01 June 2022: Articles 
Unusual Case of Dehydration Leading to Severe Symptomatic Hypercalcemia
Unknown etiology
Ojbindra KC1ABCDEF*, Punya Hari DahalDOI: 10.12659/AJCR.936204
Am J Case Rep 2022; 23:e936204
Abstract
BACKGROUND: Severe hypercalcemia is a life-threatening medical emergency. Its possible etiologies are hyperparathyroidism, malignancy, chronic granulomatous diseases, vitamin D intoxication, medication (lithium, thiazine, excessive vitamin A), hyperthyroidism, acromegaly, adrenal insufficiency, pheochromocytoma, milk-alkali syndrome, and immobilization. Dehydration is considered a consequence of hypercalcemia but not the etiology.
CASE REPORT: We present a case of a 50-year-old woman who presented with headache, nausea, dizziness, and profound weakness, with a serum calcium level of 17.3 mg/dL due to severe dehydration. The other causes of hypercalcemia were diligently excluded with extensive laboratory testing. The patient’s calcium level improved with aggressive intravenous hydration. In the subsequent follow-up visits, the calcium level remained within the reference range. The pathophysiology of severe hypercalcemia caused by dehydration is not clear. However, a feedforward mechanism has been proposed to occur, which worsens both dehydration and hypercalcemia. Dehydration as an initial insult leads to mild or transient hypercalcemia due to decreased fluid volume that affects calcium excretion via the kidneys. Subsequently, hypercalcemia interferes with the kidney’s ability to concentrate urine, leading to further dehydration. This sets up a vicious loop that worsens both dehydration and hypercalcemia, leading to profound dehydration and severe hypercalcemia.
CONCLUSIONS: Dehydration is considered a consequence of hypercalcemia but has not been identified as the etiology of severe hypercalcemia. Hyperparathyroidism and malignancy are the most common causes of severe symptomatic hypercalcemia, and dehydration is the diagnosis of exclusion. However, it is imperative to keep dehydration in the differential diagnosis for a patient presenting with severe symptomatic hypercalcemia, as highlighted by our case.
Keywords: Dehydration, Hypercalcemia, Kidney Concentrating Ability, Female, Humans, Hyperparathyroidism, Middle Aged, Neoplasms
Background
Hypercalcemia is a metabolic condition of increased calcium level in the blood above the normal limits and can lead to significant clinical consequences [1]. The reference range of serum calcium level is 8.5 to 10.5 mg/dL, and hypercalcemia is defined as a serum calcium concentration that is 2 standard deviations above the mean [2]. Calcium plays an integral role in muscle contraction, nerve transmission, cell differentiation, enzyme activation, programmed cell death, and intracellular signaling in the human body [3]. The clinical manifestations depend on the degree of hypercalcemia, which ranges from being asymptomatic to having nonspecific symptoms including fatigue, depression, constipation to severe marked symptoms of polyuria, polydipsia, nausea, muscle weakness, and altered sensorium [1,2]. Hypercalcemia etiologies include primary hyper-parathyroidism, malignancy, chronic granulomatous diseases, vitamin D intoxication, medication (lithium, thiazine, excessive vitamin A), hyperthyroidism, acromegaly, adrenal insufficiency, milk-alkali syndrome, and immobilization [4,5]. It is rare that dehydration is the primary cause of severe hypercalcemia [6,7]. We present a case of a 50-year-old woman who presented with headache, nausea, dizziness, and profound weakness, with a serum calcium level of 17.3 mg/dL due to severe dehydration.
Case Report
A 50-year-old woman with a past medical history of chronic obstructive pulmonary disease, hypertension, hyperlipidemia, bipolar disorder, autoimmune hepatitis with cirrhosis, gastroesophageal reflux disease, hypothyroidism, and type 2 diabetes mellitus presented to the hospital with headache, dizziness, nausea, and weakness of 3 days. She reported that her home had been overheated because the air conditioner stopped working 1 week ago. She had been standing close to the freezer door and eating pieces of ice to decrease the heat. However, she reported feeling heated and sweating profusely throughout the day. Her home medications included albuterol inhaler, clonidine 0.2 mg twice a day, atorvastatin 40 mg nightly, quetiapine 300 mg nightly, fluoxetine 20 mg daily, benztropine 1 mg twice a day, prednisone 5 mg daily, pantoprazole 40 mg daily, furosemide 20 mg daily, levothyroxine 50 mcg daily, metformin 1000 mg twice a day, insulin degludec 16 units every morning, and exenatide 2 mg weekly. She had a history of not adhering to her prescription medication regimen and diabetic diet in the past. She reported no recent changes in her prescription medications and denied taking over-the-counter medications, including antacids, calcium, and vitamin A supplements.
In the Emergency Department, her initial vital signs were a blood pressure of 150/90 mmHg, temperature of 37.4°C, heart rate of 105 beats per min, respiratory rate of 18 breaths per min, and oxygen saturation of 99% on room air. The patient was lethargic and had dry mucus membranes on physical examination. The rest of the physical examination was unremarkable. The laboratory test results showed hyperglycemia, with 365 mg/dL blood glucose, and severe hypercalcemia, with a calcium level of 17.3 gm/dL and ionized calcium of 1.95 mM/L. She had hyperglycemia with a normal bicarbonate and anion gap, which suggested hyperglycemia without diabetic ketoacidosis, and hence blood gas was not done at the time of admission. The rest of the laboratory test results were unre-markable, as summarized in Table 1. The electrocardiogram revealed a normal sinus rhythm without ST-segment changes. She was administered a 2-liter fluid bolus of normal saline, and was then started on the continuous infusion of normal saline at 150 mL/h and admitted to the medical floor for further management.
The patient’s calcium level gradually improved with intravenous (i.v.) hydration and normalized by the third day of hospitalization (Figure 1). The hypercalcemia improved with i.v. hydration, and bisphosphonate therapy was not needed. An extensive laboratory workup, including parathyroid hormone (PTH), parathyroid hormone-related peptide, 25-hydroxy vitamin D, 1,25-dihydroxy vitamin D, thyroid-stimulation hormone, cortisol, serum protein electrophoresis, and serum-free light chain, was done to determine the probable causes of hypercalcemia. The results of these laboratory tests were unre-markable (Table 2). The i.v. hydration was stopped after her calcium level normalized on the third hospitalization day. The patient’s symptoms had resolved, and she was discharged home on the fourth day of hospitalization, with calcium levels in the reference range.
One week after discharge, she had a follow-up visit with her primary care physician. Additional workup was done, including mammography of bilateral breast, nuclear bone imaging of the whole body, and computed tomography scan of chest abdomen and pelvis, which were unremarkable. The calcium level was within the reference range 1 week after the patient’s discharge from the hospital (Table 1).
Discussion
Calcium hemostasis is tightly regulated in the human body by hormones including PTH, 1,25-dihydroxy vitamin D3 (calcitriol), and calcitonin, with their effect on the bones, gastrointestinal tract, and kidneys [8]. The skeletal system contains 99% of total body calcium, and the remaining 1% is in the extracellular fluid and soft tissues [8]. One-half of the circulating calcium is bound to albumin, globulin, and other inorganic molecules, and the other half is free ionized calcium, which bone resorption, increased gastrointestinal absorption, and decreased renal excretion, and 1,25-dihydroxy vitamin D3 has no direct effect on the bones and kidneys but increases calcium absorption from the gastrointestinal tract [8]. Calcitonin inhibits calcium resorption from the bones and promotes calcium excretion from the kidneys [8]. These hormones increase or decrease calcium levels depending on total body stores, which are primarily measured via calcium-sensing receptors in the parathyroid gland and renal tubules [8].
Hypercalcemia is a common clinical problem that can present various clinical manifestations depending on the degree of hypercalcemia (Table 3) [11]. There are many causes of hyper-calcemia, including primary hyperparathyroidism, malignancy, chronic granulomatous diseases, vitamin D intoxication, medication (lithium, thiazine, excessive vitamin A), hyperthyroidism, acromegaly, adrenal insufficiency, pheochromocytoma, milk-alkali syndrome, and immobilization [4,5]. Primary has physiological effects on the body [9]. The normal serum calcium level ranges from 8.5 to 10.5 mg/dL and ionized calcium from 1.15 to 1.27 mg/dL, although the exact range can vary among laboratories [10]. PTH increases serum calcium by hyperparathyroidism and malignancy are the most common causes, accounting for more than 90% of cases [4,5]. To the best of our knowledge, dehydration has been reported as a primary cause of severe hypercalcemia in only a handful of case reports in the literature [6,7].
Dehydration is a known consequence of moderate to severe hypercalcemia [11]. Hypercalcemia causes natriuresis and diabetes insipidus, which lead to dehydration [12]. Natriuresis and diabetes insipidus are proposed to occur with hypercalcemia by various mechanisms, such as calcium deposition in the medulla causing tubulointerstitial injury and activation of the calcium-sensing receptor, which directly impairs renal concentrating ability by affecting the loops of Henle and collecting tubules [13,14]. Severe dehydration is known to cause mild or transient hypercalcemia due to a decrease in fluid volume that affects calcium excretion via the kidneys [15]. However, severe hypercalcemia due to dehydration as the primary cause is rare [6,7]. The pathophysiology of severe hypercalcemia caused by dehydration is not clear. A feedforward mechanism has been proposed to occur, which worsens both dehydration and hypercalcemia. Dehydration as an initial insult leads to hypercalcemia, which interferes with the kidney’s ability to concentrate urine, leading to further dehydration and setting up a vicious loop that worsens dehydration and hypercalcemia, leading to profound dehydration and severe hypercalcemia [7].
In our case, the patient had severe dehydration due to excessive sweating from an overheated room and had poor oral intake. The patient also had poorly controlled diabetes mellitus with hyperglycemia, which might have worsened dehydration due to osmotic diuresis. The other causes of hypercalcemia were diligently excluded with extensive laboratory tests (Table 2). The PTH level was markedly reduced, which suggested non-PTH-mediated hypercalcemia and ruled out hyperparathyroidism. The low PTH level was due to feedback suppression from hypercalcemia. The patient’s 25-hydroxy vitamin D and vita-min 1,25-dihydroxy vitamin D3 levels were low, which ruled out vitamin D intoxication and granulomatous diseases as the causes of hypercalcemia. The low vitamin D level suggested vitamin D deficiency, which usually causes hypocalcemia. The use of atorvastatin has been reported to cause asymptomatic hypercalcemia [16]. However, in our patient, atorvastatin was continued during and after hospitalization. The calcium level remained within the reference range after discharge, although the patient continued to take atorvastatin, which ruled out atorvastatin as the cause of hypercalcemia. The patient’s calcium level improved with the i.v. hydration and correction of dehydration. In a subsequent follow-up visit with the patient’s primary care physician, her calcium level remained within the reference range.
Conclusions
Severe hypercalcemia is a life-threatening medical emergency as it can lead to life-threatening cardiac arrhythmias or neurological complications such as stupor or coma. Dehydration is considered a consequence of hypercalcemia but has not been identified as the etiology of severe hypercalcemia. Hyperparathyroidism and malignancy are the most common causes of severe symptomatic hypercalcemia, which must be ruled out with thorough investigations, because dehydration is the diagnosis of exclusion. However, it is imperative to keep dehydration in the differential diagnosis in a patient presenting with severe symptomatic hypercalcemia, as highlighted by our case.
References:
1.. Shane E, Irani D, Hypercalcemia: Pathogenesis, clinical manifestations, differential diagnosis, and management: Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism, 2006; 6; 176-80, Washington, DC Available from: http://www.homepages.ucl.ac.uk/~ucgatma/Anat3048/PAPERS%20etc/ASBMR%20Primer%20Ed%206/Ch%2026-41%20-%20Disorders%20of%20Serum%20Minerals.pdf
2.. Sadiq NM, Naganathan S, Badireddy M, Hypercalcemia: StatPearls [Internet] Sep 11, 2021, Treasure Island (FL), StatPearls Publishing Jan. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430714/
3.. Pu F, Chen N, Xue S, Calcium intake, calcium homeostasis and heath. Food Sci Hum: Wellness, 2016; 5; 8-16
4.. Lafferty FW, Differential diagnosis of hypercalcemia: J Bone Miner Res, 1991; 6; S51-59
5.. Jacobs TP, Bilezikian JP, Rare causes of hypercalcemia: J Clin Endocrinol Metab, 2005; 90; 6316-22
6.. Fernandes LG, Ferreira NR, Cardiga R, Póvoa P, Severe hypercalcaemia and colon ischaemia: Dehydration as an unusual cause: BMJ Case Rep, 2015; 2015; bcr2014208809
7.. Acharya R, Winters DM, Rowe C, An unusual case of severe hypercalcemia: As dehydrated as a bone: J Community Hosp Intern Med Perspect, 2021; 11; 135-38
8.. Mundy GR, Guise TA, Hormonal control of calcium homeostasis: Clin Chem, 1999; 45; 1347-52
9.. Moore EW, Ionized calcium in normal serum, ultrafiltrates, and whole blood determined by ion-exchange electrodes: J Clin Invest, 1970; 49; 318-34
10.. Sinton TJ, Cowley DM, Bryant SJ, Reference intervals for calcium, phosphate, and alkaline phosphatase as derived on the basis of multichannel-analyzer profiles: Clin Chem, 1986; 32; 76-79
11.. Inzucchi SE, Understanding hypercalcemia: its metabolic basis, signs, and symptoms: Postgrad Med, 2004; 115; 69-76
12.. Christ-Crain M, Bichet DG, Fenske WK, Diabetes insipidus: Nat Rev Dis Primers, 2019; 5; 1-20
13.. Rosen S, Greenfeld Z, Bernheim J, Hypercalcemic nephropathy: Chronic disease with predominant medullary inner stripe injury: Kidney Int, 1990; 37; 1067-75
14.. Hebert SC, Extracellular calcium-sensing receptor: Implications for calcium and magnesium handling in the kidney: Kidney Int, 1996; 50; 2129-39
15.. , Hypercalcemia Mayo Clinic website [online] March 2022 [cited 2022 Jan 11]. Available from: https://www.mayoclinic.org/diseases-conditions/hypercalcemia/symptoms-causes/syc-20355523
16.. Ipekci SH, Baldane S, Sozen M, Kebapcilar L, Can atorvastatin calcium cause asymptomatic hypercalcemia?: Arch Turk Soc Cardiol, 2014; 42; 662-66
Tables
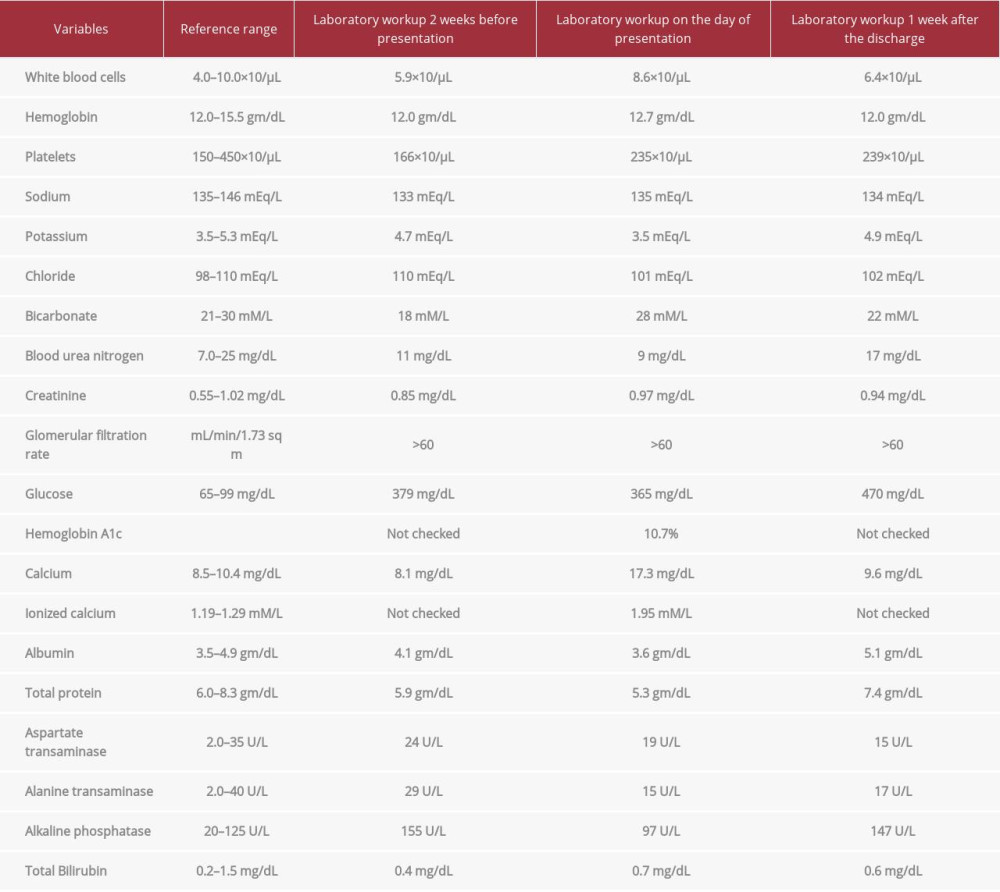 Table 1.. The laboratory test results at 2 weeks before presentation, on the day of presentation, and 1 week after discharge.
Table 1.. The laboratory test results at 2 weeks before presentation, on the day of presentation, and 1 week after discharge.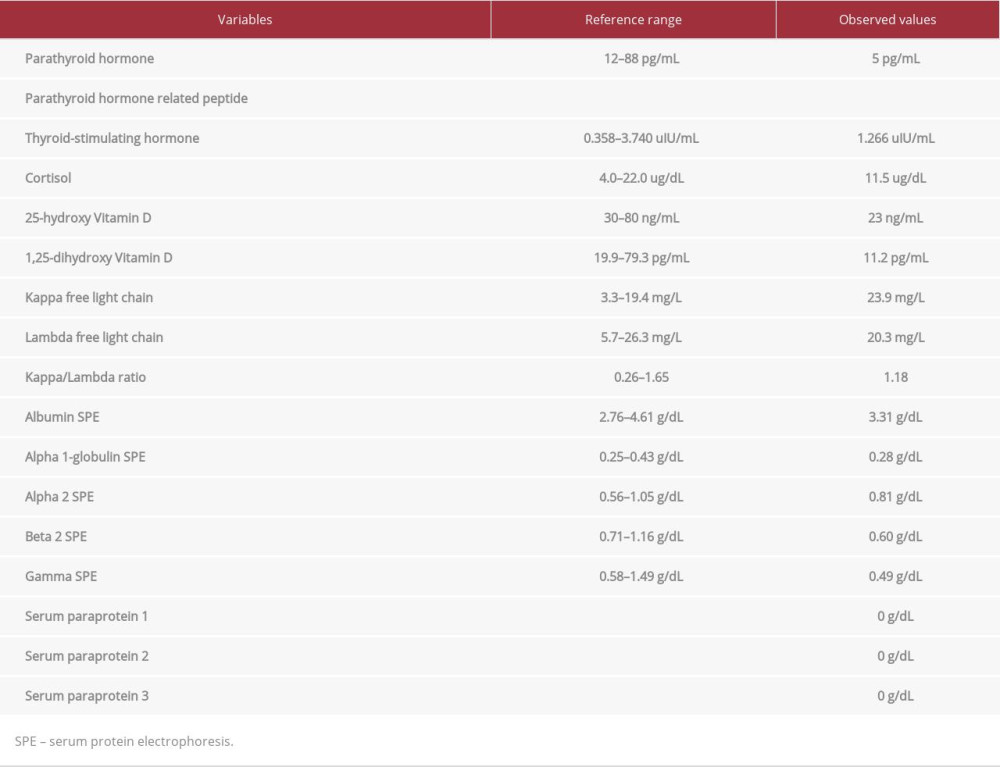 Table 2.. The laboratory workup to determine common causes of hypercalcemia.
Table 2.. The laboratory workup to determine common causes of hypercalcemia.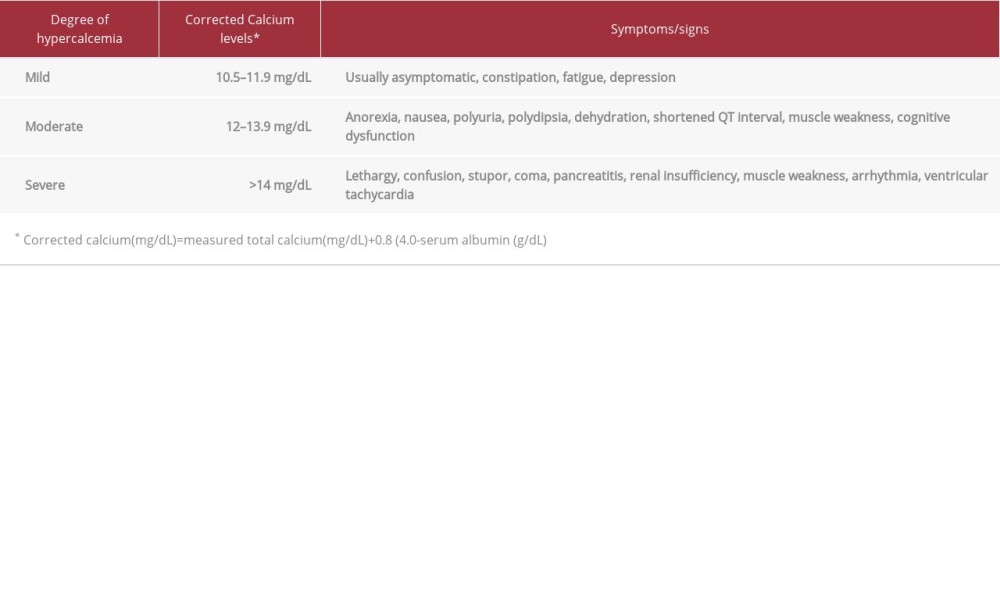 Table 3.. The degree of hypercalcemia with clinical manifestations.
Table 3.. The degree of hypercalcemia with clinical manifestations. Table 1.. The laboratory test results at 2 weeks before presentation, on the day of presentation, and 1 week after discharge.
Table 1.. The laboratory test results at 2 weeks before presentation, on the day of presentation, and 1 week after discharge. Table 2.. The laboratory workup to determine common causes of hypercalcemia.
Table 2.. The laboratory workup to determine common causes of hypercalcemia. Table 3.. The degree of hypercalcemia with clinical manifestations.
Table 3.. The degree of hypercalcemia with clinical manifestations. In Press
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943010
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943687
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943070
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943370
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250








