24 October 2020: Articles 
Chronic Lymphocytic Leukemia Concomitant with COVID 19: A Case Report
Unusual clinical course, Rare coexistence of disease or pathology
Elrazi Ali1ABDEF*, Mohamed Badawi2ADE, Elabbass Abdelmahmuod1EG, Samah Kohla3EG, Mohamed A. Yassin4ABDEDOI: 10.12659/AJCR.926062
Am J Case Rep 2020; 21:e926062
Abstract
BACKGROUND: COVID-19 is a newly emerging disease that is not yet fully understood. It is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a novel virus that is easily transmitted from human to human through the respiratory route. Usually, it presents with fever, headache, fatigue accompanied by respiratory symptoms like cough and dyspnea, and other systemic involvements. Chronic lymphocytic leukemia (CLL) is a common lymphoproliferative neoplasm characterized by absolute lymphocytosis and demonstration of clonality unlike other causes of lymphocytosis. Patients with CLL are considered immunocompromised because of impaired humoral immunity (mainly) and cellular immunity. Therefore, they are vulnerable to various infections including COVID-19. Little is known about the COVID-19 infection when it unmasks CLL.
CASE REPORT: A 49-year-old man with no significant previous illnesses, and an unremarkable family history, presented with a moderate COVID-19 infection. He initially presented to the emergency department with fever and mild shortness of breath. A complete blood count showed a high white blood cell count with absolute lymphocytosis. Flow cytometry revealed the clonality of the lymphocytes confirming the diagnosis of CLL. Despite having CLL, he developed a moderate COVID-19 infection and recovered in a few days. To the best of our knowledge, this is the first report of CLL, which presented with a COVID-19 infection as the initial presentation.
CONCLUSIONS: Lymphocytosis is an unexpected finding in patients diagnosed with COVID-19 infection and the elevated lymphocytes may be indicative of other conditions. Secondary causes of lymphocytosis like malignancy or other infections should be considered in these cases.
Keywords: Coronavirus Infections, COVID-19, Leukemia, Lymphocytic, Chronic, B-Cell, Betacoronavirus, COVID-19, Comorbidity, Immunocompromised Host, Pandemics, Pneumonia, Viral, SARS-CoV-2
Background
The COVID-19 infection is primarily a respiratory viral infection caused by a strain of coronavirus named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). It was first reported at the end of 2019, as a novel coronavirus causing a cluster of pneumonia cases in Wuhan city, China. It typically presents with fever and upper respiratory symptoms like cough and a sore throat [1], and can affect all ages. The severity of the disease ranges from mild (asymptomatic patients) to being a fatal disease with severe acute respiratory distress syndrome and respiratory failure [1]. The rate of hospitalization is high in the elderly population; mortality is high in older patients and those with comorbidities [2]. The blood profiles of COVID-19 patients classically show lymphopenia (lymphocyte count, 0.8×109/L) in 97% of patients [2]. The presence of lymphocytosis is indicative of other causes.
We report the case of a 49-year-old man who presented with COVID-19 and his blood tests revealed absolute lymphocytosis and further work-up confirmed chronic lymphocytic leukemia (CLL). CLL is a lymphoid neoplasm characterized by the accumulation of monoclonal lymphocytes, which are defective in function. There are many gaps in our knowledge of the COVID-19 infection pathogenesis and clinical spectrum, particularly when it is concomitant with other diseases like hematological malignancies. To the best of our knowledge, this is the first case of a patient with no significant past medical history who presented with typical COVID-19 symptoms and had concomitant CLL.
Case Report
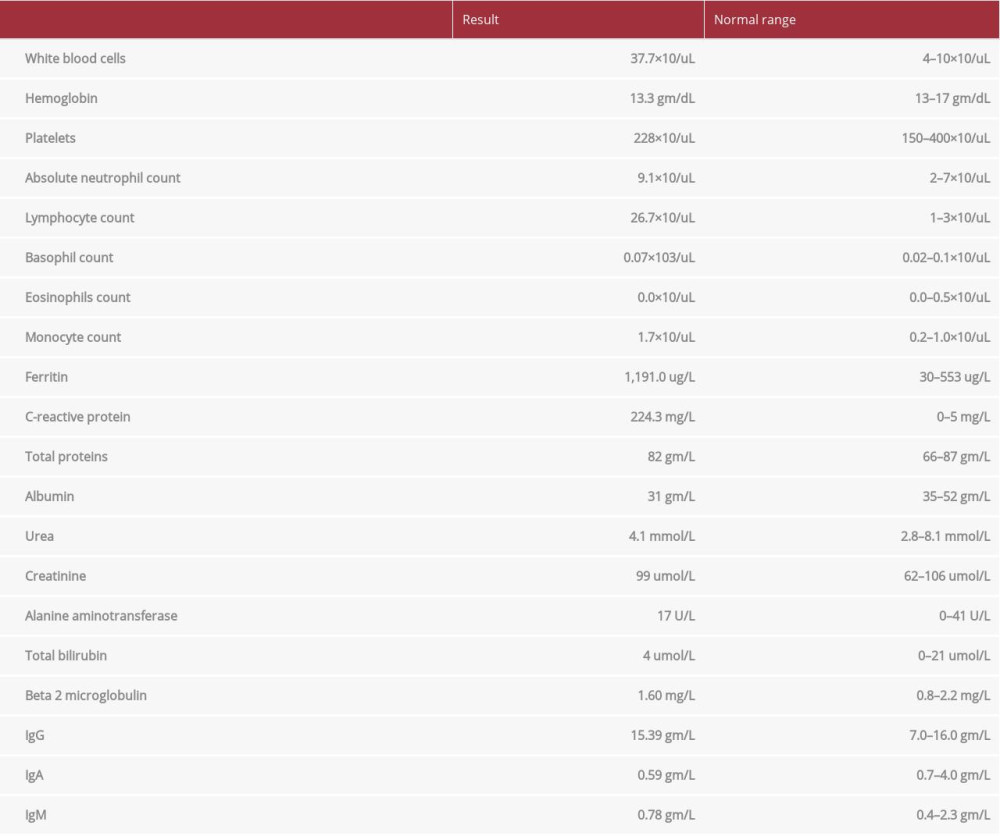
A 49-year-old man presented to the emergency department with shortness of breath, fever and body aches for 9 days. He had no significant past medical history, unremarkable family history and no previous laboratory or medical records. He reported no weight loss, no change in appetite, or night sweating. Physical examination was unremarkable except for fever reaching 39°C; there were no enlarged lymph nodes and no organomegaly. A complete blood count (CBC) showed a white blood cell (WBC) count of 37.7×103/uL (4–10×103/uL) (Table 1). The lymphocyte count was abnormally high at 26.7×103/uL. He tested positive for COVID-19 by the fully automated reverse-transcription polymerase chain reaction (RT-PCR) Cobas® 6800 (Roche, Basel, Switzerland) from nasopharyngeal and throat swabs. His ferritin level was 1,191.0 ug/L (30–553 ug/L), C-reactive protein (CRP) was 224.3 mg/L (0–5 mg/L), and his renal and liver function test results were within normal limits (Table 1). The chest X-ray showed multiple, bilateral, faint airspace shadowing representing pneumonic consolidations, suggestive of multiple atelectatic bands. His symptoms of fever, shortness of breath, and body aches were mild; he was clinically stable, did not require oxygen, and was not in acute respiratory distress. From these symptoms, it was classified as moderate COVID-19 disease according to the WHO case definition [3]. He was hospitalized for COVID-19 treatment for moderate disease, following protocol from the local institute of infectious diseases for a COVID-19 positive patient confirmed by the RT-PCR. He was prescribed oseltamivir 150 mg BID for 10 days, azithromycin 500 mg daily for 7 days, hydroxychloroquine 400 BID on the 1st day then 400 OD for 10 days, intravenous ceftriaxone 2 gm daily for 7 days, then switched to oral amoxicillin/clavulanate 625 mg 3 times per day. After 6 days, the fever and shortness of breath subsided, the body aches resolved and he was discharged home with isolation. After the initial CBC showed lymphocytosis, a peripheral blood smear was performed and the results were consistent with a lymphoproliferative disorder. There was mild normocytic normochromic anemia with a few spherocytes and increased rouleaux formation, mild neutrophilia, and marked lymphocytosis consisting mostly of small mature-looking lymphocytes with many smudge cells and platelets within normal limits. Flow cytometry was performed and the smear from the flow cytometry sample showed remarkable leukocytosis (60.0×103/uL) and lymphocytosis (51.3×103/uL), mostly small mature-looking lymphocytes with a few prolymphocytes (~2%) and many smudge cells. The flow cytometry confirmed the diagnosis of CLL; it showed a population of monotypic B cells comprising approximately 76% of the total cells, and expressing CD19, CD5, CD23, CD20 (dim), CD43, CD200 with a cytoplasmic kappa light chain restriction (dim expression). There was dim expression of FMC7 on a minority of the cells. These monotypic cells were negative for CD10, CD38, CD79b, CD103, CD25, IgM, and IgD with no significant expression of CD11c. The cells were negative for a surface light chain. There were approximately 7% T cells with a CD4: CD8 ratio of 1.4. The T cells included approximately 1% CD4/CD8 double negative cells. The natural killer cells comprised <1% of the total cells. The cells in the granulocytic gate comprised ~14% of the total cells. The fluorescence
One month after discharge, the patient came for a follow-up; the symptoms of fever, fatigue and shortness of breath were completely resolved. Currently, no specific medication is started and CLL is under observation due to the early stage (CLL stage Ria stage 1, Binet stage B).
This case report was approved by the Hamad Medical Corporation Research Center and the patient gave informed consent.
Discussion
CLL is a lymphoproliferative neoplasm that is diagnosed by meeting the 2018 International Workshop on Chronic Lymphocytic Leukemia (iwCLL) update [4]. This requires the presence of ≥5×109/L B lymphocytes in peripheral blood, sustained for at least 3 months. The clonality of the B lymphocytes needs to be confirmed by demonstrating an immunoglobulin light chain restriction using flow cytometry [4]. The main differential diagnosis for CLL is reactive lymphocytosis from infections or other types of lymphomas or leukemias. Clonality and persistence of lymphocytosis for >3 months help to differentiate CLL from other causes of lymphocytosis. The striking feature of CLL is lymphocytosis in the peripheral blood and bone marrow. They can develop other cell-line cytopathies like anemia and thrombocythemia by various mechanisms. However, unlike a COVID-19 infection, lymphopenia is not seen in CLL. In patients with COVID-19, lymphocytosis makes the diagnosis of COVID-19 more challenging, especially if the patient is not previously diagnosed with CLL [5]. Our patient initially presented with moderate COVID-19 symptoms, the lymphocyte count was high (with no previous laboratory test results to serve as a baseline), the peripheral smear showed smudge cells, and CLL was confirmed by flow cytometry. Most patients with CLL are asymptomatic and are diagnosed during routine blood work showing absolute lymphocytosis, or during evaluation for enlarged lymph nodes [6].
The lymphocyte count in CLL patients is already high; the effect of the COVID-19 infection in such a condition is unclear. In a review of 4 patients with naive CLL who were not on active treatment, 3 died and 1 had a severe COVID-19 infection. This could not apply to our patient due to the advanced age and existing comorbid conditions in these reviewed patients. However, it is reported that the lymphocyte count in patients with CLL increased 3-fold above their baseline during a COVID-19 infection, making the pathogenesis in CLL patients very different from the other patients [7]. Similarly, in the present case report the patient’s lymphocyte count doubled over 4 weeks, from 26.7×103/uL to 51.3×103/uL. This indicates that the COVID-19 infection is associated with an increase in the clonality of the B cells. Given the absence of previous records, a possible scenario is that the patient had unrecognized monoclonal B-cell lymphocytosis that was accelerated into CLL by the COVID-19 infection, as seen with other respiratory tract infections, or it could be asymptomatic CLL unmasked by the COVID-19 infection [8].
Patients with CLL have an increased risk of infections due to defective immunity (mainly humoral and cellular). They have hypogammaglobulinemia, abnormalities in T-cell subsets, defects in the complement system, and neutrophil/monocyte dys-function [9,10]. The defective cellular immunity is because these clonal cells do not participate in the normal defense against infections, and even inhibit other immune cells from appropriate immune response to infections [11]. This is evident as CLL patients have a poor response to skin antigen testing and prolonged skin-graft survival [12].
Low immunity results in making these patients susceptible to infections; most commonly respiratory infections, supporting the finding that the major immune defect is humoral in nature [13]. Having low immunity renders CLL patients especially vulnerable to COVID-19, which is primarily a droplet respiratory infection. The COVID-19 infection is usually more severe in cancer patients, especially those with hematological malignancies, and immunocompromised patients [14]. He was expected to progress to severe disease, particularly as the inflammatory markers were elevated (high CRP and ferritin). High ferritin levels are indicative of severe inflammation and the development of hemophagocytic syndrome in severe COVID-19 infection. However, the patient developed a moderate disease and was discharged after 6 days. He was on room air and did not require mechanical ventilation or oxygen.
This more indolent disease course could be explained by the fact that the COVID-19 infection, like other viral infections, is predominantly cellular and the main defect in CLL is humoral, as seen in the viral influenza infections [15]. An additional explanation could be related to a defective immune response in CLL. As a result, the lymphocytes do not respond strongly to the viral infection with excess cytokine release. This weak immune response prevents the cytokine storm, the subsequent damage and multi-organ involvement in CLL patients, acting as a protective factor against a severe COVID-19 infection. A COVID-19 infection in non-CLL patients usually shows a significant reduction in the number and functional exhaustion of lymphocytes, especially in severe disease and is associated with an increased release of cytokines [16]. Another reason for the benign course can be the age of the patient. A recent study of CLL patients with COVID-19 infection showed increased mortality due to COVID-19 infection among participants with advanced age (median 75 years), which is much more compared to our patient’s 49 years [17].
Conclusions
In summary, lymphocytosis is an unusual finding with a COVID-19 infection and its presence should raise the suspicion of additional underlying diseases. A COVID-19 infection is associated with a rise in the lymphocyte count in CLL patients, in contrast to the lymphopenia observed in non-CLL patients. The interaction between COVID-19 and CLL is not very clear, and a close follow-up and more reported cases are needed to fill the gaps in existing knowledge.
References:
1.. Wang D, Hu B, Hu C, Clinical Characteristics of 138 Hospitalized Patients with 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China: JAMA, 2020; 1323(11); 1061-69
2.. Richardson S, Hirsch JS, Narasimhan M, Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area: JAMA, 2020; 323(20); 2052-59
3.. , Clinical management of COVID-19 https://www.who.int/publications/i/item/clinical-management-of-covid-19
4.. Hallek M, Cheson BD, Catovsky D, iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLL: Blood, 2018; 131(25); 2745-60
5.. Jin XH, Zheng KI, Pan KH, COVID-19 in a patient with chronic lymphocytic leukaemia: Lancet Haematol, 2020; 7; e351-52
6.. Binet JL, Auquier A, Dighiero G, A new prognostic classification of chronic lymphocytic leukemia derived from a multivariate survival analysis: Cancer, 1981; 48(1); 198-206
7.. Paneesha S, Pratt G, Parry H, Moss P, Covid-19 infection in therapy-naive patients with B-cell chronic lymphocytic leukemia: Leuk Res, 2020; 93; 106366
8.. Anderson LA, Landgren O, Engels EA, Common community acquired infections and subsequent risk of chronic lymphocytic leukaemia: Br J Haematol, 2009; 147(4); 444-49
9.. Ravandi F, O’Brien S, Immune defects in patients with chronic lymphocytic leukemia: Cancer Immunol Immunother, 2006; 55(2); 197-209
10.. Wadhwa PD, Morrison VA, Infectious complications of chronic lymphocytic leukemia: Semin Oncol, 2006; 33(2); 240-49
11.. Orsini E, Pasquale A, Maggio R: Br J Haematol, 2004; 125(6); 720-28
12.. Miller DG, Lizardo JG, Snyderman RK, Homologous and heterologous skin transplantation in patients with lymphomatous disease: J Natl Cancer Inst, 1961; 26(3); 569-83
13.. Hamblin AD, Hamblin TJ, The immunodeficiency of chronic lymphocytic leukaemia: Br Med Bull, 2008; 87(1); 49-62
14.. Dai M, Liu D, Liu M, Patients with cancer appear more vulnerable to SARS-CoV-2: A multi-center study during the COVID-19 outbreak: Cancer, 2020; 10(6); 783-91
15.. Mcmichael AJ, Gotch FM, Noble GR, Beare PA, Cytotoxic T-cell immunity to influenza: N Engl J Med, 1983; 309(1); 13-17
16.. Diao B, Wang C, Tan Y, Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID-19): Front Immunol, 2020; 11; 827
17.. Scarfò L, Chatzikonstantinou T, Rigolin GM, COVID-19 severity and mortality in patients with chronic lymphocytic leukemia: A joint study by ERIC, the European Research Initiative on CLL, and CLL Campus: Leukemia, 2020; 34; 2354-63
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133