27 August 2020: Articles 
Acute Myocardial Infarction in a Young Bodybuilder: A Case Report and Review of the Literature
Patient complains / malpractice, Adverse events of drug therapy
Abrão José Melhem Jr.1ABDEF*, Amélia Cristina Araújo12AB, Felipe Nathan S. Figueiredo13BDE, David Livingstone A. Figueiredo14ADEDOI: 10.12659/AJCR.924796
Am J Case Rep 2020; 21:e924796
Abstract
BACKGROUND: Misuse of androgenic anabolic steroids (AAS) is a current practice associated with vigorous bodybuilding for muscular hypertrophy, especially among gym practitioners and bodybuilders, influenced by the culture of body image. In addition to liver, psychiatric, genital, urinary, dermatological, and musculoskeletal complications, AAS misuse reportedly can lead to development of cardiovascular complications, such as hypertension, dyslipidemia, cardiac hypertrophy, and early coronary disease, and potentially acute myocardial infarction (AMI) and sudden death.
CASE REPORT: A 26-year-old male farmer who was also an amateur bodybuilder developed an extensive Killip Class I AMI in the anterior wall while using AAS. A few days before the acute event, his lipid and hormone levels were measured and found to be significantly elevated. The patient was asymptomatic after left anterior descending branch angioplasty, but he had significant electrocardiographic sequelae and ventricular dysfunction.
CONCLUSIONS: We describe the case of a young male bodybuilder using AAS who presented with AMI and was treated with primary angioplasty. Documentation of high levels of lipids and hormones 1 week before the acute event suggests some relationship between AAS and cardiovascular disease. The main effects of using these steroids on the cardiovascular system are reviewed. It is time for a new global warning about the risks of misusing AAS to obtain muscle hypertrophy. Based on current medical knowledge, these hormones should not be prescribed without a clear indication for their use.
Keywords: Anabolic Agents, Androgens, Somatotypes, young adult, Doping in Sports, Heart, Steroids
Background
Use of androgenic anabolic steroids (AAS) is widespread among amateur athletes, and especially bodybuilders, to increase muscular mass with the aim of improving their performance, but the serious health effects are often overlooked [1,2]. Many individuals try anabolic androgens because of dissatisfaction with their own body image. Factors that may contribute to the dissatisfaction include not measuring up to the “ideal standards” perpetuated in mass media, fear of being diminished or embarrassed by peers, the symbolic capital of being a musclebound person, and the perception of being able to gain muscle mass easily with the drugs [3]. Factors that compromise healthy construction of masculinity also have been mentioned as a cause for the search for a quick way to develop muscles [4]. In young adults, concomitant use of AAS has been associated with early atherosclerosis and acute myocardial infarction (AMI), among other health problems [1,5,6]. When a young patient is diagnosed with chronic or acute coronary disease, one of the principal suspects should be the misuse of AAS [7]. This paper presents a case and review of the literature.
Case Report
A 26-year-old male farmer and amateur bodybuilder was admitted to the hospital with severe pain in his left hemithorax, which had begun about 2 hours earlier. The patient had been treated for a peptic gastric disease 8 years ago and had no other history of illness. Two weeks prior, he had mild epigastric pain, which subsided after he took a proton pump inhibitor. In the 24 hours before, he had mild retrosternal and transitory pain, which worsened progressively. The pain became intermittent, of high intensity, with no radiation, and it was not related to exertion and was not relieved by a common analgesic. The patient was experiencing a period of significant professional stress, anxiety, and irritability. He had no symptoms during weight training sessions. The patient did not smoke or drink alcohol, and he denied using cocaine or stimulants and reported his cholesterol and glucose levels had been tested 1 year ago and were normal. For 6 months before developing acute coronary syndrome, he had been self-injecting t trenbo-lone acetate, stanozolol, and testosterone. The patient’s family history included 2 cases of AMI. His father had an AMI at age 50 years and a paternal aunt had 3 AMIs after age 53 years; both had hypercholesterolemia.
Initial evaluation of the patient revealed the following: blood pressure (BP) of 122/78 mmHg, heart rate (HR) 92 beats per minute (BPM), respiration 12 inhalations per minute (IPM), weight 76 kg, height 176 cm, and body mass index (BMI) 24.5 kg/m2. Physical examination was normal. Hirsutism was reported by the patient but it was not visible because he shaved his body hair. There were no changes in his testicular volume.
A week before, testing of the patient’s hormonal and biochemical levels had been ordered by another clinician as part of routine care. Table 1 shows the results: low levels of luteinizing and follicle-stimulating hormones. The testing also revealed dyslipidemia, with high levels of low-density lipoproteins (LDL cholesterol), low levels of high-density lipoproteins (HDL cholesterol), and elevated C-reactive protein (CRP). Levels of estrone and estradiol and dehydroepiandrosterone (DHEA) also were elevated.
An electrocardiogram (EKG) showed ST segment elevation in leads V2 to V6 and in leads DI and aVL, with pathological Q waves in leads V4, V5, DI, and aVL (Figure 1). Markers of myocardial necrosis were elevated, as described in Table 2. Cinecoronariography and angioplasty were performed immediately, and an occlusion of the left anterior descending branch of the left coronary artery was promptly treated with angioplasty and implantation of a drug-eluting stent (Figure 2).
Doppler echocardiography (DECG) performed before the patient’s discharge revealed an increase in the size of the left atrium, reduced relaxation in the left ventricle, septal and midapical systolic dysfunction, and increased systolic dimension, with ejection fraction of 47% (Figure 3, Table 3).
The patient was discharged after 3 days in the hospital, with no symptoms, normal vital signs, a diagnosis of Killip Class I AMI, and prescriptions for ramipril (5 mg/day), carvedilol (6.25 mg twice a day), clopidogrel (75 mg/day), acetylsalicylic acid (100 mg/day), and rosuvastatin (10 mg/day).
During an outpatient assessment 4 weeks later, the patient reported having mild palpitations and insomnia. Results of a physical examination were normal (BP 118/68 mmHg, HR 76 bpm, RF 12 IPM, weight 73.6 kg, height 176 cm, BMI 23.8 kg/m2). His EKG showed infarction in the extensive anterior wall marked by pathological Q waves on V1 to V5 derivations and persistence of the ST elevation from V2 to V6, DI and aVL derivations (Figure 4). A new DECG showed improvement in the function and sizes of the left chambers, with no diastolic dysfunction or left atrial enlargement, with hypokinesis in the septal wall as the only finding (Table 4).
The patient’s dosages of carvedilol and rosuvastatin were increased and ezetimibe was added because his HR was not yet at 60 bpm and the LDL target had not been reached. Troponin was no longer elevated and the level of B-type natriuretic peptide was normal. A drastic drop in the patient’s testosterone level and slight elevations in his liver enzymes also were observed (Table 5).
Four months after his AMI, the patient was asymptomatic, working at a farm, and doing regular moderate physical activity without using steroids or any drugs other than those that he had been prescribed. His physical examination was normal, but his EKG showed an extensive inactive area in the anterior wall. Treadmill stress testing showed no symptoms, ST deviation, or arrhythmia, with excellent functional capacity (14.6 metabolic equivalent).
Discussion
REPLACEMENT THERAPY VERSUS AAS MISUSE:
Use of AAS for replacement in middle-aged or older, symptomatic men with low serum testosterone levels is a well-established practice, provided they are carefully evaluated and monitored. The dosage for testosterone replacement is approximately 400 mg every 2 or 4 weeks (maximum 800 mg/month) and the goal is to achieve a level at the low end of the normal range and recovery from hypogonadism symptoms (241 ng/dL) [6]. Even this replacement use has been linked with increased cardiovascular risks. Corona et al. [6] observed that low doses can be associated with an increased risk of MI and mortality, which underscores the significant role of dose monitoring of testosterone replacement. However, other authors have shown evidence that low testosterone levels in men with congestive heart failure are linked to poor prognosis and increased mortality [11,12]. There are reports of reduced cardiovascular and reduced mortality with testosterone replacement therapy versus untreated men. Replacement therapy has been linked to less myocardial ischemia, better exercise capacity, and better glucose metabolism [12]. The patient in this report used much higher doses of AAS, starting with 600 mg per week (2400 mg/month) [13] and had a testosterone level that was more than twice normal (Table 1).
EPIDEMIOLOGICAL FINDINGS ON AAS MISUSE:
The global prevalence of AAS use has been estimated at 3.3% to 6.4% in men [2, 14] and 1.3% in women [14,15]. Most men who use AAS to gain muscular mass gain are not athletes and their principal goal is to look and feel strong [2]. Although women have less global use of anabolic steroids, a study of North American high school students showed a 5.3% use rate, similar to the incidence of depressive symptoms, use of cigarettes, and slimming pills. Authors also observed that the use was not necessarily associated with participation in sports competitions, but appears as part of a series of dangerous attitudes aiming to achieve ephemeral feeling of well-being [15]. Our patient, a 26-year-old man who was an amateur athlete interested in bodybuilding, not in sports results, belongs to the group most likely to misuse AAS. The drugs he used – trenbolone, stanozolol, and testosterone – are the ones most frequently described in other studies in this area [16,17].
MAIN ADVERSE EFFECTS OF AAS MISUSE:
The effects of AAS have been studied for decades. Studies have described endocrine, dermatological, skeletal, genitourinary, psychiatric, hepatic, metabolic, and cardiovascular effects in bodybuilders and high-level athletes [5,6,8,18–21] (Table 6). According to the literature, psychiatric symptoms are common in users of the hormones, and our patient reported stress, anxiety, and irritability before his AMI. Although the psychiatric symptoms associated with AAS are nonspecific and underestimated, they could be a clue that an individual should consider stopping use of these hormones [22,23].
METABOLIC EFFECTS OF AAS MISUSE:
Changes in lipid profile were observed in this patient a few days before his AMI. The high LDL and low HDL cholesterol levels (204 mg/dL and 30 mg/dL, respectively) suggest hormonal action on lipid metabolism, which increases cardiovascular risk [5,24–27]. High levels of CRP (3.54 mg/L) have been associated with AAS misuse. The mechanism of action may by the hepatic activity of these hormones or the role that CRP plays as a pro-inflammatory marker [28].
EFFECTS OF AAS MISUSE IN CARDIOVASCULAR DISEASE:
Although it is not possible to conclude that AAS caused coronary plaques in this specific case, the association has been described in the literature. Baggish et al. [28] evaluated 140 weightlifters ages 34 to 54 years with DECG and coronary tomography, with 2-year follow-up, and observed a higher incidence of left ventricular systolic dysfunction and diastolic dysfunction and greater volume of coronary plaques in AAS users than in non-users.
There are not many AAS-related AMI case reports [7,30–39]. Generally, these cases occur in men ages 25 to 35 years who are bodybuilders or fighters and have few or no traditional risk factors. In some cases, no obstructions in the coronary arteries have been found. It is possible that these patients have some characteristics more suggestive of myocarditis, which is also associated with AAS, especially in cases of diffusely myocardial dysfunction, elevated troponin, and no coronary obstruction [40]. Another explanation is the development of small-vessel disease related to misuse of AAS [41].
The main pathophysiological events associated with acute coronary syndrome in AAS users are accelerated atherosclerosis, modification in lipid profile (increased LDL and reduced HDL cholesterol), coronary vasospasm (caused by inhibition of guanylate cyclase), and coronary thrombosis due to increased platelet aggregation (increased thromboxane A2 and reduced prostacyclin) [30]. We reported a case of a young man with a familial history of coronary disease and dyslipidemia. The patient was an AAS user and presented with changes in his lipid profile and many aspects of the sexual hormonal axis 1 week before having an AMI (Table 1). We believe that he was exposed to acute cardiovascular risk when the atherosclerotic process was accelerated by a pro-coagulant, pro-inflammatory, and pro-vasospastic state.
AAS MISUSE AND MORTALITY:
Misuse of AAS impacts mortality. In 2018, Helal et al. reported the risk of death was 6 to 20 times higher in athletes who used AAS than in other athletes [42]. Frati et al. (n=19) [41] and Montisci et al. (n=4) [43], studying autopsies of sudden death related to AAS misuse, identified a wide range of macro-and microscopic changes. They included concentric hypertrophy, hypertrophic cardiomyopathy, myocarditis, focal fibrosis, peripheral venous thrombosis, pulmonary, renal and hepatic thromboembolism, right ventricle dilatation, infarction with and without coronary occlusion, small-vessel disease, and intraventricular thrombosis. These findings indicate that, despite the known risks of death associated with use of AAS, the harms associated with its misuse are not fully understood.
GLOBAL ALERT FOR AAS MISUSE:
Despite all the research about the risks associated with misuse of AAS, it unfortunately continues to occur. A global warning should be sent to thousands of people, especially young AAS users, who are at increased risk of AMI, sudden death, and other diseases [5,44–46]. Our case report reinforces statements made by the Brazilian Federal Medicine Council [47] and specialty societies refuting the use of AAS for aesthetic purposes or to gain benefits in sports [44–46]. Similar alerts have been issued by European public health agencies [48] and the United States government [49].
Conclusions
We described the case of a 26-year-old male bodybuilder who used AAS, presented with an AMI, and was treated with primary angioplasty. Documentation of high serum levels of lipids and hormone changes in this patient 1 week before the acute event may suggest a role for anabolic steroids in the development of AMI in young men. Based on current evidence, AAS should not be prescribed without a clear medical indication.
Figures
Tables
Table 1.. Results of laboratory tests 1 week before AMI.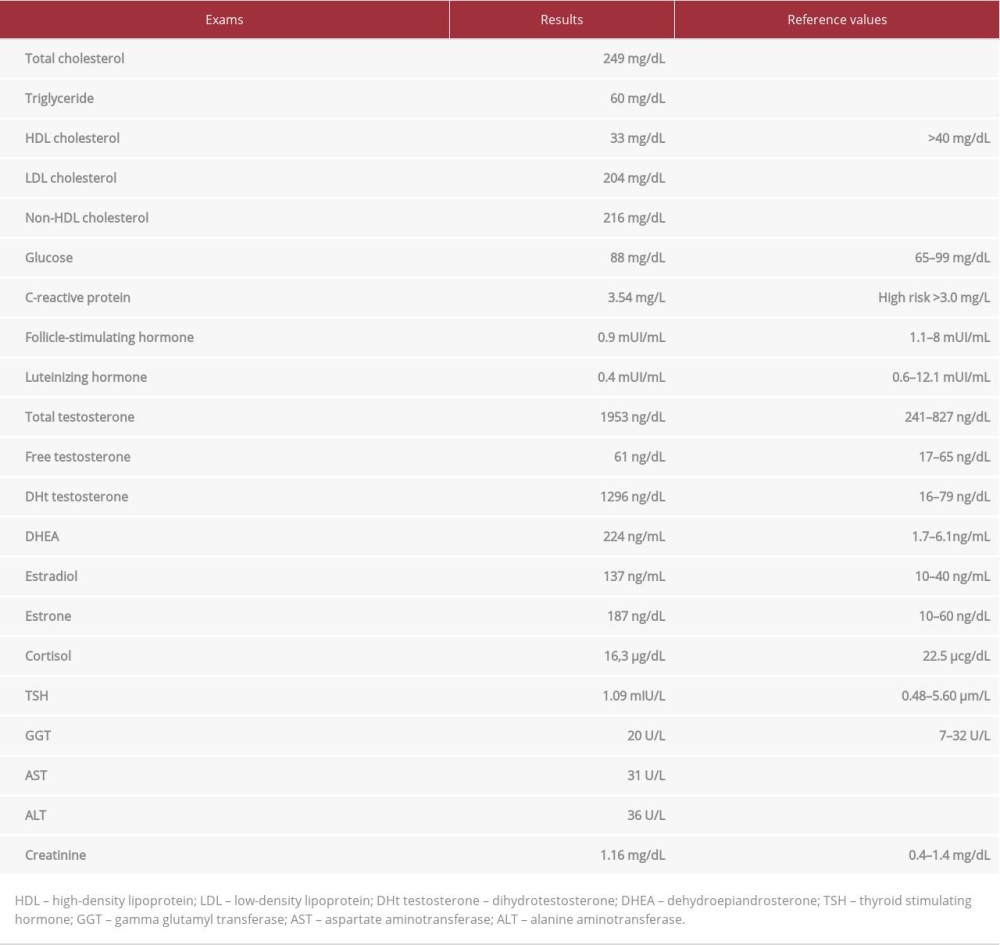 Table 2.. Myocardial necrosis markers on hospital admission.
Table 2.. Myocardial necrosis markers on hospital admission.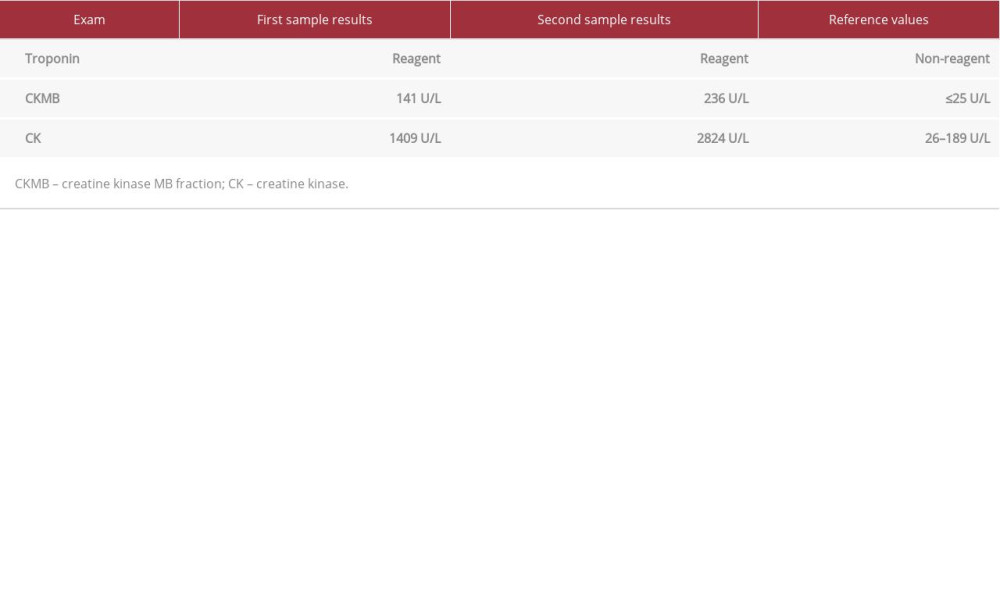 Table 3.. Pre-discharge Doppler echocardiography results.
Table 3.. Pre-discharge Doppler echocardiography results.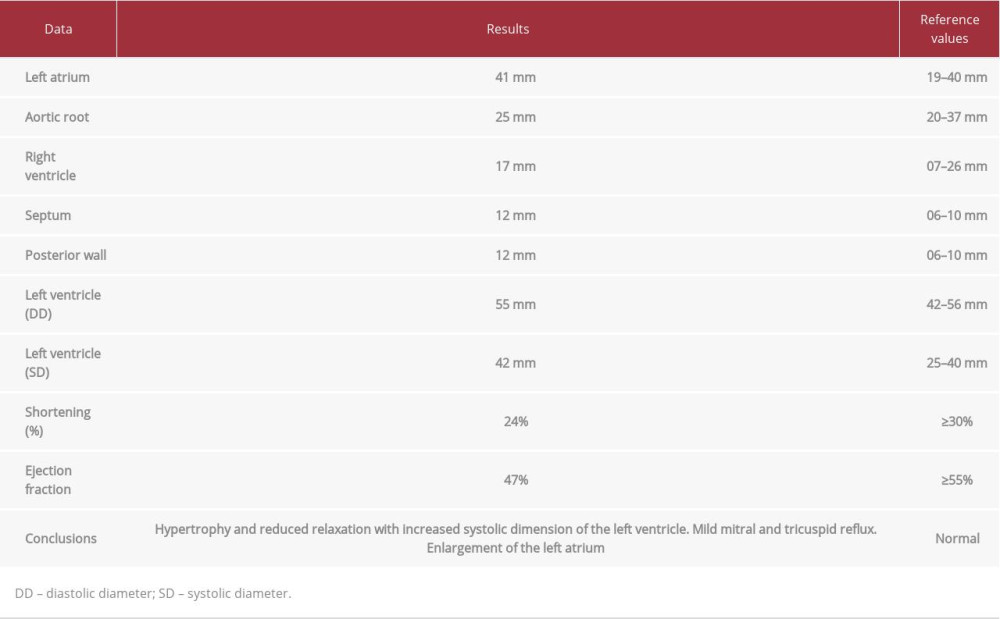 Table 4.. Doppler echocardiography 4 weeks after AMI.
Table 4.. Doppler echocardiography 4 weeks after AMI.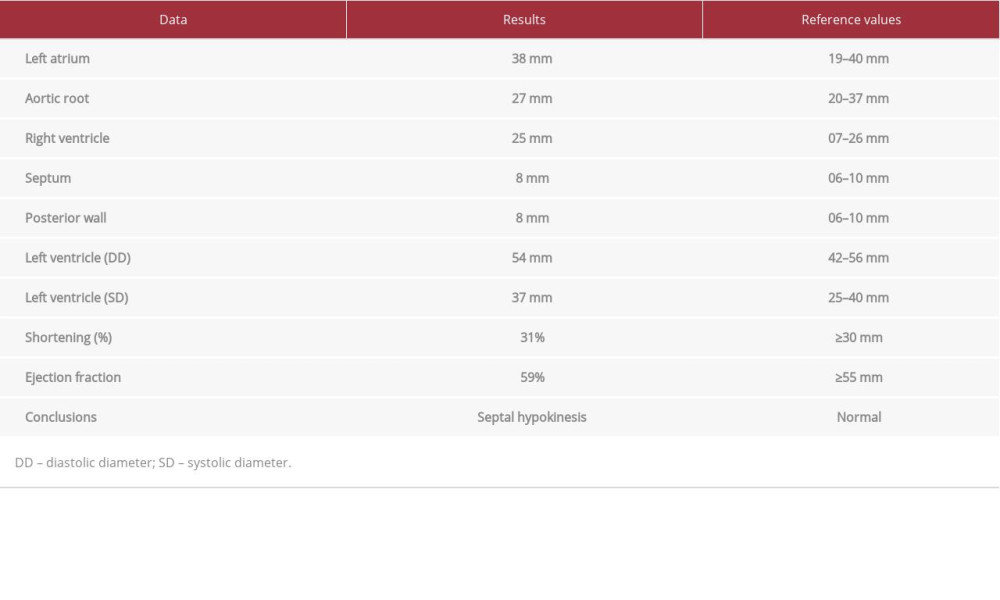 Table 5.. Laboratory results 4 weeks after AMI.
Table 5.. Laboratory results 4 weeks after AMI.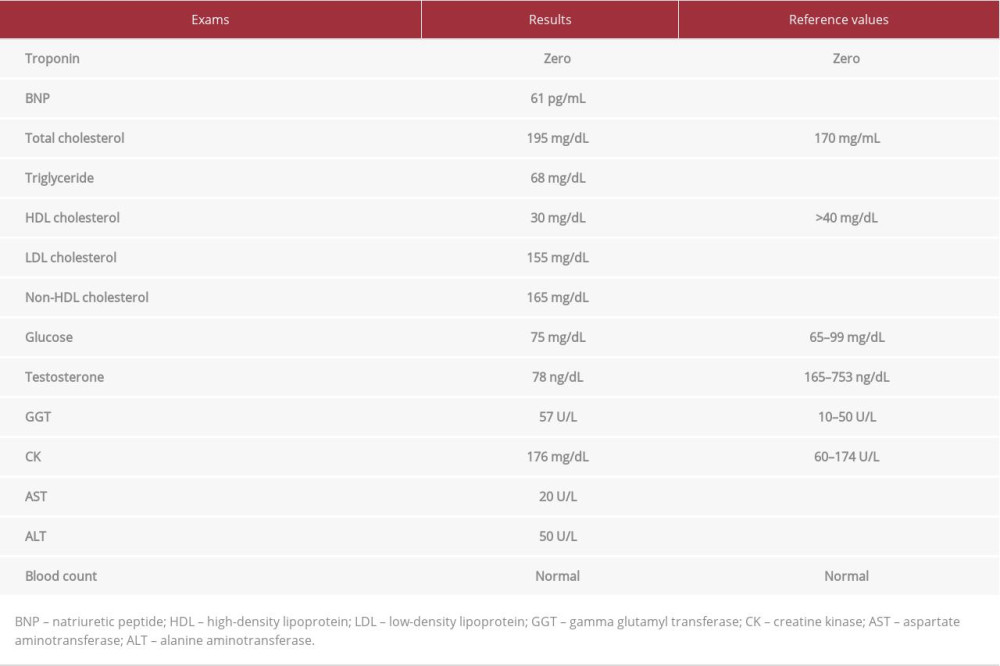 Table 6.. Adverse effects associated with the use of anabolic steroids. (Adapted from Hoffmann JR, Ratamess NA, 2006 [5]).
Table 6.. Adverse effects associated with the use of anabolic steroids. (Adapted from Hoffmann JR, Ratamess NA, 2006 [5]).![Adverse effects associated with the use of anabolic steroids. (Adapted from Hoffmann JR, Ratamess NA, 2006 [5]).](https://jours.isi-science.com/imageXml.php?i=t6-amjcaserep-21-e924796.jpg&idArt=924796&w=1000)
References:
1.. Abrahin OSC, Sousa EC, Androgenic anabolic steroids and its side effects: A critical scientific review: Rev Educ Fisuem, 2013; 24(4); 669-79
2.. Sagoe D, Molde H, Andreassen CS, The global epidemiology of anabolic-androgenic steroid use: A meta-analysis and meta-regression analysis: Annals Epidemiol, 2014; 24(5); 383-98
3.. Iriart JAB, Chaves JC, Orleans RG, [Body worship and the use of anabolics in weight trainers]: Cad Saúde Pública, 2009; 25(4); 773-82 [in Portuguese]
4.. Cecchetto F, Moraes DR, Farias OS, [Distinct approaches towards anabolic steroids: Risks to health and hypermasculinity]: Interface, 2012; 16(41); 369-82 [in Portuguese]
5.. Hoffmann JR, Ratamess NA, Medical issues associated with anabolic steroids use: Are they exaggerated?: J Sports Sci Med, 2006; 5; 182-93
6.. Corona G, Rastrelli G, Guaraldi F, An update on heart disease risk associated with testosterone boosting medications: Expert Opin Drug Safety, 2019; 18(4); 321-32
7.. Santos RP, Pereira A, Guedes H, Anabolic drugs and myocardial infarction – a clinical case report: Arq Bras Cardiol, 2015; 105(3); 316-19
8.. Powers M, The safety and efficacy of anabolic steroid precursors: What is the scientific evidence?: J Athletic Training, 2002; 37(3); 300-5
9.. Sorwell KG, Kohama SG, Urbanski HF, Testosterone increases circulating dehydroepiandrosterone sulfate levels in the male rhesus macaque: Front Endocrinol (Lausanne), 2014(5); 101
10.. Dharia S, Slane A, Jian M, Effects of aging on cytochrome b5 expression in the human adrenal gland: J Clin Endocrinol Metabol, 2005; 90(7); 4357-61
11.. Goodale T, Sadhu A, Petak S, Testosterone and the heart: Methodist Debakey Cardiovasc J, 2017; 13(2); 68-72
12.. Morgentaler A, Miner MM, Caliber M, Testosterone therapy and cardiovascular risk: Advances and controversies: Mayo Clin Proc, 2015; 90(2); 224-51
13.. , Ciclos de esteróides anabolizantes: Equipe Hipertrofia, 2019 [in Portuguese]https://www.hipertrofia.org/blog/2007/05/20/ciclos-esteroides-anabolizantes/
14.. Elliot D, Cheong JW, Moe E, Cross-sectional study of female students reporting anabolic steroid use: Arch Pediatr Adolesc Med, 2004; 161; 572-77
15.. Ip EJ, Barnett MJ, Tenerowicz MJ, Women and anabolic steroids: An analysis of a dozen users: Clin J Sport Med, 2010; 20; 475-81
16.. Cohen J, Collins R, Darkes J, A league of their own: Demographics, motivations and patterns of use of 1,955 male adult non-medical anabolic steroid users in the United States: J Int Soc Sports Nutrition, 2007; 4(1); 12-26
17.. Christiansen NK, Behavioural effects of androgen in men and women: J Endocrinol, 2001; 170; 39-48
18.. Ferner RE, Rawlins MD, Anabolic steroids: The power and the glory?: BMJ, 1988; 297(6653); 877-82
19.. Rocha M, Aguiar F, Ramos H, O uso de esteroides androgénicos anabolizantes e outros suplementos ergogénicos – uma epidemia silenciosa: Rev Port Endocrinol Diabetes Metab, 2014; 9(2); 98-105 [in Portuguese]
20.. Oliveira LL, Cavalcante-Neto JL, [Social and demographic factors, useres profile and motivations for using anabolic steroids in young adults]: Revista Brasileira de Ciências do Esporte, 2018; 40(3); 309-17 [in Portuguese]
21.. Perry HM, Wright D, Littlepage BN, Dying to be big: A review of anabolic steroid use: Br J Sports Med, 1992; 26(4); 259-61
22.. Romero-Martinez A, Moya-Albiol L, [Mechanisms facilitating anger expression through steroid abuse. Could steroids facilitate the onset of violence?]: Revista Española de Drogodependencias, 2015; 40(3); 12-26 [in Portuguese]
23.. Skärkberg K, Nyberg E, Engström I, Is there an association between the use of anabolic steroids and criminality?: Eur Addict Res, 2010; 16(4); 213-19
24.. Faludi AA, Izar MCO, Saraiva JFK, [Brazilian guidelines for dyslipidaemia and atherosclerosis prevention 2017]: Arq Bras Cardiol, 2017; 109(1); 1-76 [in Portuguese]
25.. Achar S, Rostamian A, Narayan SM, Cardiac and metabolic effects of anabolic-androgenic steroid abuse on lipids, blood pressure, left ventricular dimensions, and rhythm: Am J Cardiol, 2010; 106(6); 893-901
26.. Angell P, Chester N, Green D, Anabolic steroids and cardiovascular risk: Sports Med, 2012; 42(2); 119-34
27.. Higgins JP, Heshmat A, Higgins CL, Androgen abuse and increased cardiac risk: South Med J, 2012; 105(12); 670-74
28.. Grace FM, Davies B, Raised concentrations of C reactive protein in anabolic steroid using bodybuilders: Br J Sports Med, 2004; 38; 97-98
29.. Baggish AL, Weinee RB, Kanayama G, Cardiovascular toxicity of illicit anabolic-androgenic steroid use: Circulation, 2017; 135; 1991-2002
30.. Christou GA, Christou KA, Nikas DN, Acute myocardial infarction in a young bodybuilder taking anabolic androgenic steroids: A case report and critical review of the literature: Eur J Prev Cardiol, 2016; 23(16); 1785-96
31.. Sonmez E, Turkdogan KA, Yilmaz C, Chronic anabolic androgenic steroid usage associated with acute coronary syndrome in bodybuilder: Turkish J Emerg Med, 2016; 16(1); 35-37
32.. Poorzand H, Esfehani RJ, Hosseinzadeh P, Acute myocardial infarction in a young male wrestler: A case report: ARYA Atheroscler, 2015; 11(6); 366-69
33.. Shahsavari K, Rahmani F, Bakhtavar FE, A young man with myocardial infarction due to trenbolone acetate: A case report: Emergency, 2014; 2(1); 43-45
34.. Peoples K, Kobe D, Campana C, Hyperhomocysteinemia-induced myocardial infarction in a young male using anabolic steroids: Am J Emerg Med, 2014; 948; e1-2
35.. Ilhan E, Demirci D, Güvenç TS, Acute myocardial infarction and renal infarction in a bodybuilder using anabolic steroids: Arch Turk Soc Cardiol, 2010; 38(4); 275-78
36.. Wysocanski M, Rachko M, Bergmann SR, Acute myocardial infarction in a young man using anabolic steroids: Angiology, 2008; 59(3); 376-78
37.. Günes Y, Erbas C, Okuyan E, Myocardial infarction by anabolic steroids: Anadolu Kardiyol Derg, 2004; 4; 357-58
38.. Halvorsen S, Thorsby PM, Haug E, [Acute myocardial infarction in a young man who had been using androgenic anabolic steroids]: Tidsskr Nor Laegeforen, 2004; 124(2); 170-72 [in Norwegian]
39.. Tischer KH, Hyny-von-hausen R, Mall G, Coronary thrombosis and ectasia of coronary arteries after long term use of anabolic steroids: Z Kardiol, 2003; 92(4); 326-31 [in German]
40.. Sobreira Filho FM, Lino DOC, Belém LS, Acute myocarditis in user of anabolic hormones diagnosed by magnetic resonance imaging: A case report: Arq Bras Cardiol, 2018; 31(3); 207-10
41.. Frati P, Busardò FP, Cipolloni L, Anabolic androgenic steroid (AAS) related deaths: Autoptic, histopathological and toxicological findings: Curr Neuropharmacol, 2015; 13; 146-59
42.. Helal L, Ferrari F, Stein R, Sudden death in young Brazilian athletes: Isn’t It time we created a genuinely national register?: Arq Bras Cardiol, 2018; 111(6); 856-59
43.. Montisci M, Mazloum RE, Cechetto G, Anabolic androgenic steroids abuse and cardiac death in athletes: Morphological and toxicological findings in four fatal cases: Forensic Sci Int, 2012; 217(1–3); e13-18
44.. Simon SD: (org.): Brazilian Society of Clinical Oncology statement about not recognizing modulation hormonal specialty, 2019, Sociedade Brasileira de Oncologia Clínica [in Portuguese]https://sboc.org.br/noticias/item/1461-posicionamentoda-sboc-sobre-o-nao-reconhecimento-de-especialista-em-modulacao-hormonal
45.. Pinheiro JES: (org.): Ageing is not illness, 2017, Brazilian Society of Geriatrics and Gerontology [in Portuguese]https://sbgg.org.br/envelhecer-nao-e-doenca-sbgg-emiteposicionamento-em-retorno-as-colocacoes-expressas-pelo-pesquisador-aubrey-de-grey-que-quer-curar-o-envelhecimento/
46.. Hohl A: (org.): Clarification note to health professionals and population, 2016, Brazilian Endocrinology and Metabology Society https://www.endocrino.org.br/media/uploads/nota_oficial_sbem_12_12_16.pdf
47.. : Why hormonal modulation in not a recognized practice by Federal Medicine Council?, 2019, Conselho Federal de Medicina do Brasil https://pebmed.com.br/por-que-a-modulacao-hormonal-nao-e-uma-pratica-reconhecida-pelo-cfm
48.. Abbate V, Kicman AT, Evans-Brown M, Anabolic steroids detected in bodybuilding dietary supplements – a significant risk to public health: Drug Test Analysis, 2015; 7; 609-18
49.. , Community Drug Alert Bulletin – Anabolic Steroids, 2000 Retrieved from https://archives.drugabuse.gov/publications/nida-community-drug-alert-bulletin-anabolic-steroids
Figures
Tables
 Table 1.. Results of laboratory tests 1 week before AMI.
Table 1.. Results of laboratory tests 1 week before AMI. Table 2.. Myocardial necrosis markers on hospital admission.
Table 2.. Myocardial necrosis markers on hospital admission. Table 3.. Pre-discharge Doppler echocardiography results.
Table 3.. Pre-discharge Doppler echocardiography results. Table 4.. Doppler echocardiography 4 weeks after AMI.
Table 4.. Doppler echocardiography 4 weeks after AMI. Table 5.. Laboratory results 4 weeks after AMI.
Table 5.. Laboratory results 4 weeks after AMI. Table 6.. Adverse effects associated with the use of anabolic steroids. (Adapted from Hoffmann JR, Ratamess NA, 2006 [5]).
Table 6.. Adverse effects associated with the use of anabolic steroids. (Adapted from Hoffmann JR, Ratamess NA, 2006 [5]). Table 1.. Results of laboratory tests 1 week before AMI.
Table 1.. Results of laboratory tests 1 week before AMI. Table 2.. Myocardial necrosis markers on hospital admission.
Table 2.. Myocardial necrosis markers on hospital admission. Table 3.. Pre-discharge Doppler echocardiography results.
Table 3.. Pre-discharge Doppler echocardiography results. Table 4.. Doppler echocardiography 4 weeks after AMI.
Table 4.. Doppler echocardiography 4 weeks after AMI. Table 5.. Laboratory results 4 weeks after AMI.
Table 5.. Laboratory results 4 weeks after AMI. Table 6.. Adverse effects associated with the use of anabolic steroids. (Adapted from Hoffmann JR, Ratamess NA, 2006 [5]).
Table 6.. Adverse effects associated with the use of anabolic steroids. (Adapted from Hoffmann JR, Ratamess NA, 2006 [5]). In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








