 IU/kg was administered via i.v. infusion once a day for 3 days; then, a dose of 150 IU/kg was administered via subcutaneous injection 3 times per week. The hematocrit level increased from 13% to 37%, and the patient was discharged without a single blood transfusion [18]. Another study showed that early administration of erythropoietin was associated with a rapid increase in hematocrit levels [19]. Our patient was administered erythropoietin (100 IU/kg) at a dose of 10 000 IU via injection once daily, 3 times a week. Hence, the hematocrit levels increased from 10.9% to 33.1%. Enhanced erythropoiesis is futile without concomitant supplementation with iron, folic acid, or vitamin B12. These nutrients are important adjuncts in hemoglobin production and enhance the hemoglobin response to erythropoietin [20]. In one study, iron supplementation was shown to be beneficial and was strongly considered an adjunct in managing Jehovah’s Witness patients [21]. In the present case, the patient was administered ferrous sulfate, folic acid, iron isomaltoside, and vitamin B12 supplementation. Because of the prolonged anemia, the presence of hemolytic anemia or methemoglobinemia were also considered. Workup for hemolytic anemia did not confirm its presence. Haptoglobin (87.4 mg/dL) and reticulocyte count (1.23% of red blood cells) were within the reference range. A below normal level of lactate dehydrogenase was observed. Serum bilirubin levels were within normal limits, with only isolated events of hyperbilirubinemia (2.34 mg/dL, being the highest level). A peripheral blood smear showed relative monocytosis and eosinophilia. As for methemoglobinemia, no cyanosis was observed, and oxygen saturation was maintained between 98% and 100% throughout the hospital stay.
Correction of coagulopathy and reduction of oxygen consumption were included in our therapeutic options for managing this patient’s case. Correcting coagulopathy to minimize bleeding risk is vital in patients with trauma. Vitamin K can be administered to augment the coagulation system and increase the function of vitamin K-dependent factors. In connection with this, careful observation should be performed in patients with coagulopathy and who are on mechanical ventilation. One study showed that the risk of gastric ulcer bleeding increased in this patient group [21]. Approximately 25% of ICU patients who do not receive prophylactic measures experience gastrointestinal bleeding due to gastric ulcers [22]. In our case, vitamin K was administered at a dose of 10 mg via slow i.v. infusion once daily, and pantoprazole was administered at 40 mg via i.v. infusion once a day. Despite appropriate prophylaxis, our patient developed bleeding from a gastric ulcer, which may be attributed to ongoing coagulopathy. Moreover, there was no workup done to determine levels of clotting factors, because phlebotomy was minimized.
The effects of shock on tissue perfusion are severe. A diminished blood volume causes a decrease in oxygen delivery to tissues, in turn leading to tissue hypoxia. Hence, it is important to implement effective methods to reduce oxygen consumption. Sedation, analgesia, administration of neuromuscular blockers, and mechanical ventilation are strategies used to reduce oxygen consumption [11]. In the present case, the patient was intubated and sedated with propofol (dose titrated per demand) for 2 weeks. Adequate analgesia and appropriate neuromuscular blockers were administered.
Minimizing phlebotomy/iatrogenic blood loss was also part of our strategy [6,7]. Blood sampling was not performed for several days, which explains the absence of hemoglobin data on certain days of the patient’s hospital stay (Figure 1). Phlebotomy was only done when deemed necessary, based on the patient’s clinical condition. As transfusion of blood products is contraindicated due to religious beliefs, maintenance of adequate intravascular volume is done using crystalloid solutions. In our case and in addition to crystalloids, nutritional supplementation with amino acids, trace elements, and multivitamins was initiated to support catabolic status. Crystalloids are insufficient to maintain adequate oxygen delivery to tissues [6]. Posluszny et al [8] included in their severe anemia protocol the use of hemoglobin-based oxygen carriers (HBOC) in cases of critical anemia, which is defined as severe anemia (<7 g/dL) with hypoperfusion, shock, hemodynamic instability, and hemorrhage. This product can bridge the gap until erythropoietin can take its full effect. Case reports on the use of HBOC have been published. One was a case of a 23-year-old male patient who was struck by a car and who sustained multiple rib and pelvic fractures and grade III liver laceration. In addition to vitamin K, erythropoietin, iron supplements, and vitamin C, the patient received HBOC 150 g. After 24 h, the total hemoglobin level increase from 4.6g/dL to 7.4g/dL. The patient survived without blood transfusion [23] (Table 1). Another case was of a 21-year-old male patient who was struck and dragged by a motor vehicle. He had a left forearm and abdominal degloving injury, and extensive intraparenchymal, subdural, and subarachnoid hemorrhage. The hemoglobin level dropped to 3.5g/dL, despite efforts to enhance erythropoiesis. The patient was given HBOC (120 mg), which increased total hemoglobin from 3.2 to 7 g/dL. However, the patient succumbed to his condition [24] (Table 1). The use of HBOC has not yet been approved by the U.S. Food and Drug Administration [7]. There was concern from a previous study about the higher rates of myocardial infarction and death related to hypertension and vasoconstriction from nitric oxide scavenging [8].
Due to the complexity of disease processes, the body can become unresponsive to administered medications. The management of Jehovah’s Witness patients can be stressful and arduous, not only for the patients themselves, but also for the clinicians. In the present case, the patient experienced refractory distributive shock, increasing the requirement for norepinephrine, vasopressin, and hydrocortisone. However, the patient’s blood pressure did not improve. Arterial monitoring of the cardiac indexes showed clinical evidence of vasoplegia, which were low systemic vascular resistance in the setting of normal cardiac output. Refractory shock is mediated by nitric oxide (NO) dysregulation. NO is generated by NO synthase (NOS). NO controls the vascular tone and causes vasodilation, leading to hypotension. After exhausting all methods to increase blood pressure, methylene blue was selected as the final option. Methylene blue is a phenothiazine-based heterocyclic aromatic molecule. It plays a role in refractory shock by inhibiting NOS expression. Thus, NO production is controlled, and vascular tone is restored [25].
In this case, a high dose of methylene blue (2 mg/kg, 4 times daily) was administered to the patient. Initially, minimal response in hypotension was observed. Additional doses of methylene blue were administered, which helped to restore the patient’s blood pressure to normal and subsequently decreased the requirement for norepinephrine.
The management of bleeding in trauma without blood transfusion is taxing. A systematic approach is beneficial. In trauma patients, clinical examination of the patient should simultaneously be done with hemodynamic monitoring, immediate investigation of possible bleeding sites, initiation of fluid resuscitation, and blood sampling for blood typing and blood count. If an overt source of bleeding is identified, prompt control should be done via embolization or surgical approach. Erythropoiesis should be augmented early on with monitoring of the hemoglobin count, as well as an investigation of causes of anemia. Respiratory and cardiovascular support should be done as indicated. Administration of appropriate medications should be based on the patient’s clinical situation. Nutritional support must be initiated from the start to counter the effects of catabolism. Furthermore, judicious decision-making is vital in each step of management, especially in the setting of surgery.
With these strategies, a critically ill trauma patient with life-threatening anemia was successfully treated without blood transfusion. Nevertheless, clinicians are still faced with predicaments that include the availability of appropriate therapeutic options, high hospital costs, and appropriate allocation of ICU resources to patients who need them the most. Most importantly, the degree of psychological and emotional stress that the situation caused to the clinicians and the patient’s family was also one of the factors that makes it challenging to manage patients who refuse blood transfusion. However, the successful outcome for the patient was rewarding. In a dilemma between patients’ autonomy and physicians’ oath to a save life wherever possible using the ideal measures, respect and value of each individual’s chosen belief comes in between. This respect and values have encouraged clinicians to employ all other options of managing severe anemia without blood transfusion.
Conclusions
This report has presented the clinical challenges of managing the case of a patient who requires but declines blood transfusion and has highlighted the approach to clinical care while respecting the wishes of the patient.
References:
1.. Rossaint R, Bouillon B, Cerny V, Task Force for Advanced Bleeding Care in Trauma. Management of bleeding following major trauma: an updated European guideline.: Crit Care., 2010; 14(2); R52
2.. : Advanced Trauma Life Support (ATLS)., 2018, Chicago, American College of Surgeons
3.. , The American Religious Landscape in 2020. July 8, 2021 Retrieved from: https://www.prri.org/research/2020-census-of-american-religion
4.. : Jehovah’s Witnesses [editorial]., 2023, Watch Tower Bible and Tract Society of Republic of Korea
5.. Thompson HA, Blood transfusions and Jehovah’s Witnesses: Tex Med, 1989; 85; 57-59
6.. Hughes DB, Ullery BW, Barie PS, The contemporary approach to the care of Jehovah’s Witnesses: J Trauma, 2008; 65; 237-47
7.. Resar LMS, Wick EC, Almasri TN, Bloodless medicine: Current strategies and emerging treatment paradigms: Transfusion, 2016; 56; 2637-47
8.. Posluszny JA, Napolitano LM, How do we treat life-threatening anemia in a Jehovah’s Witness patient?: Transfusion, 2014; 54; 3026-34
9.. Nishant , Kumari R, Surgical management in treatment of Jehovah’s Witness in trauma surgery in Indian subcontinent: J Emerg Trauma Shock, 2014; 7; 215-21
10.. DeLoughery TG, Transfusion replacement strategies in Jehovah’s Witnesses and others who decline blood products: Clin Adv Hematol Oncol Dec, 2020; 18(12); 826-36
11.. Kulvatunyou N, Heard SO, Care of the injured Jehovah’s Witness patient case report and review of the literature: J Clin Anesth, 2004; 16; 548-53
12.. Lorentzen K, Kjaer B, Jørgensen J, Supportive treatment of severe anemia in a Jehovah’s Witness with severe trauma: Blood Transfus, 2013; 11(3); 452-53
13.. Melendez SL, Doty CI, Rib Fracture: Medscape. Jan 18, 2023 [cited 2023 Feb 4]. Available from: https://emedicine.medscape.com/article/825981-overview
14.. Moore FD, Effects of hemorrhage on body composition.: New Engl J Med, 1965; 273; 567-77
15.. Crowther M, Podolak-Dawidziak M, Anemia: General considerations.: McMaster Textbook of Internal Medicine., Cracow, Medycyna Praktyczna Available from: https://empendium.com/mcmtextbook/chapter/B31.II.15.1
16.. Weiskopf RB, Kramer JH, Viele M, Acute severe isovolemic anemia impairs cognitive function and memory in humans.: Anesthesiology, 2000; 92; 1646-52
17.. Ng KO, Chow LH, Wang CC, Successful management of massive blood loss to extremely low hemoglobin in an elderly woman receiving spinal surgery: Acta Anaesthesiol Sin, 2000; 38; 89-92
18.. Koestner JA, Nelson LD, Morris JA, Safcsak K, Use of recombinant human erythropoietin (r-HuEPO) in a Jehovah’s Witness refusing transfusion of blood products: Case report: J Trauma, 1990; 30; 1406-8
19.. DeMeester SR, Marsh EE, Gerkin TM, Rodriguez J, Immediate use of recombinant erythropoietin in a Jehovah’s Witness following major blunt trauma: Contemp Surg, 1994; 45; 228-32
20.. Jung M, Harish V, Wijewardena A, Management strategies for perioperative anaemia in the severely burn-injured Jehovah’s Witness patients who decline a blood transfusion: A systematic review with illustrative case reports: Burns, 2023; 49; 716-29
21.. Georgiou C, Inaba K, DuBose J, Optimizing outcomes in the Jehovah’s Witness following trauma: Special management concerns for a unique population: Eur J Trauma Emerg Surg, 2009; 35; 383
22.. Marino PL: The ICU Book, 2007; 68, Philadelphia, PA, USA, Lippincott Williams & Wilkins
23.. Mackenzie CF, Morrison C, Jaberi M, Management of hemorrhagic shock when blood is not an option: J Clin Anesth, 2008; 20(7); 538-41
24.. Marinaro J, Smith J, Tawil I, HBOC-201 use on traumatic brain injury: Casae report and review of literature: Transfusion, 2009; 49(10); 2054-59
25.. Naoum EE, Dalia AA, Roberts RJ, Methylene blue for vasodilatory shock in the intensive care unit: a retrospective, observational study: BMC Anesthesiol, 2022; 22; 199
26.. Digieri LA, Pistelli IP, de Carvalho CE, The care of a child with multiple trauma and severe anemia who was a Jehovah’s Witness: Hematology, 2006; 11(3); 187-91
27.. Ayiomamitis GD, Alkari B, Owera A, Ammori BJ, Emergency laparoscopic splenectomy for splenic trauma in a Jehovah’s Witness patient: Surg Laparosc Endosc Percutan Tech, 2008; 18(6); 626-30
28.. Barsun A, Sen S, Palmieri TL, Greenhalgh DG, Reducing postburn injury anemia in a Jehovah’s Witness patient: J Burn Care Res, 2014; 35(4); e258-61
29.. Gilcreast DM, Avella P, Camarillo E, Mullane G, Treating severe anemia in a trauma patient who is a Jehovah’s Witness: Crit Care Nurse, 2001; 21(2); 69-72
30.. Posluszny JA, Napolitano LM, Hemoglobin-based carrier for traumatic hemorrhagic shock treatment in a Jehovah’s Witness: Arch Trauma Res, 2016; 5(2); e30610
31.. Streubel PN, Arndt S, Armitage MS, Fatal knee dislocation in a morbidly obese Jehovah’s Witness. A case report: Obes Surg, 2010; 20(9); 1316-18
32.. Dipasquale T, Greiwe RM, Simmons P, Temporary partial intra-iliac balloon occlusion for the treatment of acetabulum fracture in a Jehovah’s Witness: A case report: J Orthop Trauma, 2005; 19(6); 415-19
33.. Vaziri K, Roland JC, Robinson LL, Extreme anemia in an injured Jehovah’s Witness: A test for understanding of the pathophysiology of severe anemia and the threshold for blood transfusion: J Trauma, 2009; 67(1); E11-13
34.. Cothren C, Moore EE, Offner PJ, Blood substitute nad erythropoietin therapy in a severely injured Jehovah’s Witness: N Engl J Med, 2002; 346(14); 1097-98
35.. Lanthaler M, Freund M, Margreiter R, Nehoda H, Unusual impalement injury in a Jehovah’s Witness: J Thorac Cardiovasc Surg, 2005; 129(5); 1179-80
36.. Muller P, Lozano D, Control of blood loss in the treatment of a Jehovah’s Witness with massive thermal injuries using a fibroblast derived temporary skin substitute: Burns, 2004; 30(5); 483-87
37.. Rizzo G, Astuto M, Withington DE, Extensive lower limbs injuries in a child complicated by complex pain management and profound anemia: Paediatr Anaesth, 2008; 18(8); 779-81
38.. Fitzgerald MC, Chan JY, Ross AW, A synthetic haemoglobin-based oxygen carrier and the reversal of cardiac hypoxia secondary to severe anemia following trauma: Med J Aust, 2011; 194(9); 471-73
In Press
19 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942660
19 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943174
19 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943136
21 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943645
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250