17 December 2020: Articles 
Sedation After a Trial of Mixed Amphetamine Salts in a Boy with Attention-Deficit/ Hyperactivity Disorder
Unusual or unexpected effect of treatment, Unexpected drug reaction
Raed Al Awami1EF, Ammar Albanna23AEF*DOI: 10.12659/AJCR.928269
Am J Case Rep 2020; 21:e928269
Abstract
BACKGROUND: Attention-deficit/hyperactivity disorder (ADHD) is a common neurodevelopmental disorder that manifests in early childhood. Pharmacotherapy, including psychostimulants, is considered the cornerstone of ADHD management. Although stimulants have been associated with adverse effects, sedation following the administration of an amphetamine-based stimulant is an extremely rare adverse effect.
CASE REPORT: We report the case of a 6-year-old boy presenting with ADHD and a history of autism spectrum disorder (ASD). After discussing treatment options with his parents, he was started on a low dose of a methylphenidate medication. He was unable to tolerate the medication due to anorexia, insomnia, and irritability despite multiple adjustments in the dosages. A trial of immediate-release mixed amphetamine salts was initiated, starting from a low dose. The boy developed sedation and lethargy shortly after the administration of this medication.
CONCLUSIONS: Sedation is a rare adverse drug reaction to mixed amphetamine salts. Clinicians should proactively monitor for the possible adverse effects in patients with ASD and ADHD, including unexpected symptoms such as sedation. Reporting of adverse drug reactions should be encouraged to promote the post-marketing surveillance of medications.
Keywords: Amphetamine, Attention Deficit Disorder with Hyperactivity, Central Nervous System Stimulants, Child Development Disorders, Pervasive, Dextroamphetamine, Drug-Related Side Effects and Adverse Reactions, case reports, Autism Spectrum Disorder, Child, Methylphenidate, Salts
Background
Attention-deficit/hyperactivity disorder (ADHD) is a neurodevelopmental disorder that manifests in childhood with symptoms of inability to sustain attention, hyperactivity, and/or impulsivity, that could affect behavioral, academic, and social functioning [1]. Other psychiatric conditions commonly coexist with ADHD, including oppositional defiant disorder, anxiety disorders, depression, and learning disorders.
ADHD is a chronic condition that requires active family involvement in setting-up treatment goals and ongoing management. Evidence-based treatment options for ADHD typically include behavioral modifications and pharmacotherapy [2]. Among pharmacological options, stimulants are considered as the first-line treatment for ADHD. While stimulants are considered safe, they may have some potential adverse effects. Here, we describe the case of a child with ADHD and comorbid autism spectrum disorder (ASD) who developed a rare adverse effect of sedation after a trial of immediate-release mixed amphetamine salts for ADHD.
Case Report
We present the case of a 6-year-old boy who was first brought to our outpatient psychiatry clinic, within a tertiary care pediatric hospital, when he was 3 years of age due to parental concerns regarding speech and language delay. His speech was limited to single words and there was absence of progression in his language acquisition since the age of 18 months. His parents had a stable relationship and there was no history of expressive language delay in his family. His medical history and surgical history were unremarkable. He was born at 32 weeks of gestation and was delivered via emergency lower-segment cesarean section due to fetal distress and was admitted for a few days to a neonatal intensive care unit due to respiratory distress. He was discharged in a healthy state and he was generally healthy during his childhood. His gross and fine motor skills are appropriate for his age.
The child did not display any dysmorphic features and his growth parameters were within normal limits. He underwent a comprehensive audiology assessment, which revealed normal hearing bilaterally. Following structured comprehensive assessment, including administering the Second Edition of the Autism Diagnostic Observation Schedule (ADOS-2) [3], he was diagnosed with ASD. In addition to his speech delay, he exhibited restricted patterns of behavior with excessive adherence to routines and resistance to change that significantly impaired his daily functioning. Hence, he was enrolled in an early behavioral intervention program based on applied behavior analysis.
At the age of 6 years, his parents brought him in for evaluation of comorbid and impairing symptoms, including significant difficulty in sustaining attention, and significant hyperactivity and impulsivity symptoms. These symptoms occurred in multiple environments and were present for more than 6 months. Based on parental feedback, and feedback from his teachers, in addition to the clinical assessment, the child met DSM-5 criteria for comorbid ADHD [1]. This was comprehensively explained to the parents and the management options, including behavioral interventions and pharmacological treatments, were discussed. The patient was prescribed a small dose of methylphenidate medication, both in long-acting form (osmotic-controlled release oral delivery system methylphenidate, 18 mg) and short-acting forms (immediate-release methylphenidate, 2.5 mg). The child was unable to tolerate these medications due to irritability, insomnia, and anorexia. Hence, he was switched to immediate-release mixed amphetamine salts (Adderall® XR) at a starting dose of 5 mg. His parents reported that at about 1 hour after initiating the first dose, the child became sedated and lethargic. He had decreased arousal and developed excessive sleepiness. Hence, his parents did not give him any further doses and sought medical attention. After 1 day, the medication was re-administered in immediate-release form Adderall® IR at 1.25 mg and the child exhibited the same reaction of becoming sleepy and had minimal responsiveness. The parents reported giving the medication at 10 a.m. and the child was brought to the clinic at 2 p.m. for evaluation. The parents confirmed that the child had not received any medication other than Adderall® IR. His vital signs were within the normal limits and there was no other medical condition explaining his decreased activity. Due to parental concerns and possible adverse effects, the medication was then changed to atomoxetine 10 mg twice/day, on which the patient showed moderate improvement as reported by the parents in the next follow-up visits.
Discussion
We describe here the case of a 6-year-old boy with ADHD and ASD who developed lethargy and sedation after a trial of immediate-release mixed amphetamine salts, which is an extremely rare adverse effect.
In school-age children, stimulants are often used as the first-line treatment for ADHD. While the exact mechanism for stimulants in ADHD is not entirely clear, stimulants increase dopamine and norepinephrine in the prefrontal cortex, which enhances the efficiency of information processing by pyramidal neurons, resulting in improvement in the symptoms of ADHD [4].
Stimulants, including methylphenidate and amphetamines, are generally considered safe and have a well-established efficacy profile [5]. Most adverse effects are mild and can be ameliorated by adjustments of doses and dosing intervals [6,7]. Common adverse effects include anorexia, weight loss, poor growth, and emotional lability [7]. Despite the concerns of their cardiovascular adverse effects, stimulants were not shown to have increased risk of serious cardiac complications, including sudden cardiac death. Moreover, stimulants are known to cause small elevations in the heart rate and blood pressure [8]. Rare adverse effects include priapism, psychosis, tics, and potential misuse [9–11].
In contrast to other agents, including methylphenidate, atomoxetine, clonidine, and guanfacine [12–15], there is no randomized controlled trial for the use amphetamines for the treatment of ADHD and ASD [16]. Prior studies suggested that children with ASD and ADHD have a lower response rate to methylphenidate, but also exhibit a higher rate of adverse effects to methylpheni-date [12,17–19]. For example, the discontinuation of methylphenidate due to adverse effects in a clinical trial was 18%, which was 10 times higher than the in the control group [19]. While the reason for this higher rate of adverse effects remains unclear, there is some evidence suggesting that patients with ASD may have aberrant GABAergic neural systems that can cause paradoxical responses to medications [20]. Similarly, the present case may suggest a possible alteration of catecholaminergic neural systems in patients with ASD.
It is well known that the discontinuation of stimulants can result in rebound symptoms, including increased appetite and excessive sleepiness [21]. In the present case, however, the patient experienced lethargy and sedation shortly after the administration of the medication and after a washout period from the previous medication, which is very unusual. This unusual adverse effect was previously reported with the use of lisdexamfetamine, a prodrug of d-amphetamine, in ADHD [22].
It is possible that the present patient had a fast metabolism profile resulting in rapid alterations in the drug blood level, and the observed sedation could be a symptom of withdrawal from stimulants [23]. However, it should be noted that the patient had received a prolonged stimulatory-effect methylphenidate, which has similar pharmacokinetic characteristics as amphetamines. Additionally, dizziness has been reported in association with stimulants [24]. It is reasonable that the child developed this adverse effect, but he was not able to express it due to his language and social communication disorder. Furthermore, the paradoxical depressant effect has been experimentally demonstrated with the administration of low doses of stimulants [25].
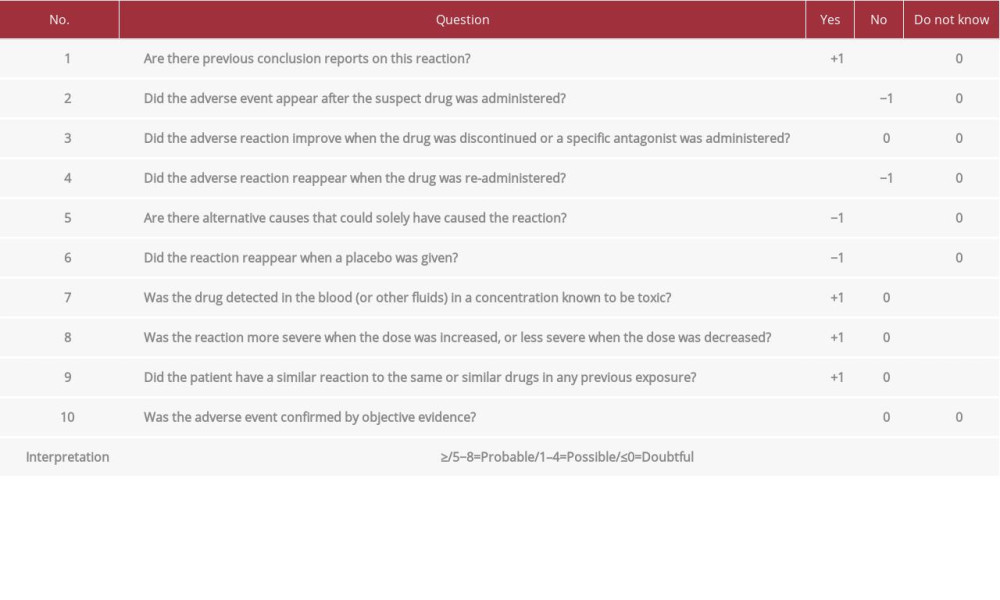
Recognition of adverse drug reactions is essential in promoting patient safety. Accurate attribution of causality of such reactions in daily practice could be challenging considering that the assessment often relies on subjective judgment which may falsely decrease the assessment of the safety of the medication. Naranjo et al. [26] developed a scale to allow for an objective evaluation of adverse drug reactions. A careful assessment of the present patient using the Naranjo algorithm revealed that sedation is a definite adverse drug reaction to the mixed amphetamine salts (Table 1).
Conclusions
Sedation is a rare adverse drug reaction of mixed amphetamine salts in patients. Such paradoxical reactions to amphetamines should be noted in the patient information leaflet if encountered in additional patients. Clinicians should have proactive monitoring for the possible adverse effects in patients with ASD and ADHD. Reporting of adverse drug reactions should be encouraged in order to promote the post-marketing surveillance of medications.
References:
1.. , 2013
2.. Pliszka S, Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder: J Am Acad Child Adolesc Psychiatry, 2007; 46(7); 894-921
3.. Lord C, Risi S, Lambrecht L, The Autism Diagnostic Observation Schedule– Generic: A standard measure of social and communication deficits associated with the spectrum of autism: J Autism Dev Disord, 2000; 30(3); 205-23
4.. Stahl SM, Mechanism of action of stimulants in attention-deficit/hyperactivity disorder: J Clin Psychiatry, 2010; 71(1); 12-13
5.. Clavenna A, Bonati M, Safety of medicines used for ADHD in children: A review of published prospective clinical trials: Arch Dis Child, 2014; 99(9); 866-72
6.. Wolraich ML, Hagan JF, Allan C, Clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents: Pediatrics, 2019; 144(4); e20192528
7.. Storebø OJ, Pedersen N, Ramstad E, Methylphenidate for attention deficit hyperactivity disorder (ADHD) in children and adolescents-assessment of adverse events in non-randomised studies: Cochrane Database Syst Rev, 2018(5); CD012069
8.. Hennissen L, Bakker MJ, Banaschewski T, Cardiovascular effects of stimulant and non-stimulant medication for children and adolescents with ADHD: A systematic review and meta-analysis of trials of methylphenidate, amphetamines and atomoxetine: CNS Drugs, 2017; 31(3); 199-215
9.. Moran LV, Ongur D, Hsu J, Psychosis with methylphenidate or amphetamine in patients with ADHD: N Engl J Med, 2019; 380(12); 1128-38
10.. Kelly BD, Lundon DJ, McGuinness D, Brady CM, Methylphenidate-induced erections in a prepubertal child: J Pediatr Urol, 2013; 9(1); e1-2
11.. Wilens TE, Gignac M, Swezey A, Characteristics of adolescents and young adults with ADHD who divert or misuse their prescribed medications: J Am Acad Child Adolesc Psychiatry, 2006; 45(4); 408-14
12.. Pearson DA, Santos CW, Aman MG, Effects of extended release methylphenidate treatment on ratings of attention-deficit/hyperactivity disorder (ADHD) and associated behavior in children with autism spectrum disorders and ADHD symptoms: J Child Adolesc Psychopharmacol, 2013; 23(5); 337-51
13.. Harfterkamp M, van de Loo-Neus G, Minderaa RB, A randomized double-blind study of atomoxetine versus placebo for attention-deficit/hyper-activity disorder symptoms in children with autism spectrum disorder: J Am Acad Child Adolesc Psychiatry, 2012; 51(7); 733-41
14.. Jaselskis CA, Cook EH, Fletcher KE, Leventhal BL, Clonidine treatment of hyperactive and impulsive children with autistic disorder: J Clin Psychopharmacol, 1992; 12(5); 322-27
15.. Politte LC, Scahill L, Figueroa J, A randomized, placebo-controlled trial of extended-release guanfacine in children with autism spectrum disorder and ADHD symptoms: An analysis of secondary outcome measures: Neuropsychopharmacology, 2018; 43(8); 1772-78
16.. Howes OD, Rogdaki M, Findon JL, Autism spectrum disorder: Consensus guidelines on assessment, treatment and research from the British Association for Psychopharmacology: J Psychopharmacol, 2018; 32(1); 3-29
17.. Quintana H, Birmaher B, Stedge D, Use of methylphenidate in the treatment of children with autistic disorder: J Autism Dev Disord, 1995; 25(3); 283-94
18.. Handen BL, Johnson CR, Lubetsky M, Efficacy of methylphenidate among children with autism and symptoms of attention-deficit hyperactivity disorder: J Autism Dev Disord, 2000; 30(3); 245-55
19.. Research Units on Pediatric Psychopharmacology Autism N, Randomized, controlled, crossover trial of methylphenidate in pervasive developmental disorders with hyperactivity: Arch Gen Psychiatry, 2005; 62(11); 1266-74
20.. Bruining H, Passtoors L, Goriounova N, Paradoxical benzodiazepine response: A rationale for bumetanide in neurodevelopmental disorders?: Pediatrics, 2015; 136(2); e539-43
21.. Nishino S, Rebound hypersomnolence, stimulant abuse, and DAT-mediated dopamine release: Sleep, 2009; 32(11); 1407-9
22.. Majeed MH, Wasiq S, Mumtaz A, A case of sedation secondary to a trial of lisdexamfetamine: Prim Care Companion CNS Disord, 2019; 21(3); 18 l02396
23.. Weiss MD, Surman CB, Elbe D, Stimulant ‘rapid metabolizers’: Wrong label, real phenomena: Atten Defic Hyperact Disord, 2018; 10(2); 113-18
24.. Tatro DS, Drug interactions facts, facts and comparisons division: JB Lippincott Company, 2006
25.. Tecce JJ, Cole JO, Amphetamine effects in man: paradoxical drowsiness and lowered electrical brain acitivity (CNV): Science, 1974; 185(4149); 451-53
26.. Naranjo CA, Busto U, Sellers EM, A method for estimating the probability of adverse drug reactions: Clin Pharmacol Ther, 1981; 30(2); 239-45
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133