10 September 2020: Artilces 
Autoimmune Myelofibrosis in Sjögren’s Syndrome: Report of a Case
Rare disease
Seiji Kakiuchi1ABDEF*, Ikumi Takagi1ABD, Hiroaki Akiyama1BD, Hiroyuki Matsuba1BD, Junpei Rikitake1BD, Kazuyoshi Kajimoto2BD, Yoshitake Hayashi3BD, Nobuko Iwata1BDDOI: 10.12659/AJCR.924983
Am J Case Rep 2020; 21:e924983
Abstract
BACKGROUND: Autoimmune myelofibrosis (AMF) is a rare clinicopathologic entity of bone marrow fibrosis that occurs in association with autoimmune disorders. Steroids are very effective for treatment of AMF and the disease has a good prognosis and should be distinguished from primary myelofibrosis.
CASE REPORT: A 49-year-old man with bleeding and petechial hemorrhage of the extremities presented to our institution. His platelet count was 1×10⁹/L. Bone marrow aspiration revealed a dry tap, and bone marrow biopsy confirmed small lymphocyte infiltration and increased reticular fibers, consistent with immune thrombocytopenia. Testing for mutations in JAK2, MPL, and CALR was negative. Because the patient had a history of Raynaud’s phenomenon, he was suspected to have collagen disease. Anti-Sjögren’s-syndrome-related antigen-A antibody testing, Schirmer’s test, and fluorescein staining all came back positive, which led to a diagnosis of Sjögren’s syndrome. Given the bone marrow findings, the patient also was diagnosed with AMF. Treatment with steroids resulted in an immediate improvement in his platelet count.
CONCLUSIONS: In the present case, treatment with steroids resulted in prompt improvement in platelet counts and subsequent marrow biopsy showed MF-0 reticulin fibrosis. Bone marrow fibrosis rarely is seen in association with autoimmune disease, and its significance and mechanism are still to be determined.
Keywords: Autoimmune Diseases, Bone Marrow Examination, primary myelofibrosis, Sjogren's Syndrome, Steroids, Bone Marrow, Thrombocytopenia
Background
Myelofibrosis is a disease that causes widespread bone marrow fibrosis as a result of proliferation of reticular and collagen fibers, and bone marrow fibroblasts. There are 2 types of myelofibrosis: primary myelofibrosis of unknown cause (PMF) and myelofibrosis secondary to other diseases [1,2]. Myelofibrosis that occurs secondary to autoimmune abnormalities is termed autoimmune myelofibrosis (AMF) [3]. Here, we describe an extremely rare case in which a patient with Sjögren’s syndrome received concomitant treatment with steroids for PMF.
Case Report
A 49-year-old Japanese man presented to our hospital with a 3-day history of oral bleeding and epistaxis. The patient was admitted when blood testing revealed a platelet count of 1×109/L. He reported a long history of Raynaud’s phenomenon, although the exact duration was unknown. An examination 8 months prior to the current hospitalization revealed no abnormalities other than a slight decrease in platelet count to 110×109/L.
A physical examination of the patient revealed a blood pressure of 133/96 mmHg, heart rate of 109 beats per minute, respiratory rate of 16 inspirations per minute, and body temperature of 37.4°C. Lymph nodes in the liver, spleen, and other areas of the body were not palpable. Petechiae were present on the buccal mucosa and limbs. Blood testing showed marked thrombocytopenia, mild lactate dehydrogenase elevation (398 U/L), and haptoglobin decline (46 mg/dL). Increased levels of antinuclear antibodies (ANAs) (×1280), rheumatoid factor (200 IU/mL), anti-Sjögren’s-syndrome-related antigen-A (anti-SS-A) antibodies (×256), and platelet-associated immunoglobulin (PAIgG) (3240 ng/107 cells) were observed. Direct and indirect Coombs tests were negative. A thoracoabdominal computed tomography scan showed a cyst in the lung field and no hepatosplenomegaly. A bone marrow biopsy showed hyperplastic bone marrow, with a slight increase in the number of megakaryocytes to 44 cells/mm2; however, no atypia or aggregation was observed. Immunostaining revealed aggregation of CD3- and CD20-positive, atypical small lymphocytes, and no clonality of κ or λ chains. Silver staining showed MF-2 fibrosis (Figure 1A). Fluorescence
Bone marrow examination on admission showed a dry tap and collagen fibers in the patient’s bone marrow biopsy stamp specimen. Because of his oral bleeding, a platelet transfusion was performed; however, the platelet elevation was only temporary, his anemia progressed gradually, and he developed malaise.
After the patient was diagnosed with AMF, he was treated with dexamethasone, (40 mg/day for 4 days), followed by predniso-lone (PSL) (1 mg/kg), which was continued and gradually dose-tapered. Immediately after the treatment began, the patient’s platelet counts increased, blood transfusion was considered unnecessary, and his anemia also improved. On Day 175, PSL was discontinued after being tapered. On Day 182, a bone marrow examination was performed and aspiration was possible. The bone marrow biopsy showed no accumulation of lymphocytes and silver staining showed improvement in fibrosis to MF-0 (Figure 1B). On the same day, the patient’s serum transforming growth factor-β1(TGF-β1) level was 1.80 ng/mL versus a pretreatment value of 1.75 ng/mL; it was within the standard range and did not decrease. On Day 257, the patient’s platelet count dropped to 110×109/L, and PSL was resumed at a dose of 10 mg/day. Currently, about 14 months later, he is taking PSL 5 mg/day and has experienced no cytopenia.
Discussion
Distinguishing AMF and PMF histologically is extremely important because they differ greatly in treatment and prognosis. Peripheral blood findings may show teardrop erythrocytes and leukoblastosis in PMF, although these findings are rare in AMF. There are also marked pathological differences in bone marrow findings. In contrast to PMF, AMF does not present with megakaryocyte aggregation or atypia, granulocytic or erythroblastic atypia, or granulocytic hyperplasia, but it does produce erythroblastic or megakaryocytic hyperplasia. The majority of patients with AMF have disease classified under the European Consensus grading system for bone marrow fibrosis as MF-1, with osteosclerosis and fibrosis with systemic hyperplasia. Other characteristics that distinguish AMF from PMF are lack of or slight splenomegaly, positive autoantibodies, and no mutations in
Conditions with serologically positive autoantibodies but not clinically diagnosed as autoimmune disease with myelofibrosis are called primary AMF, and are closely related to immune thrombocytopenia (ITP), autoimmune hemolytic anemia, and Evans syndrome [5]. In contrast, those secondary to autoimmune diseases, such as systemic lupus erythematosus (SLE) and rheumatoid arthritis, are specifically called secondary AMF. Regarding the treatment of secondary AMF, a review of data from 32 cases of AMF secondary to SLE showed that 29 patients achieved improvement with steroids, one-third of them within 2 weeks after initiation of treatment. Reports indicate that in patients with treatment resistance or in whom disease improvement is poor, intravenous immunoglobulin, azathioprine, and cyclophosphamide are considered effective [3]. In approximately 50% of cases, however, bone marrow fibrosis cannot be improved, even if a normal blood count is restored [6].
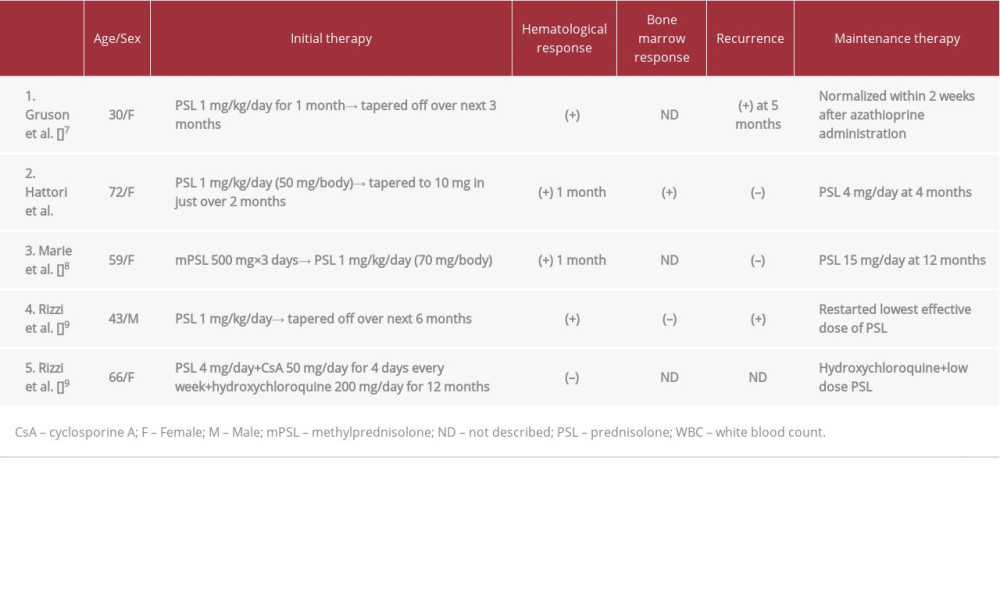
It is extremely rare to have AMF with Sjögren’s syndrome, and there are only 5 reported AMF cases published in the English literature [7–10] (Table 1); 4 of 5 patients in these cases were female. Although their responsiveness to steroid treatment reportedly was good, 2 of them experienced exacerbations, as happened in our case, and they were retreated with steroids or treated with azathioprine. In the present case, the therapeutic effect was again observed when steroid administration resumed, and the myelofibrosis gradually decreased again. Notably, our patient’s myelofibrosis resolved following treatment with steroids, unlike in the other 4 cases.
Although the distinction between AMF and PMF is important, there is often an overlap in pathological features that results in not all diagnostic criteria being met [2,5,11,12]. Another factor that makes diagnosis difficult is that ANAs and rheumatoid factors are not serologically specific to AMF [11]. In particular, cases of thrombocytopenia alone, as in the present report, are difficult to differentiate from ITP, and some cases have been reported that were initially misdiagnosed as ITP [13]. Therefore, immunological testing and bone marrow biopsy are essential to make an accurate diagnosis. In the present case, AMF was characterized by dry tap, increased rheumatoid factor, and progression of malaise and anemia during the clinical course, which is characteristic of the condition [2].
The mechanism by which AMF causes bone marrow fibrosis remains unclear. According to a report by Harrison et al., it has been hypothesized that in patients with AMF, TGF-β and substance P are produced from monocytes and CD4-positive cells in peripheral blood. Thus TGF-β and substance P levels become higher than in healthy individuals, resulting in myelofibrosis [14]. However, in our patient, TGF-β1 was within the reference range and did not decrease before or after treatment. Reports of more such cases may help to elucidate the mechanism of action.
Conclusions
This is an extremely rare case report describing the resolution of myelofibrosis after treatment with steroids for AMF secondary to Sjögren’s syndrome. Because AMF is often accompanied by an underlying collagen disease, careful immunological testing and bone marrow biopsy are crucial. The clinical significance of myelofibrosis in patients with AMF is not yet clear. The condition is probably underrecognized and needs further study.
References:
1.. Tefferi A, Primary myelofibrosis: 2014 update on diagnosis, risk-stratification, and management: Am J Hematol, 2014; 89; 915-25
2.. Vergara-Lluri ME, Piatek CI, Pullarkat V, Autoimmune myelofibrosis: An update on morphologic features in 29 cases and review of the literature: Hum Pathol, 2014; 45; 2183-91
3.. Koduri PR, Parvez M, Kaza S, Vanajakshi S, Autoimmune myelofibrosis in systemic lupus erythematosus report of two cases and review of the literature: Indian J Hematol Blood Transfus, 2016; 32; 368-73
4.. Marcellino B, El Jamal SM, Mascarenhas JO, Distinguishing autoimmune myelofibrosis from primary myelofibrosis: Clin Adv Hematol Oncol, 2018; 16; 619-26
5.. Bass RD, Pullarkat V, Feinstein DI, Pathology of autoimmune myelofibrosis. A report of three cases and a review of the literature: Am J Clin Pathol, 2001; 116; 211-16
6.. Paquette RL, Meshkinpour A, Rosen PJ, Autoimmune myelofibrosis. A steroid-responsive cause of bone marrow fibrosis associated with systemic lupus erythematosus: Medicine (Baltimore), 1994; 73; 145-52
7.. Gruson B, Brevet M, Vaida I, Myelofibrosis and cytopenia are not always malignant: Eur J Intern Med, 2006; 17; 136-37
8.. Marie I, Levesque H, Cailleux N, An uncommon association: Sjögren’s syndrome and autoimmune myelofibrosis: Rheumatology (Oxford), 1999; 38; 370-71
9.. Rizzi R, Pastore D, Liso A, Autoimmune myelofibrosis: Report of three cases and review of the literature: Leuk Lymphoma, 2004; 45; 561-66
10.. Takahashi T, Autoimmune myelofibrosis accompanied by Sjogren’s syndrome in a 47, XXX/46, XX mosaic woman: Intern Med, 2014; 53(7); 783-87
11.. Piatek CI, Vergara-Lluri ME, Pullarkat V, Autoimmune myelofibrosis: Clinical features, course, and outcome: Acta Haematol, 2017; 138; 129-37
12.. Pullarkat V, Bass RD, Gong JZ, Primary autoimmune myelofibrosis: Definition of a distinct clinicopathologic syndrome: Am J Hematol, 2003; 72; 8-12
13.. Hua J, Matayoshi S, Uchida T, Primary autoimmune myelofibrosis with severe thrombocytopenia mimicking immune thrombocytopenia: A case report: Mol Clin Oncol, 2016; 5; 789-91
14.. Harrison JS, Corcoran KE, Joshi D, Peripheral monocytes and CD4+ cells are potential sources for increased circulating levels of TGF-beta and substance P in autoimmune myelofibrosis: Am J Hematol, 2006; 81; 51-58
In Press
05 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942032
06 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942937
12 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943244
13 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943275
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250