09 December 2020: Articles 
Propylthiouracil-Induced Anti-Neutrophil Cytoplasmic Antibody Vasculitis Presenting with Red Eye Followed by Pulmonary Hemorrhage: Diagnostic and Management Considerations
Challenging differential diagnosis, Rare disease, Adverse events of drug therapy
Marjan Alidoost1EF, Jennifer Cheng2EF, Deborah R. Alpert3ABEF*DOI: 10.12659/AJCR.925200
Am J Case Rep 2020; 21:e925200
Abstract
BACKGROUND: Drug-induced anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) should be suspected in patients on certain medications who present with inflammatory ocular, constitutional, pulmonary, and/or renal manifestations. Here, we present a case of propylthiouracil (PTU)-induced AAV presenting initially with red eye, and review important diagnostic and management considerations for this uncommon disorder.
CASE REPORT: A 34-year-old woman with hyperthyroidism taking PTU presented with red eye, later followed by fevers and hemoptysis. She was found to have episcleritis, diffuse alveolar hemorrhage, and microhematuria. The infectious diseases workup was unrevealing. Laboratory evaluations were notable for a high-titer perinuclear ANCA and elevated anti-myeloperoxidase antibodies. Renal function was normal. She was ultimately diagnosed with PTU-induced AAV. PTU was promptly discontinued and she was treated with pulse-dose methylprednisolone for 3 days, followed by prednisone 60 mg daily. A kidney biopsy revealed pauci-immune focal segmental necrotizing and crescentic glomerulonephritis. Given an allergy to methimazole, she underwent thyroidectomy and was ultimately treated with rituximab. Her steroid doses are progressively being tapered and she has complete resolution of symptoms.
CONCLUSIONS: PTU-induced AAV is a rare and serious condition. Our patient presented with ocular symptoms prior to more commonly recognized pulmonary and renal manifestations. Patients may have favorable outcomes if PTU is discontinued promptly, but patients with vital-organ involvement may require treatment with steroids and may need additional immunosuppression.
Keywords: Anti-Neutrophil Cytoplasmic Antibody-Associated Vasculitis, Conjunctivitis, Hemorrhage, Propylthiouracil, Antibodies, Antineutrophil Cytoplasmic, Antithyroid Agents, Peroxidase
Background
Vasculitis is characterized by inflammation in blood vessel walls that can cause damage to visceral organs, which may occur via ischemia or necrosis [1]. It is often challenging to diagnose vasculitis, since it may present with a wide variety of symptoms that can potentially affect any organ system. Vasculitis can have numerous presentations, including constitutional symptoms such as fever, arthralgias, and myalgias, as well as various organ manifestations affecting the eyes, skin, nervous system, heart, lungs, gastrointestinal tract, and kidneys [2]. Vasculitis can be classified by size of affected vessels, as well as by primary versus secondary cause. In vasculitis patients, it is estimated that 90% of c-ANCA is directed against proteinase 3 (PR3), while 80-90% of p-ANCA is directed against myeloperoxidase (MPO) [3]. An important subtype of secondary vasculitis is drug-induced vasculitis, which is often an ANCA-associated necrotizing small-vessel vasculitis [2]. Drugs that may be associated with ANCA-associated vasculitis (AAV) include hydralazine, minocycline, ciprofloxacin, phenytoin, levamisole-adulterated cocaine, and propylthiouracil (PTU) [4]. We describe a patient taking PTU for hyperthyroidism who presented initially with episcleritis, prior to a pulmonary-renal syndrome that was diagnosed as PTU-induced vasculitis. Our case highlights the importance of prompt recognition of PTU-induced vasculitis presenting initially with ocular symptoms, cessation of the offending agent, immunosuppressive treatment, and appropriate treatment of the underlying thyroid disease.
Case Report
A 34-year-old woman with a history of Grave’s hyperthyroidism maintained on PTU for 2 years presented to the Emergency Department with irritated bilateral red eyes for 3 weeks. She denied any visual change or eye discharge. She also reported sneezing and coughing, and denied myalgias, arthralgias, fever, or fatigue. Her ocular symptoms were initially attributed to allergic conjunctivitis, but as they did not improve, she was given a trial of besifloxacin eye drops. A chest X-ray showed a patchy right lower-lobe airspace opacity with air bronchograms (Figure 1). At that time, she was diagnosed with community-acquired pneumonia and discharged with a course of oral levofloxacin. As an outpatient, she was subsequently evaluated by ophthalmology and diagnosed with episcleritis.
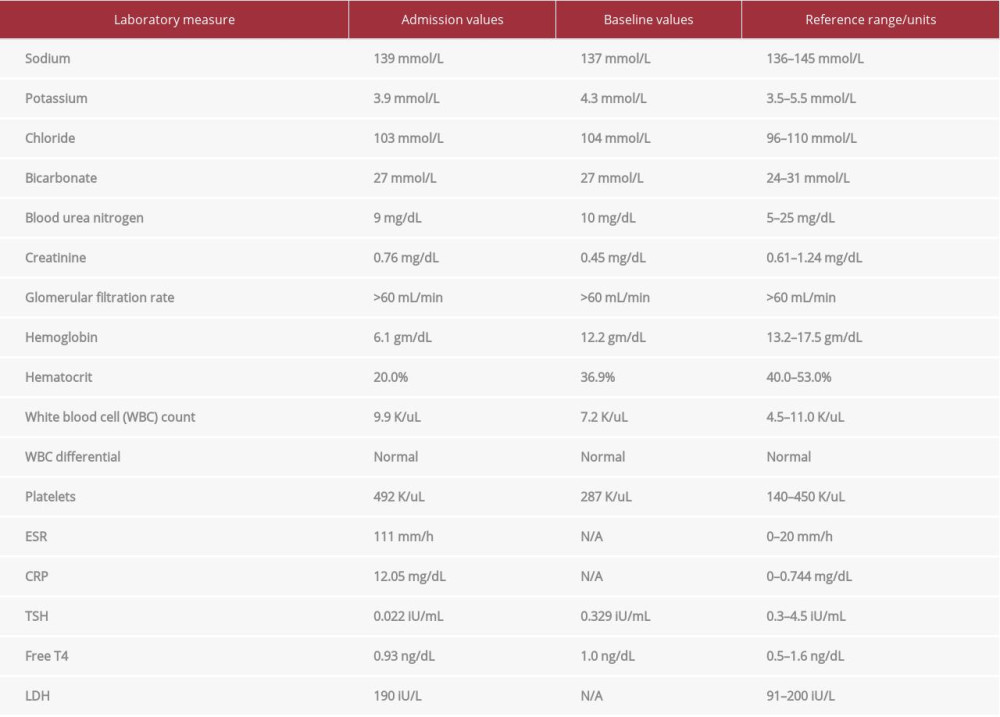
One month later, she presented to the hospital with hemoptysis for 2 weeks, coughing up 20–30 mL of coagulated blood with each episode. At that time, she also reported fevers with a maximum temperature of 38°C, fatigue, arthralgias, and myalgias. She denied decreased urine output, rash, hematuria, or hematochezia. Upon hospital admission, she was found to have severe anemia, with a hemoglobin of 6.1 g/dL, for which she was transfused with 2 units of packed red blood cells (Table 1). A CT angiogram on admission showed symmetric ground-glass opacities in the lungs, suggestive of pulmonary hemorrhage (Figure 2). Her respiratory status remained stable, with a respiratory rate of 14 breaths per minute and oxygen saturation levels of 97% on room air. An infectious diseases workup was negative, including blood and sputum cultures, fungal serologies, and testing for influenza and tuberculosis. Serologic evaluation revealed a negative antinuclear antibody (ANA), but a positive p-ANCA with a titer of 1: 5120. The anti-myeloperoxidase (MPO) level was 168 AU/ml (normal 0–19 AU/mL), and the anti-proteinase 3 (PR3) level was undetectable. Anti-glomerular basement membrane antibody was not detected. Anti-double-stranded DNA and anti-histone antibodies were not detected. Renal function was normal, but urinalysis revealed red blood cells too numerous to count, 15–20 white blood cells, and 3–5 hyaline casts. A kidney biopsy was performed, which revealed a pauci-immune focal segmental necrotizing and crescentic glomerulonephritis (Figure 3).
Since our patient previously had an urticarial reaction with methimazole, she was treated with potassium iodine (SSKI) and cholestyramine in addition to pulse-dose steroids. While her PTU was being held, she was given cholestyramine to increase thyroid hormone excretion via the enterohepatic circulation, and SSKI to block iodine uptake by the thyroid gland to minimize thyroid hormone production. Due to our patient’s intolerance of thionamides, she required a more permanent treatment for Grave’s disease, with either radioactive iodine (RAI) ablation or thyroidectomy. Total thyroidectomy was favored due to the risk for development of Grave’s ophthalmopathy with RAI ablation, and concern for abiding by radiation precautions with young children in the home [5]. She ultimately underwent total thyroidectomy and was later discharged to home on prednisone 60 mg daily, levothyroxine 125 mcg daily, and calcium carbonate with vitamin D supplements.
The patient was unfortunately lost to rheumatologic follow-up for 4 months. During that time, she continued taking prednisone 60 mg daily, although the steroid taper would have ideally been started sooner. Treatment with 2 doses of intravenous rituximab 1000 mg given 2 weeks apart was ultimately administered as an outpatient, and she is presently undergoing steroid taper. She remains with full resolution of symptoms, and her most recent p-ANCA titer had decreased to 1: 160, with the anti-MPO level decreased to 67 AU/ml (normal 0–19 AU/mL).
Discussion
Our patient initially presented with red eye, followed by infiltrate on chest X-ray. The main diagnostic consideration for her lung infiltrate at that time was community-acquired pneumonia.
She ultimately failed empiric antibiotic treatment for pneumonia and developed hemoptysis. At that point, an autoimmune process was considered. The differential diagnosis for episcleritis is broad, with approximately 20% to 30% of cases being idiopathic [6]. Autoimmune diseases, such as systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), Crohn’s disease, and vasculitis, may also cause episcleritis. Systemic vascular diseases and infections such as hepatitis, syphilis, and Lyme disease are other possible causes of episcleritis [6]. Her serologic workup, including serologies for infections, SLE, and RA, were negative. In the setting of microscopic hematuria, hemoptysis, and a high-titer p-ANCA with anti-myeloperoxidase antibodies, a diagnosis of AAV (specifically, PTU-induced AAV) was strongly suspected. Patients with PTU-induced vasculitis typically have p-ANCA rather than c-ANCA, although they can have a mixed picture with both p-ANCA and c-ANCA [6].
The pathophysiology of PTU-induced AAV is not entirely clear. One theory is that when PTU binds to MPO, it changes its structure and antigenicity, thus potentially triggering PTU-induced AAV [7]. PTU-induced AAV may develop after initiating PTU treatment or may develop after being on PTU for multiple years [8]. In patients with PTU-induced vasculitis, renal involvement is most common, affecting an estimated 72% of patients, and the pulmonary system is affected in approximately 26% of patients [9]. In patients with pulmonary manifestations, pulmonary hemorrhage is the most common manifestation, which can present as cough, dyspnea, or hemoptysis [10]. Other reported pulmonary sequelae include acute respiratory distress syndrome, hilar lymphadenopathy, and pulmonary infiltrates [11]. Fever and arthralgias are also common, and some patients present with isolated cutaneous findings [12].
Our patient had multiple symptoms of PTU-induced vasculitis. She initially presented with episcleritis, followed by pulmonary hemorrhage and microscopic hematuria. Data from 2 large randomized controlled trials of AAV demonstrated that 17.0% of patients initially presented with ocular involvement. Conjunctivitis and episcleritis were most common, while uveitis and retinal involvement were the rarest. Other ocular findings in AAV include proptosis, scleritis, and retinal hemorrhage [13]. We found 1 report of a 14-year-old girl who was diagnosed with PTU-induced AAV in the setting of anemia, followed by fever, arthritis, and episcleritis [14]. To the best of our knowledge, the present report is the first to describe a patient with PTU-induced AAV presenting initially with episcleritis.
PTU has many well-known adverse effects, ranging from rash to hepatotoxicity [12]. Importantly, PTU can cause drug-induced lupus (DIL), as well as AAV, as seen in our patient. Clinically, patients with DIL usually have more musculoskeletal and gastrointestinal symptoms. With PTU-induced DIL, ANA, anti-double-stranded DNA, and anti-histone antibodies may be positive, with accompanying low complement levels. Our patient did not have serologies consistent with DIL.
There are no formal guidelines for the treatment of PTU-induced AAV. PTU-induced AAV has classically been treated similarly to primary AAV with cyclophosphamide, rituximab, or, in some cases, plasmapheresis, if clinically indicated [15]. Patients who have no vital-organ involvement may be treated with withdrawal of the causative agent alone and monitored [2]. Those who have vital-organ involvement (e.g., renal or pulmonary involvement) are treated with pulse-dose, followed by oral steroids [1]. Patients with severe organ involvement, such as necrotizing glomerulonephritis and diffuse alveolar hemorrhage, often require treatment with pulse-dose steroids and another immunosuppressive agent such as cyclophosphamide or rituximab. The RAVE trial suggests that rituximab is non-inferior to cyclophosphamide in inducing remission in patients with primary AAV [16]. Some small retrospective studies have suggested that plasmapheresis can benefit patients with massive pulmonary hemorrhage [17]. However, given our patient’s prompt favorable response to PTU discontinuation and pulse-dose steroids, treatment with plasmapheresis was not deemed necessary. Of note, a recent randomized controlled trial with 352 patients comparing plasmapheresis with steroids to steroids alone for the treatment of AAV did not show a decrease in mortality or development of end-stage renal disease [18].
Patients with drug-induced vasculitis (DIV) tend to have better outcomes than those with primary AAV. A 2008 study by Gao et al. reviewed 15 patients with PTU-induced vasculitis, and all but 2 patients went into clinical remission [1]. Prior to use of immunosuppressive agents, primary AAV had a morbidity rate of 93% [19], while fatalities are considered rare for DIV if the offending agent is discontinued [1,19]. Additionally, primary AAV has a higher recurrence rate, estimated at 11–57% [1]. This has been attributed to the fact that DIV has a pathophysiologic trigger that can be removed [20]. Many patients with DIV improve and achieve remission simply with the cessation of the offending drug, whereas patients with primary AAV still require induction and maintenance treatment [19]. However, not all patients with DIV have complete resolution of symptoms with cessation of the offending agent. For instance, in a case series of 19 patients with PTU-induced AAV, 1 of 4 patients not treated with immunosuppressive agents had progression to end-stage renal disease [21]. It is unclear whether patients with DIV need maintenance therapy, but they are often given immunosuppressive therapy for 6-12 months [1]. Since our patient presented with diffuse alveolar hemorrhage and glomerulonephritis, despite improvement with PTU withdrawal and steroid treatment, the clinical decision was made to treat with rituximab to minimize risk of recurrence.
Conclusions
In this case report we describe a patient with PTU-induced AAV who initially presented with ocular symptoms, which are less common as the initial presentation of AAV and drug-induced AAV. If the suspicion for drug-induced AAV is high, it is important to promptly discontinue the offending medication and confirm the diagnosis. Treatment with high-dose steroids and, possibly, additional immunosuppressive agents may be considered, especially if there is involvement of vital organs. PTU-induced AAV is a rare condition that often has a good prognosis if recognized early. Our case highlights the importance of recognizing both uncommon and common manifestations of drug-induced AAV, to allow for prompt diagnosis and treatment.
Figures
References:
1.. Gao Y, Chen M, Ye H, Long-term outcomes of patients with propylthiouracil-induced anti-neutrophil cytoplasmic auto-antibody-associated vasculitis: Rheumatology, 2008; 47(10); 1515-20
2.. Radic M, Martinovic Kaliterna D, Radic J, Drug-induced vasculitis: A clinical and pathological review: Neth J Med, 2012; 70(1); 12-17
3.. Radice A, Sinico R, Antineutrophil cytoplasmic antibodies (ANCA): Autoimmunity, 2005; 35(1); 93-103
4.. Bossuyt X, Tervaert J-WC, Arimura Y, Revised 2017 international consensus on testing of ANCAs in granulomatosis with polyangiitis and microscopic polyangiitis: Nat Rev Rheumatol, 2017; 13(11); 683-92
5.. Walsh JP, Dayan CM, Potts MJ, Radioiodine and thyroid eye disease: BMJ, 1999; 319(7202); 68-69
6.. Schonberg S: Episcleritis, 2019, StatPearls, U.S National Library of Medicine https://www.ncbi.nlm.nih.gov/books/NBK534796/
7.. Shikha D, Harris J, Resta C, Park P, Antineutrophilic cytoplasmic antibody positive vasculitis associated with methimazole use: Case Rep Endocrinol, 2015; 2015; 530319
8.. Tetikkurt C, Yuruyen M, Tetikkurt S, Propylthiouracil-induced lupus-like or vasculitis syndrome: Multidisciplinary Respiratory Medicine, 2012; 7(1); 14
9.. Quax RAM, Swaak AJG, Baggen MGA, Churg-Strauss syndrome following PTU treatment: Int J Rheumatol, 2009; 2009; 504105
10.. Chen B, Yang X, Sun S, Propylthiouracil-induced vasculitis with alveolar hemorrhage confirmed by clinical, laboratory, computed tomography, and bronchoscopy findings: A case report and literature review: Iran Red Crescent Med J, 2016; 18(4); e23320
11.. Pillinger M, Staud R, Wegeners granulomatosis in a patient receiving propylthiouracil for Graves disease: Semin Arthritis Rheum, 1998; 28(2); 124-29
12.. Demir M, Yaylaci S, Tamer A, Ayturk S, Propylthiouracil induced leukocytoclastic vasculitis: A rare manifestation: Indian J Endocrinol Metabol, 2013; 17(2); 339
13.. Cai S, Papaliodis G, Lu L, Ocular manifestations of ANCA-associated vasculitis [abstract]: Arthritis Rheumatol, 2016; 68(Suppl. 10) Abstract Number: 1947
14.. Schamp V, Verfaillie C, Bonroy C, Propylthiouracil induced ANCA-associated vasculitis in a 14-year-old girl: Acta Clin Belg, 2015; 70(2); 127-29
15.. Khan TA, Luk FCY, Uqdah HT, A fatal case of propylthiouracil-induced ANCA-associated vasculitis resulting in rapidly progressive glomerulonephritis, acute hepatic failure, and cerebral angiitis: Clin Nephrol, 2015; 83(5); 309-14
16.. Stone JH, Merkel PA, Spiera R, Rituximab versus cyclophosphamide for ANCA-associated vasculitis: N Engl J Med, 2010; 363(3); 221-32
17.. Klemmer PJ, Chalermskulrat W, Reif MS, Plasmapheresis therapy for diffuse alveolar hemorrhage in patients with small-vessel vasculitis: Am J Kidney Dis, 2003; 42(6); 1149-53
18.. Walsh M, Merkel PA, Peh CA, Plasma exchange and glucocorticoids in severe ANCA-associated vasculitis: N Engl J Med, 2020; 382(7); 622-31
19.. Irani F, Elkambergy H, Naraharisetty K, Jamal R, Propylthiouracil-induced leucocytoclastic vasculitis with pulmonary hemorrhage treated with plasmapheresis: Am J Med Sci, 2009; 337(6); 470-72
20.. Chen Y-X, Zhang W, Chen X-N, Propylthiouracil-induced antineutro-phil cytoplasmic antibody (ANCA)-associated renal vasculitis versus primary ANCA-associated renal vasculitis: a comparative study: J Rheumatol, 2012; 39(3); 558-63
21.. Chen XC, Yu HJ, Ni LY, Propylthiouracil-associated antinutrophil cytoplasmic autoantibody-positive vasculitis: retrospective study of 19 cases: J Rheumatol, 2007; 34; 2451-56
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133