02 August 2020: Articles 
Takayasu Arteritis Associated with Latent Tuberculosis Infection: A 39-Year-Old Woman Is the First Case in Gabon
Challenging differential diagnosis, Management of emergency care, Patient complains / malpractice, Rare disease
Guignali Laurette Mangouka1ABCDEF, Berthe Amélie Iroungou2ABCDEF, Berthold Bivigou-Mboumba3BCDEF*, Davy Ngabou4DF, El Medhi Badidi Moulay5ADF, Abdelatif Darbi6DF, Jean Raymond Nzenze1AEFGDOI: 10.12659/AJCR.920195
Am J Case Rep 2020; 21:e920195
Abstract
BACKGROUND: Takayasu disease is a rare disease affecting large vessels, particularly the aorta and its main branches. The affected arteries become partially occluded. The disease is more frequent in women under 40 years of age, with a ratio of women to men of 8: 1.
CASE REPORT: We report the case of a 39-year-old woman who was admitted to the medical ward at the Military Hospital in Gabon. She presented with multiple aneurysms, arterial stenosis, and a positive tuberculin skin test and was diagnosed with Takayasu disease associated with latent tuberculosis infection. This rare case is the first to be reported in Gabon, with a delay in diagnosis of approximately 7 months.
CONCLUSIONS: This is the first case of Takayasu arteritis that has been described in Gabon and has generated medical interest beyond the country regarding the diagnostic, therapeutic, and prognostic approach.
Keywords: latent tuberculosis, Takayasu arteritis, Aneurysm, Axillary Artery, computed tomography angiography, Constriction, Pathologic, delayed diagnosis, Gabon, Iliac Artery, Imaging, Three-Dimensional
Background
Takayasu disease, also called pulseless disease or aortic arch syndrome, is a granulomatous inflammatory arteritis which can affect the aorta and its main branches, as well as the innominate, brachiocephalic, carotid, subclavian, and renal arteries [1]. The affected arteries become partially occluded. The disease is more frequent in women under 40 years of age, with a ratio of women to men of 8: 1 [2]. It is rare in Europe and the United States, with 1–3 new cases per million population per year, but it is more frequent in Mexico and Japan. The African figure is unknown. Symptoms include pain and tenderness in the area of the affected arteries, fever, sweats, fatigue, weight loss, arthralgia, myalgia, and anemia. Vascular failure may be accompanied by headache, syncope, limb claudication, and angina. Hypertension may be present and suggests involvement of the renal arteries. Clinical results include loss of peripheral pulses, bruits, and blood pressure discrepancies between the 2 arms. Findings in the eye include amblyopia, blindness, cataract, and atrophy of the iris, optic nerve, and retina, and glaucoma. Laboratory results typically show normocytic and normochromic anemia, elevated erythrocyte sedimentation rate (ESR), and leucocytosis. The chest X-ray shows cardiomegaly and an enlarged aorta. Intravenous digital subtraction angiography may show aortic arch or branch stenosis (type I), atypical coarctation with descending aorta involvement (type II), or mixed imaging (type III). The treatment is primarily high-dose oral prednisolone (1–2 mg/kg per day). The 5-year survival rate in treated patients is 90%.
Case Report
We report the case of a 39-year-old Gabonese woman who was referred from the Surgical Department to the Internal Medicine Department of the Military Hospital in Gabon in September 2017. The patient had become symptomatic in July 2016 when she reported to the hospital with intermittent numbness and tingling in the left arm. This was followed by the appearance of a left axillary pulsatile mass. She was first seen and treated as an outpatient for multiple aneurysmal dilatations and arterial stenosis. The Doppler ultrasound, performed in August 2016, revealed a 5-cm aneurysm with a peripheral thrombus and additional 3-cm long dilatations of the humeral artery in its cephalic part towards the left axillary artery. The clinical and Doppler results justified admission to the Surgical Department of the Omar Bongo Ondimba Military Hospital (Omar Bongo Ondimba Army Training Hospital). The surgeon removed the left axillary aneurysm and inserted a prosthesis in late August 2016. The results of the laboratory tests are presented in Tables 1–3.
Histopathological results from surgical tissues showed multiple fibrin fragments mixed with polymorphic inflammatory infiltrate. One vessel was narrowed by fibrous thickening and contained macrophages and fibroblasts. The absence of a thrombus, granulomas, or giant cells was noted.
In February 2017, another left femoral pulsatile mass without fever appeared. There was no evidence of arthromyalgia, oral ulceration, or uveitis. However, the patient reported a weight loss of 15 kilograms (kg) in the past 3 years. She was admitted back for surgery in April 2017 and underwent a surgical resection of the left femoral aneurysm.
In September 2017, the patient, who already had a history of multiple aneurysms and 2 surgical procedures (left axillary and left femoral artery), was referred to the Department of Medicine for further assessment and care.
At the time of the examination, vital signs and results were: pulse 69 beats per min, blood pressure 195/174 mmHg on the left arm and blood pressure 155/92 mmHg on the right arm, and 99% oxygen saturation in ambient air. Her weight was 70 kg and her axillary temperature was 36.8°C. Heart sounds were normal, but there was no peripheral pulse (radial, femoral, popliteal, and tibial pulses were not palpable). There was a pulsatile mass around the right femoral artery and a loss of sensation along the left arm. We performed the tuberculin skin test (TST) using Sanofi Pasteur MSD’s TUBERTEST as recommended by Gabon’s Ministry of Health. According to the standard protocol, we used the Mantoux method for the intradermal injection of 0.1 ml (5 IU) of purified tuberculin protein derivative. TST was highly positive after 72 h, with an induration diameter of 30 mm revealing an inflammatory, erythematous, and phlyctenular reaction (Figure 1).
The thoraco-abdominal-pelvic CT scan and renal artery Doppler ultrasound showed no abnormalities.
To make the differential diagnoses, we considered infectious diseases and non-infectious diseases. In the differential diagnosis, infectious causes were excluded because there was no history of fever, diarrhea, or recent trips abroad. There was no heart murmur and no evidence of skin damage or urinary tract infection. For infectious endocarditis, the differential diagnosis was also considered, yet later dismissed, as there was no history of fever and the blood culture was aseptic. The transthoracic echocardiogram showed no abnormalities or valve lesions. In addition, histopathological analysis of the left axillary artery revealed no neutrophil infiltration1. In this context, there was no evidence which suggested sepsis or endocarditis. Moreover, the absence of clinical signs suggesting secondary syphilis and negative serological results (Treponema tests: TPHA/VDRL) excluded the diagnosis of syphilitic aortitis [2].
With regard to the non-infectious causes of Takayasu arteritis, we considered an atheromatous origin, but this was unlikely because of the age of the patient, the absence of dyslipidemia, and the absence of histological lesions suggesting atherosclerosis [3,4].
Autoimmune diseases were considered, but clinical history and results in addition to negative autoantibody test results excluded immune causes such as lupus erythematosus, rheumatoid arthritis, and granulomatosis with polyangiitis [5,6].
Bechet’s disease [7] was excluded due to the absence of genital ulceration, uveitis, venous thrombosis, and HLA-B52. The histological report of the affected vessels excluded the diagnosis of giant cell arteritis. Considering the patient’s relatively young age, the existence of intermittent vascular claudication of the left upper limbs, the absence of peripheral pulse, and the difference in systolic blood pressure greater than 10 mmHg between the 2 arms, we reached a clinical diagnosis of Takayasu disease [8–13].
In February 2017, we performed an angio-computed tomography (angio-CT) of the lower limbs with two-dimensional reformations images using the volume rendered technique (VRT) which showed arterial ectasia and stenosis of the lower limbs (Figure 2). There was an ectasia of the renal abdominal aorta over 7 cm in length and 31 mm in diameter at the bifurcation. The right lower limb showed a 13-mm-long femoral artery ectasia and a 2-cm parietal infiltration with superficial femoral artery stenosis greater than 8 cm (Figure 2A).
An angio-CT showing significant stenosis of the iliac arteries in the right lower limb. The left lower limb also had a saccular aneurysm at the origin of the superficial femoral artery measuring 5×5 cm in diameter and containing a 2.4-cm thrombus (Figure 2B).
Repeated CT angiography in June 2017, after the second surgery, showed a new aneurysm of the right axillary artery, and the left axillary artery showed a 4-cm aneurysm (Figure 3). An angio-CT showed saccular aneurisms of the abdominal aorta; one was 22×20 mm on the lateral part of the infra-renal abdominal aorta, with thrombosis (i). An aneurysm after the bifurcation on the right iliac artery measured 30x25 mm (ii) [13,14].
Discussion
Our diagnosis was made on the basis of the criteria of the American College of Rheumatology for the diagnosis of Takayasu arteritis, published in 1990 [8]. This patient presented 5 criteria, with 98% specificity, as shown in Table 4. Several clinical and laboratory characteristics supported the diagnosis of latent tuberculosis and uncontrolled high blood pressure. Thus, our patient developed a Takayasu disease associated with latent tuberculosis. To date, although studies show a high prevalence of TB in patients with TA, there is no evidence of a causal relationship between these 2 diseases [15,16]. Furthermore, the association tuberculosis and systemic disease has been rarely reported in the African literature despite the endemicity of tuberculosis in our region (high prevalence of tuberculosis in Gabon according to WHO) and the underlying systemic diseases [17].
In October 2017, the patient began treatment with oral prednisone at a dose of 1 mg/kg/day (70 mg/day) and started tuberculosis chemotherapy with rifampicin and isoniazid.
To control hypertension, the patient received an increased dose of antihypertensive treatment combination of Perindopril/Indapamide (10 mg/1.25 mg): 1 tablet per day and 50 mg Atenolol per day. We maintained antiplatelet therapy with aspirin at 160 mg daily.
One month after the introduction of antihypertensive therapy, her blood pressure was normalized (systolic blood pressure on the right hand: 103 mmHg, systolic blood pressure on the left hand 120 mmHg) and there was a decrease in the pulsation of the right femoral artery aneurysm.
One month after the start of prednisone and rifampin/isoniazid therapy, an axillary and aortic Doppler ultrasound showed the appearance of a left axillary hematoma surrounding an enlarged axillary artery with an impression of aneurysmal fissure and right axillary aneurysm. Aortic ultrasonography showed 2 saccular aneurysms of the abdominal aorta, one measuring 22×20 mm on the lateral part of the infra-renal aorta containing thrombosis and the second aneurysm on the terminal part of the abdominal aorta at the bifurcation of the right proximal iliac artery measuring 30×25 mm (Figure 4).
Some authors suggest that the relationship between vasculitis and tuberculosis is linked to cross-reactivity against vascular peptides which mimic the antigens of Mycobacterium tuberculosis and thus act as infectious triggers of vasculitis [18]. Other studies have shown that the association of Takayasu arteritis with latent tuberculosis might be related to the existence of giant Langerhans-like cells and granulomas in Takayasu arteritis, similar to those found in tuberculous lesions. However, due to the lack of convincing evidence, further investigation is needed to identify an association between the pathogenesis of Takayasu arteritis and tuberculosis.
Conclusions
Our final diagnosis was Takayasu arteritis in a young, pulseless Gabonese woman who met the criteria for Takayasu arteritis according to the American College of Rheumatology and criteria for latent tuberculosis. This is the first case of Takayashu arteritis ever described in Gabon, with a diagnostic delay of more than 9 months. Because of this long diagnostic delay, the physician must keep in mind that this pathology can lead to death without immunosuppressive therapy. The fact that we are now observing this disease not only in developed countries but also in low-income countries suggests that this vasculitis is becoming ubiquitous.
Figures
References:
1.. Johnston SL, Lock RJ, Gompels MM, Takayasu arteritis: A review: J Clin Pathol, 2002; 55(7); 481-86
2.. Mansour MK, Letourneau AR, Wallace ZS, Case 15-2017 – A 27-year-old woman with anemia, thrombocytosis, and skin lesions after travel abroad: N Engl J Med, 2017; 376(20); 1973-81
3.. Seyahi E, Ugurlu S, Cumali R, Atherosclerosis in Takayasu arteritis: Ann Rheum Dis, 2006; 65(9); 1202-7
4.. Maleszewski JJ, Inflammatory ascending aortic disease: Perspectives from pathology: J Thorac Cardiovasc Surg, 2015; 149(2 Suppl.); S176-83
5.. Vaideeswar P, Deshpande JR, Pathology of Takayasu arteritis: A brief review: Ann Pediatr Cardiol, 2013; 6(1); 52-58
6.. Skeik N, Rodriguez AJ, Engstrom B, Rare case of Takayasu arteritis with concurrent aneurysmal dilation and stenosis: Int J Angiol, 2015; 24(3); 244-48
7.. Sato N, Irie K, Kouzuma R, A case of 37 year long Behcet disease resembling Takayasu arteritis: An autopsy report: Pathol Int, 2016; 66(3); 164-68
8.. Arend WP, Michel BA, Bloch DA, The American College of Rheumatology 1990 criteria for the classification of Takayasu arteritis: Arthritis Rheum, 1990; 33(8); 1129-34
9.. Arnaud L, Haroche J, Limal N, Takayasu arteritis in France: A single-center retrospective study of 82 cases comparing white, North African, and black patients: Medicine (Baltimore), 2010; 89(1); 1-17
10.. Karadag O, Aksu K, Sahin A, Assessment of latent tuberculosis infection in Takayasu arteritis with tuberculin skin test and Quantiferon-TB Gold test: Rheumatol Int, 2010; 30(11); 1483-87
11.. Kechaou M, Frigui M, Ben Hmida M, Bahloul Z, Takayasu arteritis in southern Tunisia: A study of 29 patients: Presse Med, 2009; 38(10); 1410-14
12.. Lim AY, Lee GY, Jang SY, Comparison of clinical characteristics in patients with Takayasu arteritis with and without concomitant tuberculosis: Heart Vessels, 2016; 31(8); 1277-84
13.. Nooshin D, Neda P, Shahdokht S, Ali J, Ten-year investigation of clinical, laboratory and radiologic manifestations and complications in patients with Takayasu’s arteritis in three University Hospitals: Malays J Med Sci, 2013; 20(3); 44-50
14.. Zhu FP, Luo S, Wang ZJ, Takayasu arteritis: Imaging spectrum at multidetector CT angiography: Br J Radiol, 2012; 85(1020); e1282-92
15.. Zhang Y, Fan P, Luo F, Tuberculosis in Takayasu arteritis: A retrospective study in 1105 Chinese patients: J Geriatr Cardiol, 2019; 16(8); 648-55
16.. Pedreira ALS, Santiago MB, Association between Takayasu arteritis and latent or active Mycobacterium tuberculosis infection: A systematic review [published online ahead of print, 2019 Nov 15]: Clin Rheumatol, 2020; 39(4); 1019-26
17.. Ndongo S, Diallo S, Tiendrebeogo J, [Systemic vasculitis: Study of 27 cases in Senegal]: Med Trop (Mars), 2010; 70(3); 264-66 [in French]
18.. van Timmeren MM, Heeringa P, Kallenberg CG, Infectious triggers for vasculitis: Curr Opin Rheumatol, 2014; 26(4); 416-23
Figures
Tables
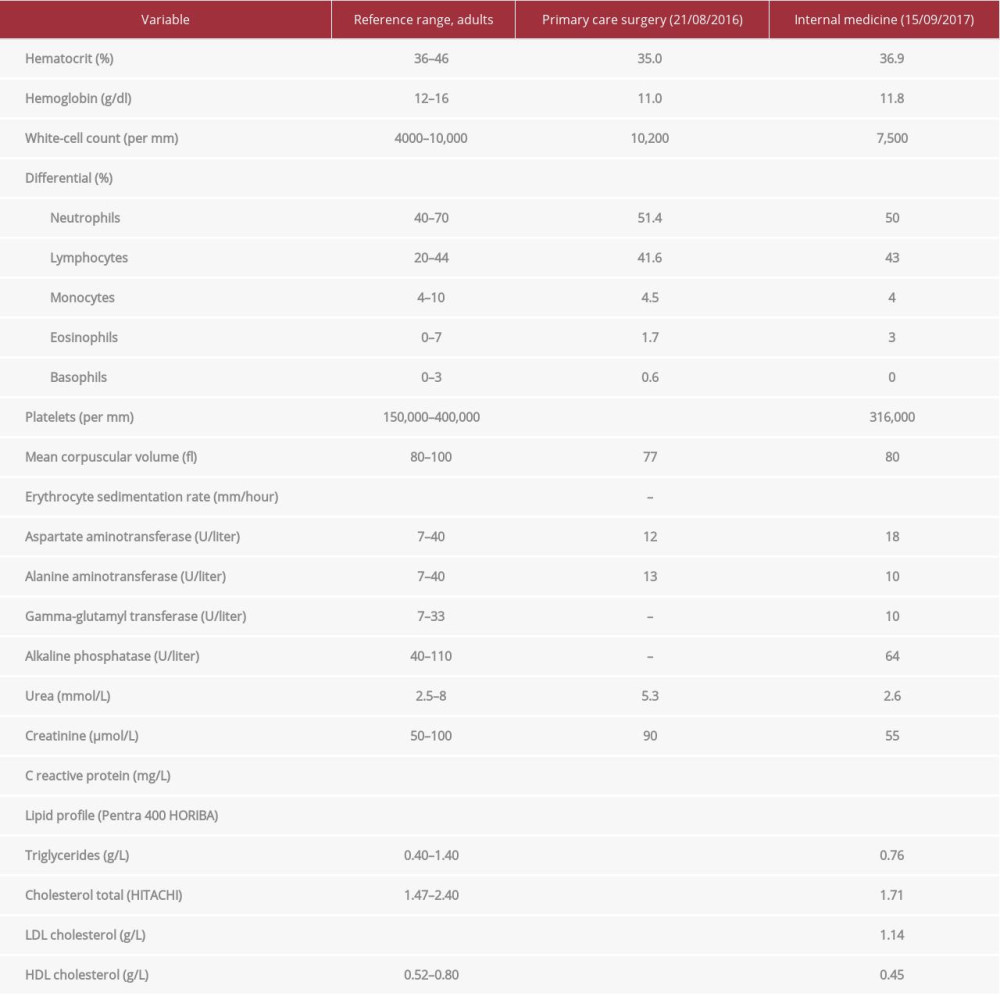 Table 1.. Laboratories tests/laboratory data.
Table 1.. Laboratories tests/laboratory data.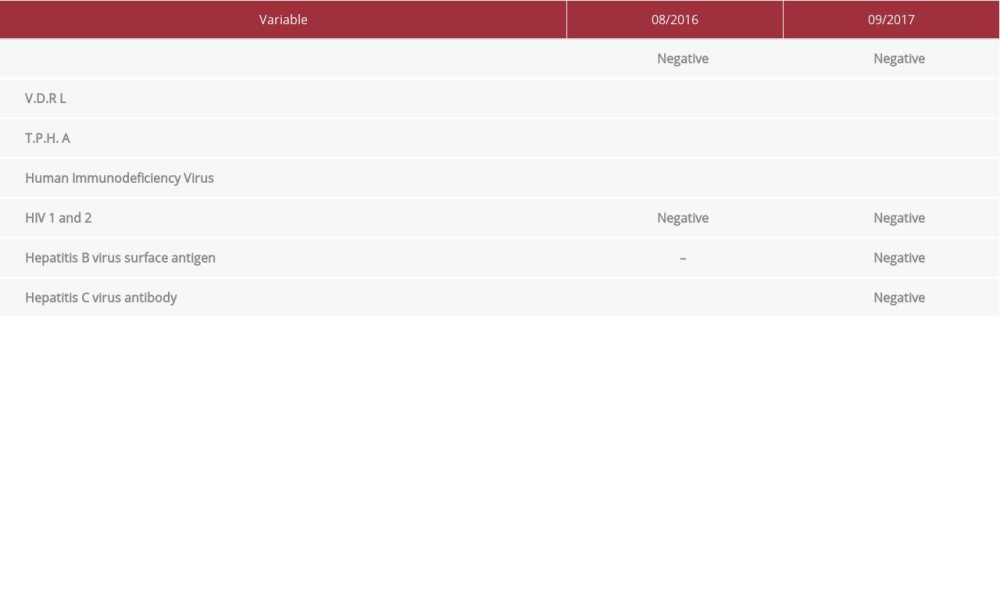 Table 2.. Microbiological data.
Table 2.. Microbiological data.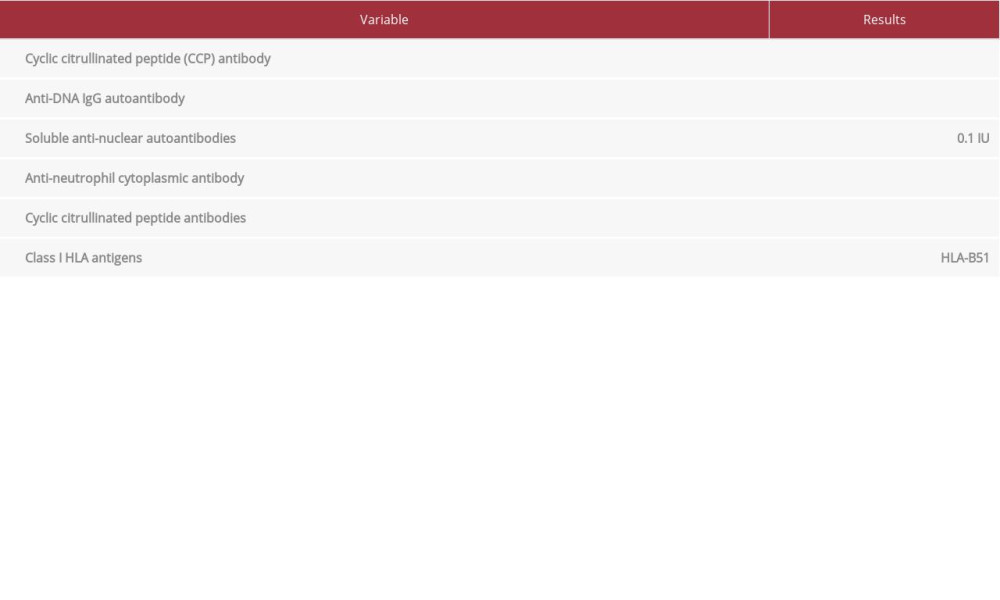 Table 3.. Autoimmune tests and HLA typing.
Table 3.. Autoimmune tests and HLA typing. Table 4.. American College of Rheumatology Criteria for Takayasu arteritis.
Table 4.. American College of Rheumatology Criteria for Takayasu arteritis. Table 1.. Laboratories tests/laboratory data.
Table 1.. Laboratories tests/laboratory data. Table 2.. Microbiological data.
Table 2.. Microbiological data. Table 3.. Autoimmune tests and HLA typing.
Table 3.. Autoimmune tests and HLA typing. Table 4.. American College of Rheumatology Criteria for Takayasu arteritis.
Table 4.. American College of Rheumatology Criteria for Takayasu arteritis. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








