28 August 2020: Articles 
Gastrointestinal Bleeding in a Pancreas Transplant Recipient: A Case to Remember
Unusual clinical course, Challenging differential diagnosis
Jeevan Prakash Gopal1EFG, James E. Jackson2E, Andrew Palmer1E, David Taube1E, Anand Sivaprakash Rathnasamy Muthusamy13E*DOI: 10.12659/AJCR.923197
Am J Case Rep 2020; 21:e923197
Abstract
BACKGROUND: Pancreas transplantation has proven to be the most effective therapeutic option for insulin-dependent diabetes mellitus. However, despite advances in surgical technique and continuously improving outcomes, pancreas transplantation has the highest complication rate among all solid-organ transplants. Vascular complications in particular can be catastrophic, with graft- and life-threatening potential. Ectopic variceal bleeding is less common and is rarely reported in the literature.
CASE REPORT: A 51-year-old man presented with recurrent intermittent gastrointestinal bleeding (GIB) associated with hepatic dysfunction and portal hypertension 4 years after a successful pancreas-after-kidney transplant. Apart from positive serology for hepatitis E virus, all the other liver disease screening results were negative. He was extensively investigated with 6 computed tomography (CT) scans, 3 esophago-gastro-duodenoscopies (EGD), 3 colonoscopies, and 1 visceral arteriogram before the plausible diagnosis of ectopic trans-anastomotic variceal bleeding involving the pancreas transplant was established. Selective variceal catheterization and embolization were done with 3% sodium tetradecyl sulphate (STD). He remained free of bleeding after embolization.
CONCLUSIONS: This case report adds to the scanty literature on the management of ectopic variceal bleeding in a pancreas transplant recipient. Diagnosis of ectopic varix is usually challenging and frequently requires a visceral arteriogram. We describe a novel minimally-interventional technique to obtain source control and also discuss the complexity involved in the management, along with future implications.
Keywords: Embolization, Therapeutic, Gastrointestinal Hemorrhage, Pancreas Transplantation, Esophageal and Gastric Varices, Kidney Transplantation, Pancreas
Background
A successful pancreas transplantation enables durable glycemic control with survival benefits for diabetic patients [1]. The success of pancreas transplantation is predominantly influenced by the associated surgical complications. Due to the continuing advancement in surgical techniques, technical failure rates have decreased to around 8% [2]. Vascular complications associated with pancreas transplantation are not infrequent, the most common of which is allograft thrombosis, which usually occurs within the first 6 weeks. Bleeding due to arterial complications (arterio-venous fistula, pseudoaneurysm, arterio-enteric, or arterio-cystic fistula) have been reported but are relatively infrequent. Ectopic variceal bleeding in pancreas transplant recipients is even rarer, with only 2 cases reported in the literature. Diagnosis is usually challenging and frequently needs exhaustive work-up. Therapeutic options are endovascular intervention and transplant pancreatectomy with or without vascular reconstruction.
Case Report
A 51-year-old white man with long-standing, poorly controlled type-1 diabetes mellitus, complicated by peripheral vascular and coronary artery disease (CAD), underwent pancreas-after-kidney (PAK) transplantation from a 19-year-old donation after brainstem death (DBD) donor. He was on long-term amoxicillinclavulanic acid for chronic foot ulcers. The pancreas allograft was implanted extraperitoneally in the left iliac fossa with enteric exocrine and systemic venous drainage into the recipient left external iliac vein. Immunosuppression consisted of induction with methylprednisolone and maintenance with tacrolimus, mycophenolate mofetil, and a short course of steroids. The initial postoperative period was uncomplicated, with primary function of the pancreas.
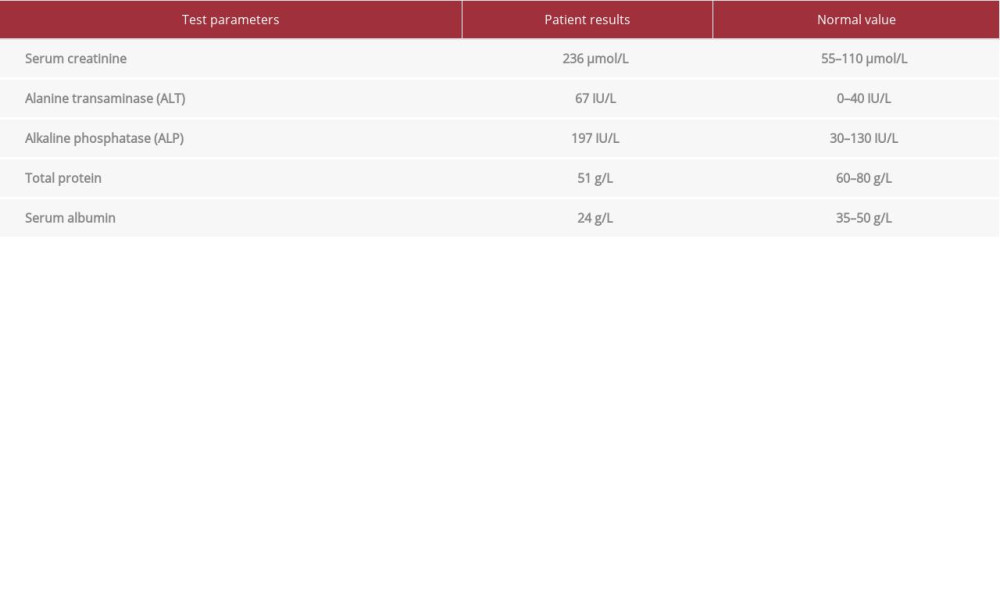
Four years after PAK transplantation, he was admitted due to ascites with kidney graft dysfunction, an infected foot ulcer, and abnormal liver function tests (LFTs) (Table 1). Contrast-enhanced computed tomography of the abdomen and pelvis (CTAP) showed moderate ascites with pericholecystic and periportal edema, morphologically normal liver and spleen, no varices, patent porto-spleno-mesenteric axis, and well-perfused transplanted organs. Polymerase chain reaction (PCR) analyses for cytomegalovirus (CMV), Epstein-Barr virus (EBV), and hepatitis viral markers were negative. Markers for autoimmune liver disease, serum iron, and copper studies were all within normal limits. The foot infection responded well to broad-spectrum antibiotics, and kidney function recovered completely. During the hospital stay, he had significant melena and subsequent episodes of hematochezia associated with encephalopathy, necessitating multiple transfusions of packed red blood cells (PRBC). Prominent gastric antral erosions and gastritis consistent with gastric antral vascular ectasia (GAVE) without any stigmata for recent or remote bleeding were found on EGD. Colonoscopy failed to reveal any bleeding source. At this stage, he developed positive serology (Immunoglobulin M) for hepatitis E virus (type 3 genotype, viral load 2 million IU/mL). Ribavirin 200 mg/day was started, along with reduction in tacrolimus dose and temporary cessation of mycophenolate mofetil (MMF). CT and MRI (magnetic resonance imaging) of the brain were unrevealing and his neurology status improved spontaneously. A repeat contrast-enhanced CTAP showed large-volume ascites without any evidence of liver disease or portal hypertension, globally perfused transplanted organs, and dilatation and hyperenhancement of the transplant duodenum. The ascites resolved with diuretics and albumin supplementation. He was stabilized and discharged.
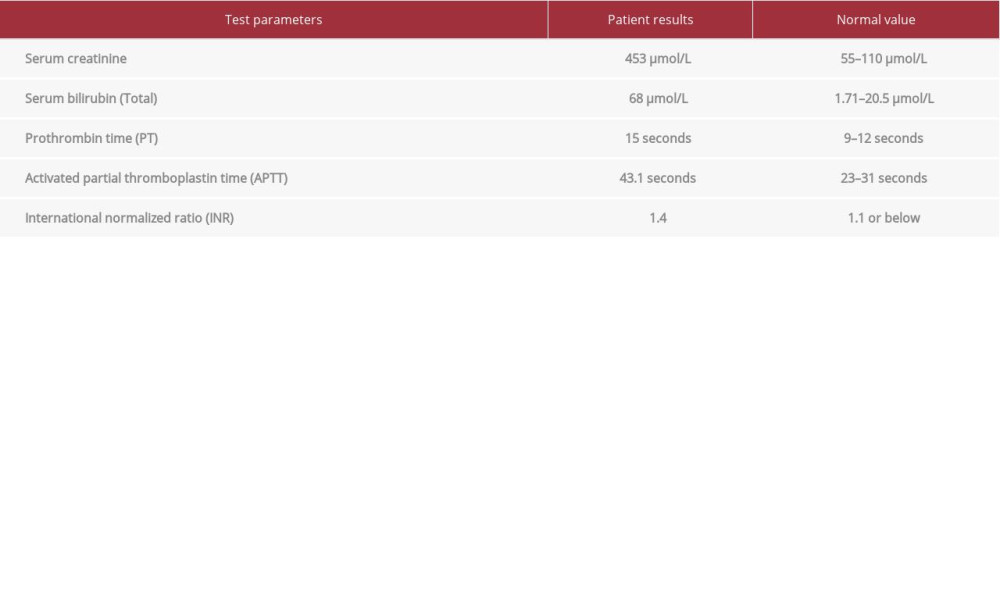
He was subsequently re-admitted with ascites re-accumulation, worsening kidney function, and deranged LFTs (Table 2). Repeat serology for hepatitis B/C virus and chronic liver disease screening were negative. During this admission, he had recurrent episodes of lower-GI bleeding associated with hemoglobin drop and hemodynamic instability. He had 3 further CT abdominal angiograms, all of which showed persisting contrast enhancement around the graft pancreas but no evidence of local intraluminal hemorrhage. He had 2 further upper-GI endoscopies, with no evidence of active upper-gastrointestinal source of bleeding apart from moderate antral-predominant gastritis (although a biopsy was normal). LFTs subsequently normalized except for serum albumin (31 g/L) and the ascites resolved spontaneously. He also underwent 2 colonoscopies and both were unrevealing. He received a total of 13 units of PRBC, 2 units of fresh frozen plasma, 2 pools of platelets, and 1 unit of cryoprecipitate. The patient then developed an acute coronary syndrome. As he continued to have lower-GI bleeding, a further CTAP was performed, in which the areas of small-bowel varices related to the anastomosis between the transplant duodenum and the recipient jejunum (Figure 1A, 1B) were thought likely to be the source of recurrent hemorrhage. Endovascular obliteration of these varices was recommended. Retrospective review of previous imaging demonstrated subtle but present features of ectopic varices around the transplant duodenum, previously described as “hyperenhancement of the pancreas”.
Portal venography was performed via a percutaneous transhepatic approach, which demonstrated retrograde flow within a dilated jejunal vein (Figure 2A) communicating with the graft duodenal vein via multiple trans-anastomotic varices and draining through the graft portal vein into the recipient left external iliac vein (Figure 2B). These varices were selectively catheterized and were embolized with a total of 3 mL of 3% sodium tetradecyl sulphate (STD). A final portography confirmed complete occlusion of the varices with obliteration of the porto-systemic shunt and preserved antegrade flow in the superior mesenteric and main portal vein (Figure 3). Ascites increased after the procedure and 3.125 mg carvedilol was commenced. A trans-jugular liver biopsy and hepatic portal venous gradient measurement were deferred due to clinical instability of the patient. The patient remained well, with no episodes of GI bleeding post-embolization; however, he then had a second acute coronary event leading to cardiac arrest with consequent irreversible hypoxic brain injury and he ultimately died.
Discussion
Arterial complications after pancreas transplantation can cause massive life-threatening GIB. Ectopic variceal bleeding due to porto-systemic trans-anastomotic varices between the transplant duodenum and recipient small bowel is a rare cause of GIB, with only 2 cases reported in the literature [3,4]. Fontana et al. [3] described peri-transplant duodenal varices in a patient after kidney–pancreas transplant in the setting of portal hypertension, who was treated successfully with coil embolization followed by liver transplantation. Rostambegi et al. [4] reported a similar patient in whom variceal embolization was performed but who subsequently required surgical resection of the involved segment of bowel due to recurrent bleeding.
The authors of the report [4] did not measure portal vein pressure but concluded that portal hypertension was not present despite the presence of the trans-anastomotic varices.
Porto-systemic varices develop in portal hypertension (PHT) and are most commonly gastro-oesophageal. Ectopic varices are much less common but are known to develop across bowel anastomoses that allow porto-systemic communication; a typical example is those that occur around a stoma where portal venous branches develop communications with abdominal wall veins. Such varices, which are neo-vessels rather than enlargement of pre-existing porto-systemic communications as is the case with gastro-esophageal varices, are very friable and are particularly prone to hemorrhage, which can be massive. The incidence of ectopic variceal bleed is 1% to 5% in cirrhosis, 27% to 40% in extrahepatic portal hypertension, and up to 64% in coexisting portal venous occlusion and hepatobiliary surgery [5].
The rarity of peri-transplant ectopic varices could have contributed to the delay in radiological diagnosis given the variability of the location of the transplant-to-recipient bowel anastomosis, and typical appearances are not obvious on CT angiography. Ectopic varices can be very difficult to manage. Both endoscopic and interventional radiological techniques may be helpful, although surgery may be necessary in some cases. Endoscopic access to ectopic varices depends on the location of the porto-systemic anastomosis, and in our patient’s situation would have been difficult to access (apart from an entero-scope or a capsule endoscope) for diagnosis. Emergency surgery in the form of transplant pancreatectomy is possible, although the morbidity rate is high. In the patient reported here, surgical options were kept as a last resort, given his co-morbidities and the desire not to explant a functioning pancreas allograft.
The underlying etiology of portal hypertension in this case was not established, although there were multiple putative risk factors. Amoxycillin-clavulanic acid-induced liver injury is a possible cause, as this agent is known to cause varying degrees of hepatic dysfunction, with cases of acute liver failure and chronic liver disease being described [6,7]. Other etiologies that should be considered are idiopathic non-cirrhotic portal hyper-tension or chronic hepatitis E virus (HEV) infection. Hepatitis E infection is becoming more common and infection with type-3 genotype can progress to chronic infection, leading to cirrhosis and liver failure in solid-organ transplant recipients [8].
Bleeding from ectopic varices around the pancreas transplant remain the most likely cause, as there were no typical varices in the oesophagus or rectum, and there was complete resolution of symptoms following radiological embolization of the varices. Unfortunately, due to the subsequent cardiac events that led to the death of the patient, establishing a pathological diagnosis was not possible.
Conclusions
This case report adds to the scarce literature on the management of ectopic variceal bleeding in pancreas transplant recipients, describing a novel minimally-interventional technique to obtain source control. Diagnosis can be very difficult, particularly as other signs of portal hypertension may be lacking, as was the case in the patient reported here. Management should be tailored to the patient’s clinical state, the severity of hemorrhage, and availability of appropriate expertise.
Figures
References:
1.. Dean PG, Kukla A, Stegall MD, Pancreas transplantation: BMJ, 2017; 357; j1321
2.. Troppmann C, Complications after pancreas transplantation: Curr Opin Organ Transplant, 2010; 15(1); 112-18
3.. Fontana I, Bertocchi M, Di Domenico S, Percutaneous embolization of periduodenal varix due to portal hypertension in a patient with kidney–pancreas transplantation: A case report: Transplant Proc, 2010; 42(6); 2162-63
4.. Rostambeigi N, Shrestha P, Dunn TB, Recurrent ectopic variceal bleed after pancreas transplantation with no portal hypertension: Case report and outcomes of endovascular onyx embolization: Vasc Endovascular Surg, 2019; 53(5); 415-19
5.. Helmy A, Al Kahtani K, Al Fadda M, Updates in the pathogenesis, diagnosis and management of ectopic varices: Hepatol Int, 2008; 2(3); 322-34
6.. Fontana R, Shakil AO, Greenson JK, Acute liver failure due to amoxicillin and amoxicillin/clavulanate: Dig Dis Sci, 2005; 50(10); 1785-90
7.. Ryley NG, Fleming KA, Chapman RW, Focal destructive cholangiopathy associated with amoxycillin/clavulanic acid (Augmentin): J Hepatol, 1995; 23(3); 278-82
8.. Donnelly MC, Scobie L, Crossan CL, Review article: Hepatitis E-a concise review of virology, epidemiology, clinical presentation and therapy: Aliment Pharmacol Ther, 2017; 46(2); 126-41
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133