27 January 2024: Articles 
Intrahepatic Cholangiocarcinoma in Wilson’s Disease: A Case Report
Rare disease
Eun Jeong JangDOI: 10.12659/AJCR.942372
Am J Case Rep 2024; 25:e942372
Abstract
BACKGROUND: Wilson’s disease is a rare autosomal recessive disorder characterized by excessive accumulation of copper in the liver, brain, and kidneys. Although it affects only approximately 1 in 30 000 individuals, it leads to progressive liver damage and neurological issue. Wilson’s disease presents a wide spectrum of clinical manifestations related to hepatic disease, ranging from asymptomatic cases to acute liver failure. The occurrence of hepatobiliary malignancies, including intrahepatic cholangiocarcinoma, is relatively uncommon in Wilson’s disease, even among patients with cirrhosis. Only 14 cases have been published so far, including the present report, and its etiology remains unclear.
CASE REPORT: We report the successful treatment of intrahepatic cholangiocarcinoma in a 39-year-old woman with Wilson’s disease. Twenty-two years after being diagnosed with Wilson’s disease, intrahepatic cholangiocarcinoma was diagnosed. She had an intrahepatic mass that was found to be a 4.3-cm ill-defined hypodense lesion in liver segment 3/4, with features suggesting infiltrative intrahepatic cholangiocarcinoma rather than hepatocellular carcinoma. Laboratory results showed slightly elevated liver enzymes and tumor markers. There was no evidence of metastasis on chest computed tomography or positron emission tomography, and the tumor was resectable, so surgery was the first-choice treatment option. Left hepatectomy was performed successfully, and the final pathology confirmed adenocarcinoma with clear resection margins. The patient received adjuvant chemotherapy with capecitabine. To date, the patient has been doing well without evidence of recurrence or metastasis.
CONCLUSIONS: Despite limited knowledge regarding hepatic malignancy in Wilson’s disease, it is crucial to prioritize careful monitoring and develop suitable treatment strategies upon diagnosis to achieve favorable outcomes, considering the potential occurrence of intrahepatic cholangiocarcinoma in Wilson’s disease.
Keywords: Cholangiocarcinoma, Hepatolenticular Degeneration, case reports, Female, Humans, adult, Bile Ducts, Intrahepatic, Bile Duct Neoplasms
Background
Wilson’s disease (WD) is characterized by the accumulation of excessive copper in vital organs such as the liver, brain, and kidneys [1]. This autosomal recessive disorder, which affects copper metabolism, occurs in approximately 1 in 30 000 individuals [2]. It causes progressive liver damage and neurological impairment [3]. WD presents a wide spectrum of clinical manifestations related to hepatic disease, ranging from asymptomatic patients to acute liver failure [4,5]. Hepatobiliary malignancies are common in other chronic liver diseases. Hepatobiliary malignancies appear to be uncommon in WD, even among patients with cirrhosis, although the exact reasons for this finding are unclear. Contradictory theories exist regrading copper accumulation in the liver: some propose it has a protective effect against carcinoma through chromatin stabilization, while others associate it with elevated hepatic malignancy risk [6–8].
There have been occasional reports of hepatocellular carcinoma (HCC) in WD. However, the incidence of intrahepatic cholangiocarcinoma (IHCC) is even rarer in this condition [1]. Here, we report a case of successful treatment of IHCC in a 39-year-old woman and discuss IHCC in WD.
Case Report
A 39-year-old woman diagnosed with IHCC was referred for surgery. She was first diagnosed with WD at the age of 17 years and had been prescribed D-penicillamine for 10 years and had been taking zinc acetate, without any specific findings. A 19-mm intrahepatic mass was suspected on follow-up ultrasonography 22 years after the WD diagnosis.
Further evaluation was conducted with computed tomography (CT) and magnetic resonance imaging (MRI). CT showed an ill-defined, 4.3-cm, hypodense lesion in the liver in segment (S) 3/4, heterogenous hypodense on arterial phase and portal venous phase, heterogenous hyperdense on delayed phases, encasement of umbilical portion of the left portal vein, mild dilation of the bile duct(B)3 and B4, and no enlarged lymph nodes in the abdomen (Figure 1). MRI showed a 4.3-cm mass in the liver S3/S4, delayed centripetal enhancement, invasion of the umbilical portion of the left portal vein, mild dilatation of the B3/B4, and targetoid diffusion restriction. These findings were indicative of infiltrative intrahepatic cholangiocarcinoma rather than hepatocellular carcinoma (Figure 2).
The laboratory findings were as follows: alanine transaminase, 45 (normal range: 0–35 U/L); aspartate transaminase, 46 (normal range: 0–35 U/L); alkaline phosphatase, 96 (normal range: 30–120 U/L); total bilirubin, 0.8 (normal range: 0.2–1.2 mg/dL); prothrombin time (INR) 1.11 (normal range: 0.85–1.2 INR); alpha-fetoprotein, 9.32 (normal range: 0–7 ng/mL); carcinoembryonic antigen, 0.546 (normal range: 0–5 ng/mL); and carbohydrate antigen 19–9, 46.6 (normal range: 0–27 U/mL). There was no evidence of metastasis on chest CT or positron emission tomography, and left hepatectomy was the first treatment of choice.
Left hepatectomy was performed for 120 min, and the IHCC was located in segments 2, 3, and 4 grossly, with minimal liver cirrhosis. During surgery, there was no evidence of metastasis, significant lymph node involvement, or intraoperative complications. The patient was discharged after 7 days without any postoperative complications. Upon histological examination, the mass was diagnosed as a cholangiocarcinoma, moderately differentiated. Grossly, it was an ill-defined mass with dimensions of 42 mm×26 cm with low- to high-grade biliary intraepithelial neoplasia. Clear bile and parenchymal resection margins were also observed (margin of clearance: 0.9×0.8 cm). Immunochemical studies showed positive results for cytokeratin 7, 19, and P53 (Figure 3).
As adjuvant chemotherapy, 8 cycles of capecitabine were administered postoperatively, and the patient remained healthy without recurrence for 15 months after surgery (Figure 4).
Discussion
Hepatic malignancy, including HCC and IHCC, is common in patients with liver disease, regardless of whether they have cirrhosis [9]. Although chronic liver damage is a potential risk factor for primary liver cancer, it appears to be rare in WD, accounting for approximately 1.2% of cases [1]. The incidence of HCC and IHCC in individuals with WD is 0.7% and 0.5%, respectively. Although the exact reasons for the low incidence of primary liver cancer in WD remain unclear and controversial, some theories suggest that the accumulation of excessive copper in the liver protects against carcinoma development, and this mechanism is related to the stabilization of cellular chromatin [1,6–8]. However, other studies have demonstrated that accumulation of copper increases susceptibility to HCC, a risk that can potentially be prevented through the administration of D-penicillamine. This research suggests that carcino-genesis may arise from liver damage, subsequently leading to chronic inflammation and cirrhosis due to chronic copper accumulation [10,11]. Recent research indicates that certain genetic variations, particularly the c.3972C > T variant at exon 28 of the
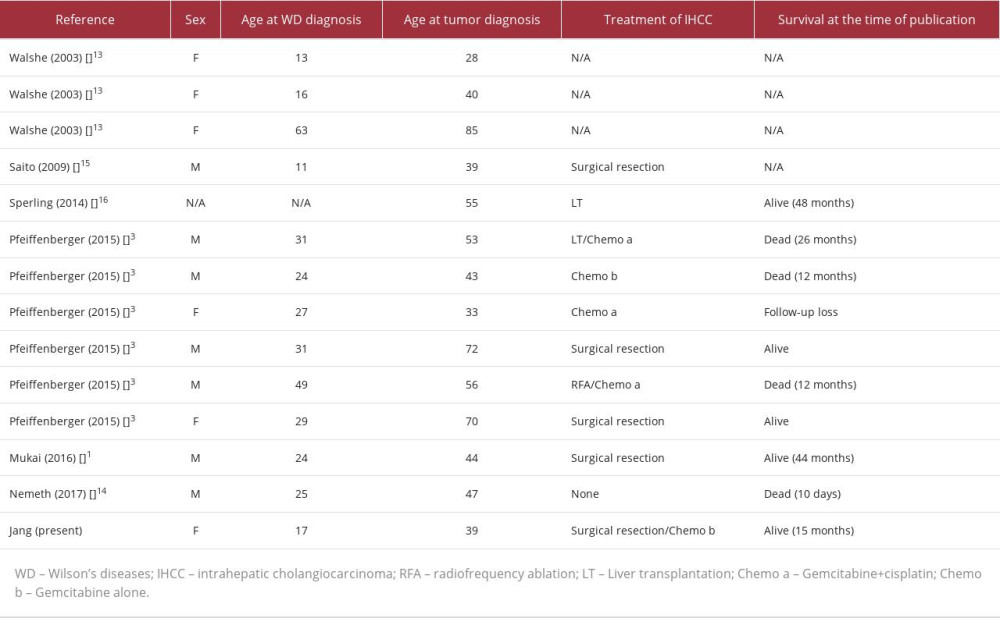
Among the hepatic malignancies, there are fewer IHCC cases in WD, with only 14 cases reported to date, including the present study. Two of the reports were cohort studies [3,13], whereas the others were case reports [1,14–16]. Although details for these cases were not included, 7 of the 14 cases were treated surgically, including our case, and various treatments for the remaining cases were applied, including liver transplantation, radiofrequency ablation, and chemotherapy according to the tumor and liver condition at the time of diagnosis (Table 1).
Generally, IHCC is the second most common malignancy arising from the liver, accounting for 3% of all cases of gastrointestinal cancer [17]. Liver resection is a fundamental component of any curative treatment approach for IHCC, although only approximately 20% of cases are eligible for resection at the time of diagnosis [18]. The primary objective of surgery is to achieve a complete margin-negative resection (R0) while considering the functional capacity of the liver and ensuring oncologic radicality. These factors, along with the patient’s performance status, are crucial determinants of surgical intervention success. In the present case, the tumor was resectable by the time of diagnosis, and the sufficient residual liver volume and oncologic radicality were obtained for successful treatment. Oncological radicality pertains to the goal of achieving complete removal of the tumor, often including the removal of surrounding tissues and lymph nodes to minimize the risk of cancer recurrence.
Approximately 50% of patients with WD have liver nodules detected on imaging, most of which are typically benign [19]. Although it is a very low probability, the livers of patients with WD should be carefully followed up owing to the possibility of hepatic malignancy. Close observation enables early detection, which, in turn, facilitates timely surgical intervention and contributes to favorable outcomes, as observed in the present case.
Conclusions
In summary, despite the limited understanding of the causes and rarity of hepatic malignancy in WD, owing to the possibility of IHCC, careful monitoring should be implemented and appropriate treatment plans established by the time of diagnosis for good outcomes.
Figures
References:
1.. Mukai Y, Wada H, Eguchi H, Intrahepatic cholangiocarcinoma in a patient with Wilson’s disease: A case report: Surg Case Rep, 2016; 2; 29
2.. Merle U, Schaefer M, Ferenci P, Clinical presentation, diagnosis and long-term outcome of Wilson’s disease: A cohort study: Gut, 2007; 56; 115-20
3.. Pfeiffenberger J, Mogler C, Gotthardt DN, Hepatobiliary malignancies in Wilson disease: Liver Int, 2015; 35; 1615-22
4.. Medici V, LaSalle JM, Genetics and epigenetic factors of Wilson disease: Ann Transl Med, 2019; 7(Suppl. 2); S58
5.. Roelofsen H, Wolters H, Van Luyn MJ, Copper-induced apical trafficking of ATP7B in polarized hepatoma cells provides a mechanism for biliary copper excretion: Gastroenterology, 2000; 119; 782-93
6.. Meacham KA, Cortés MP, Wiggins EM, Altered zinc balance in the Atp7b(-/-) mouse reveals a mechanism of copper toxicity in Wilson disease: Metallomics, 2018; 10; 1595-606
7.. Geierstanger BH, Kagawa TF, Chen SL, Base-specific binding of copper(II) to Z-DNA. The 1.3-A single crystal structure of d(m5CGUAm 5CG) in the presence of CuCl2: J Biol Chem, 1991; 266; 20185-91
8.. Wilkinson ML, Portmann B, Williams R, Wilson’s disease and hepatocellular carcinoma: Possible protective role of copper: Gut, 1983; 24; 767-71
9.. Schroeder SM, Matsukuma KE, Medici V, Wilson disease and the differential diagnosis of its hepatic manifestations: A narrative review of clinical, laboratory, and liver histological features: Ann Transl Med, 2021; 9; 1394
10.. Jong-Hon K, Togashi Y, Kasai H, Prevention of spontaneous hepato-cellular carcinoma in Long-Evans cinnamon rats with hereditary hepatitis by the administration of d-penicillamine: Hepatology, 1993; 18; 614-20
11.. van Meer S, de Man RA, van den Berg AP, No increased risk of hepatocellular carcinoma in cirrhosis due to Wilson disease during long-term follow-up: J Gastroenterol Hepatol, 2015; 30; 535-39
12.. Brandi G, Rizzo A, Deserti M, Wilson disease, ABCC2 c.3972C > T polymorphism and primary liver cancers: Suggestions from a familial cluster: BMC Med Genet, 2020; 21; 225
13.. Walshe JM, Waldenström E, Sams V, Abdominal malignancies in patients with Wilson’s disease: QJM, 2003; 96; 657-62
14.. Németh D, Folhoffer A, Smuk G, Cholangiocarcinoma in Wilson’s disease – a Case Report: J Gastrointestin Liver Dis, 2017; 26; 305-8
15.. Saito T, Watanebe M, Tanaka H, Cholangiocellular carcinoma associated with Wilson’s disease: Jpn J Gastroenterol Surg, 2009; 42; 1574-79
16.. Sperling J, Justinger C, Schuld J, Intrahepatic cholangiocarcinoma in a transplant liver – selective internal radiation therapy followed by right hemihepatectomy: Report of a case: World J Surg Oncol, 2014; 12; 198
17.. Hoyos S, Navas M-C, Restrepo J-C, Current controversies in cholangiocarcinoma: Biochim Biophys Acta Mol Basis Dis, 2018; 1864; 1461-67
18.. Krenzien F, Nevermann N, Krombholz A, Treatment of intrahepatic cholangiocarcinoma – a multidisciplinary approach: Cancers (Basel), 2022; 14; 362
19.. Dohan A, Vargas O, Dautry R, MR imaging features of focal liver lesions in Wilson disease: Abdom Radiol (NY), 2016; 41; 1811-24
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133