29 September 2020: Articles 
A Case of Postoperative Methicillin-Resistant Enterocolitis in an 81-Year-Old Man and Review of the Literature
Rare disease
Kapil Gururangan12ABCDEF*, Marisa K. Holubar2DEGDOI: 10.12659/AJCR.922521
Am J Case Rep 2020; 21:e922521
Abstract
BACKGROUND: Nosocomial diarrhea affects 12% to 32% of hospitalized patients. Before the development of the Clostridium difficile cytotoxin assay in the 1970s, Staphylococcus aureus was frequently implicated as a cause of hospital-acquired infectious colitis, particularly in association with recent antibiotic therapy or abdominal surgery. Decreased utilization of stool culture has reduced the recognition of S. aureus as a rare, but historically important, cause of enterocolitis.
CASE REPORT: An 81-year-old man with no recent history of travel, exposure to potential infectious sources (e.g., sick contacts, animals, undercooked foods), or antibiotic or proton-pump inhibitor use was admitted for a Whipple procedure (expanded intraoperatively with total pancreatectomy, splenectomy, and portal vein resection) for stage III pancreatic adenocarcinoma. On postoperative day (POD) 5, the patient developed large-volume watery diarrhea that did not improve with tube feeding cessation and oral pancreatic enzyme replacement. He subsequently became clinically septic on POD10, and workup revealed severe radiographic sigmoid and rectal colitis and methicillin-resistant S. aureus (MRSA) bacteremia. Polymerase chain reaction testing for C. difficile was negative twice (POD5 and POD12). He was diagnosed with MRSA proctocolitis and improved with initiation of oral and intravenous vancomycin.
CONCLUSIONS: We describe a case of staphylococcal enterocolitis, a previously common cause of nosocomial diarrhea that has become increasingly underappreciated since the advent of culture-independent stool testing for C. difficile. Increased awareness of this entity, especially when Clostridium assays are negative, may guide more effective treatment of hospital-acquired infection.
Keywords: Clostridium difficile, Cross Infection, diarrhea, Enterocolitis, Methicillin-resistant Staphylococcus aureus, Staphylococcus aureus, Aged, 80 and over, Anti-Bacterial Agents, Pancreatic Neoplasms, Staphylococcal Infections
Background
Before the development of the
Case Report
An 81-year-old man with stage III pancreatic adenocarcinoma without neoadjuvant treatment was admitted for a Whipple procedure, which was expanded intraoperatively with total pancreatectomy, splenectomy, and portal vein resection due to repeatedly positive surgical margins. Portal vein reconstruction was performed with end-to-end anastomosis of remaining portal vein to superior mesenteric vein without vascular graft. He received cefazolin 2 g intravenously prior to surgical incision; additional intraoperative or perioperative antibiotics were not given. He had a medical history of hypertension, hyperlipidemia, gastroesophageal reflux disease, glaucoma, genital herpes, asthma, and a prior renal exophytic mass (fine-needle aspiration showed no evidence of neoplasm) status after cryoablation 2 years prior. It was during annual surveillance imaging for this renal mass that a pancreatic head mass was found. He had no recent diarrhea, travel, antibiotic or proton-pump inhibitor use, or hospitalization, and no history of inflammatory bowel disease. MRSA nares screen (routinely performed on admission at our institution) was positive.
Postoperatively, he was hypotensive and hypovolemic, which was suspected to be secondary to intraoperative fluid losses in addition to further fluid losses from peri-anastomotic Jackson-Pratt drains, and he developed prerenal acute kidney injury that improved with bolus and maintenance intravenous (IV) fluids without requiring vasopressors. No central venous access was required, but the indwelling urinary (Foley) catheter placed intraoperatively was continued postoperatively to closely monitor urine output until it was removed on postoperative day (POD) 5, after which he developed urinary retention that was managed with doxazosin and intermittent straight catheterization. He developed brittle diabetes after total pancreatectomy and required an insulin drip, which was transitioned to scheduled subcutaneous insulin injections on POD4. By POD3, the patient was noted to have waxing and waning encephalopathy consistent with hospitalization-associated delirium, which was managed with quetiapine as needed.
On POD5, the patient developed large-volume, foul-smelling, nonbloody loose stools that were attributed to tube feeding via nasogastric tube and pancreatic insufficiency. He was afebrile with an expected postsplenectomy leukocytosis and without vital sign changes, so he was not started on any empiric antibiotics but was given oral pancreatic enzyme replacement. Blood and urine cultures and
Discussion
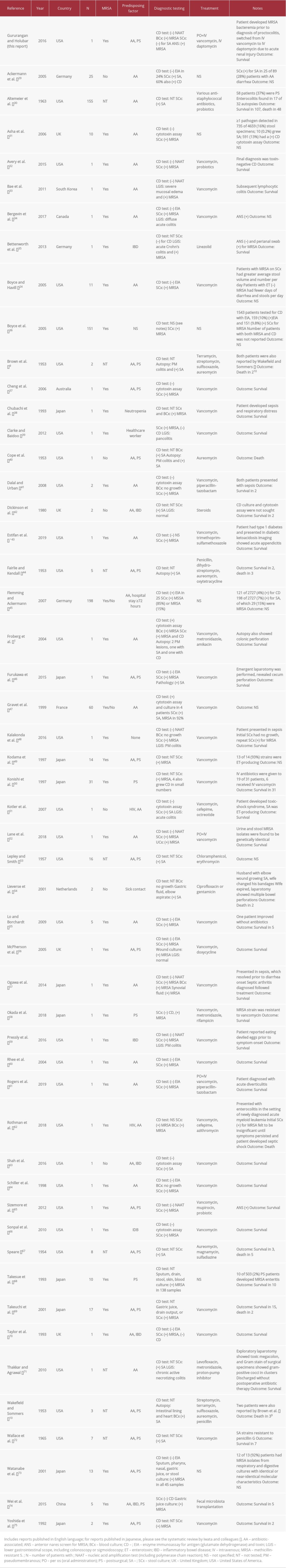
The history of staphylococcal enterocolitis – from its recognition in the 1940s to its eclipse by
Prior cases of
Our patient’s putative diagnosis of MRSA proctocolitis is a controversial one, especially in the absence of definitive endoscopic evidence (due to high risk of anastomotic compromise) or confirmatory stool culture, which is infrequently performed in current clinical practice and is often limited by delayed results and low negative predictive value [11–14]. However, our diagnosis is supported by the following lines of evidence. The likelihood of clinically significant
Conclusions
The management of diarrhea in hospitalized patients is complex and requires both the investigation of multiple noninfectious etiologies, especially in postsurgical and oncologic patients, and the consideration of pathogens not included in routine laboratory testing. Our case highlights the potential for staphylococcal enterocolitis or translocation of colonizing staphylococcal species into the bloodstream to cause severe
References:
1.. Froberg MK, Palavecino E, Dykoski R: Clin Infect Dis, 2004; 39(5); 747-50
2.. Lin Z, Kotler DP, Schlievert PM, Sordillo EM, Staphylococcal enterocolitis: Forgotten but not gone?: Dig Dis Sci, 2010; 55(5); 1200-7
3.. Auchtung TA, Fofanova TY, Stewart CJ, Investigating colonization of the healthy adult gastrointestinal tract by fungi: mSphere, 2018; 3(2); e00092-18
4.. Kane SV, Sandborn WJ, Rufo PA, Fecal lactoferrin is a sensitive and specific marker in identifying intestinal inflammation: Am J Gastroenterol, 2003; 98(6); 1309-14
5.. Holland TL, Arnold C, Fowler VG: JAMA, 2014; 312(13); 1330-41
6.. Singer M, Deutschman CS, Seymour CW, The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3): JAMA, 2016; 315(8); 801-10
7.. Rhodes A, Evans LE, Alhazzani W, Surviving sepsis campaign: International guidelines for management of sepsis and septic shock: 2016: Crit Care Med, 2017; 45(3); 486-52
8.. Iwata K, Doi A, Fukuchi T: BMC Infect Dis, 2014; 14(1); 247-59
9.. Brown WJ, Winston R, Sommers SC, Membranous staphylococcal enteritis after antibiotic therapy; Report of two cases: Am J Dig Dis, 1953; 20(3); 73-75
10.. Wakefield RD, Sommers SC, Fatal membranous staphylococcal enteritis in surgical patients: Ann Surg, 1953; 138(2); 249-52
11.. Bauer TM, Lalvani A, Fehrenbach J: JAMA, 2001; 285(3); 313-19
12.. Miller JM, Binnicker MJ, Campbell S, A guide to utilization of the Microbiology Laboratory for Diagnosis of Infectious Diseases: 2018 Update by the Infectious Diseases Society of America and the American Society for Microbiology: Clin Infect Dis, 2018; 67(6); e1-e94
13.. Imdad A, Retzer F, Thomas LS, Impact of culture-independent diagnostic testing on recovery of enteric bacterial infections: Clin Infect Dis, 2018; 66(12); 1892-98
14.. Axelrad JE, Freedberg DE, Whittier S, Impact of gastrointestinal panel implementation on health care utilization and outcomes: J Clin Microbiol, 2019; 57(3); e01775-18
15.. Polage CR, Solnick JV, Cohen SH: Clin Infect Dis, 2012; 55(7); 982-89
16.. O’Horo JC, Jones A, Sternke M: Mayo Clin Proc, 2012; 87(7); 643-51
17.. Polage CR, Gyorke CE, Kennedy MA: JAMA Intern Med, 2015; 175(11); 1792-801
18.. Murad YM, Perez J, Ybazeta G: BMC Infect Dis, 2016; 16; 430
19.. Wertheim HFL, Melles DC, Vos MC: Lancet Infect Dis, 2005; 5(12); 751-62
20.. Acton DS, Plat-Sinnige MJ, van Wamel W: Eur J Clin Microbiol Infect Dis, 2009; 28(2); 115-27
21.. Claassen-Weitz S, Shittu AO, Ngwarai MR: Front Microbiol, 2016; 7; 449
22.. Gorwitz RJ, Kruszon-Moran D, McAllister SK: J Infect Dis, 2008; 197(9); 1226-34
23.. Salgado CD, Farr BM, Calfee DP: Clin Infect Dis, 2003; 36(2); 131-39
24.. Boyce JM, Havill NL: Am J Gastroenterol, 2005; 100(8); 1828-34
25.. Matsubara Y, Uchiyama H, Higashi T, Nasal MRSA screening for surgical patients: Predictive value for postoperative infections caused by MRSA: Surg Today, 2014; 44(6); 1018-25
26.. Libby DB, Bearman G: Int J Infect Dis, 2009; 13(5); e305-9
27.. Naber CK: Clin Infect Dis, 2009; 48(Suppl. 4); S231-37
28.. Lima NL, Guerrant RL, Kaiser DL, A retrospective cohort study of nosocomial diarrhea as a risk factor for nosocomial infection: J Infect Dis, 1990; 161(5); 948-52
29.. Ackermann G, Thomalla S, Ackermann F, Prevalence and characteristics of bacteria and host factors in an outbreak situation of antibiotic-associated diarrhoea: J Med Microbiol, 2005; 54(2); 149-53
30.. Altemeier WA, Hummel RP, Hill EO, Staphylococcal enterocolitis following antibiotic therapy: Ann Surg, 1963; 157(6); 847-57
31.. Asha NJ, Tompkins D, Wilcox MH: J Clin Microbiol, 2006; 44(8); 2785-91
32.. Avery LM, Zempel M, Weiss E: Am J Health Syst Pharm, 2015; 72(11); 943-51
33.. Bae JH, Han DS, Park HS, A case of methicillin-resistant staphylococcal enterocolitis with subsequent development of lymphocytic colitis: Intest Res, 2011; 9(2); 139-43
34.. Bergevin M, Marion A, Farber D, Severe MRSA enterocolitis caused by a strain harboring enterotoxins D, G, and I: Emerg Infect Dis, 2017; 23(5); 865-67
35.. Bettenworth D, Nowacki TM, Friedrich A: World J Gastroenterol, 2013; 19(27); 4418-21
36.. Boyce JM, Havill NL, Maria B: J Clin Microbiol, 2005; 43(12); 5992-95
37.. Cheng AC, Wade A, Harris OC: ANZ J Surg, 2006; 76(8); 763
38.. Chubachi A, Hashimoto K, Miura AB: Am J Hematol, 1993; 43(4); 327
39.. Clarke K, Baidoo L: Int J Colorectal Dis, 2012; 27(3); 417-18
40.. Cope O, Hanelin J, Shaw RS, Staphylococcal membranous enteritis: N Engl J Med, 1953; 249(1); 29-33
41.. Dalal A, Urban C: Infect Dis Clin Pract, 2008; 16(4); 222-23
42.. Dickinson RJ, Dixon MF, Axon AT, Staphylococcal enterocolitis and inflammatory bowel disease: J Clin Pathol, 1980; 33(6); 604-5
43.. Estifan E, Nanvati S, Kumar V, Unusual presentation of MRSA colitis complicated with acute appendicitis: Am J Gastroenterol, 2019; 114; S1647-48
44.. Fairlie CW, Kendall RE, Fatal staphylococcal enteritis following penicillin and streptomycin therapy: JAMA, 1953; 153(2); 90-94
45.. Flemming K, Ackermann G: Infection, 2007; 35(5); 356-58
46.. Furukawa K, Tsutsumi J, Takayama S: Int Surg, 2015; 100(3); 414-16
47.. Gravet A, Rondeau M, Harf-Monteil C: J Clin Microbiol, 1999; 37(12); 4012-19
48.. Kalakonda A, Garg S, Tandon S, A rare case of infectious colitis: Gastroenterol Rep (Oxf), 2016; 4(4); 328-30
49.. Kodama T, Santo T, Yokoyama T: Surg Today, 1997; 27(9); 816-25
50.. Konishi T, Idezuki Y, Kobayashi H: Surg Today, 1997; 27(9); 826-32
51.. Kotler DP, Sandkovsky U, Schlievert PM, Sordillo EM, Toxic shock-like syndrome associated with staphylococcal enterocolitis in an HIV-infected man: Clin Infect Dis, 2007; 44(12); e121-23
52.. Lane AB, Copeland NK, Onmus-Leone F, Lawler JV: Case Rep Infect Dis, 2018; 2018; 3106305
53.. Lepley D, Smith MB, Staphylococcal enterocolitis: AMA Arch Surg, 1957; 75(3); 377-87
54.. Lieverse RJ, van Hooft J, Fedder G, Necrotizing enterocolitis in a married couple due to a staphylococcal toxin: Eur J Gastroenterol Hepatol, 2001; 13(5); 595-97
55.. Lo TS, Borchardt SM: Diagn Microbiol Infect Dis, 2009; 63(4); 388-89
56.. McPherson S, Ellis R, Fawzi H, Panter SJ: Eur J Gastroenterol Hepatol, 2005; 17(11); 1225-27
57.. Ogawa Y, Saraya T, Koide T: BMC Res Notes, 2014; 7(1); 21-27
58.. Okada N, Fujita T, Kanamori J: Int J Surg Case Rep, 2018; 52; 75-78
59.. Pressly KB, Hill E, Shah KJ: BMJ Case Rep, 2016; 2016; bcr2016215225
60.. Rhee KY, Soave R, Maltz C: J Clin Gastroenterol, 2004; 38(3); 299-300
61.. Rogers E, Dooley A, Vu S: Cureus, 2019; 11(6); e5013
62.. Rothman A, Lio J, Lee Y, MRSA colitis: An under-recognized cause of septic shock: Am J Respir Crit Care Med, 2018; 197; A5187
63.. Shah D, Fisher C, Wilson S, Gutkin E: Am J Gastroenterol, 2016; 111; S5835
64.. Schiller B, Chiorazzi N, Farber BF, Methicillin-resistant staphylococcal enterocolitis: Am J Med, 1998; 105(2); 164-66
65.. Sizemore EN, Rivas KM, Valdes J, Caballero J: BMJ Case Rep, 2012; 2012; bcr2012006366
66.. Sonpal N, Mathur S, Arya M: Am J Gastroenterol, 2010; 105; S359
67.. Speare GS, Staphylococcus pseudomembranous enterocolitis, a complication of antibiotic therapy: Am J Surg, 1954; 88(4); 523-34
68.. Takesue Y, Yokoyama T, Kodama T: Surg Today, 1993; 23(1); 4-8
69.. Takeuchi K, Tsuzuki Y, Ando T: Eur J Surg, 2001; 167(4); 293-96
70.. Taylor M, Ajayi F, Almond M: Lancet, 1993; 342(8874); 804
71.. Thakkar S, Agrawal R: Gastroenterol Hepatol, 2010; 6(2); 115-17
72.. Wallace JF, Smith RH, Petersdorf RG, Oral administration of vancomycin in the treatment of staphylococcal enterocolitis: N Engl J Med, 1965; 272(19); 1014-15
73.. Watanabe H, Masaki H, Asoh N: Microbiol Immunol, 2001; 45(9); 629-34
74.. Wei Y, Gong J, Zhu W: BMC Infect Dis, 2015; 15; 265
75.. Yoshida D, Fukunari H, Hojo I: Bull Tokyo Med Dent Univ, 1992; 39(2); 31-34
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133