06 October 2020: Articles 
Peritoneal Dialysis in a Patient with Acute Kidney Injury, Thrombocytopenia, Urosepsis, and Liver Cirrhosis: A Case Report
Challenging differential diagnosis, Diagnostic / therapeutic accidents, Management of emergency care, Educational Purpose (only if useful for a systematic review or synthesis), Rare coexistence of disease or pathology
Krzysztof Cieszyński1ABDEF, Alicja E. Grzegorzewska12ADEG*DOI: 10.12659/AJCR.926226
Am J Case Rep 2020; 21:e926226
Abstract
BACKGROUND: We present the possibility of successful peritoneal dialysis (PD) treatment in acute kidney injury (AKI) patients with multiple comorbidities.
CASE REPORT: A 60-year-old woman with chronic kidney disease (CKD, stage G3b), liver cirrhosis (Child-Pugh class A score), and thrombocytopenia developed AKI due to urosepsis. Laboratory tests showed serum creatinine 430.5 µmol/L, urea 44.0 mmol/L, potassium 5.7 mmol/L, C-reactive protein 208 mg/L, procalcitonin 8 ng/mL, platelets 14×10⁹/L, hemoglobin 5.83 mmol/L, and albumin 30 g/L. Due to hemodynamic instability with profound hypotension and the potentially high bleeding risk when doing central venous catheter insertion or using anticoagulants, PD was selected as the AKI treatment. The PD catheter was implanted by the surgical method after the transfusion of platelet concentrate. Automated PD in tidal mode was implemented using 1.5% and 2.3% glucose: basic inflow volume 1200 mL and a tidal volume of 700 mL. Effective dialysis with ultrafiltration up to 1200 mL/day was achieved. The patient was discharged home in good condition. After 1 month, PD was discontinued due to the renal function returning to its pre-septic state of CKD category G3b. The PD catheter was removed 3 weeks later.
CONCLUSIONS: PD can be an effective method for AKI treatment in patients with sepsis, hemodynamic instability, thrombocytopenia, and liver cirrhosis.
Keywords: Acute Kidney Injury, Liver Cirrhosis, Peritoneal Dialysis, Sepsis, Thrombocytopenia
Background
Peritoneal dialysis (PD) can be used as the first-line dialysis treatment in most cases with end-stage renal disease, and it can also be a method of renal replacement therapy (RRT) in acute kidney injury (AKI) [1]. The great challenge for nephrologists is the need for RRT in AKI patients with liver cirrhosis. Cirrhotic patients needing emergency care for AKI have relative contraindications for extracorporeal RRT and PD [2,3].
We describe the case of a patient with 2 severe chronic diseases, liver cirrhosis and chronic kidney disease (CKD), who had a sudden deterioration of renal function due to septicemia of renal origin. Other health problems were thrombocytopenia causing a high risk of hemorrhagic diathesis and hemodynamic instability due to septic shock. Additionally, a less popular RRT method, PD, was used for the AKI treatment. We consider our patient as attractive from the clinical point of view because of the coincidence of multiple co-occurring diseases (AKI, CKD, thrombocytopenia, urosepsis, liver cirrhosis, type 2 diabetes, hypertension, and hypothyroidism), of which acute deteriorations (AKI, aggravated thrombocytopenia, and urosepsis) were successfully treated. Our case report may also have an educational value by showing that PD can be a valuable method for AKI treatment in selected patients with substantial comorbidities.
Case Report
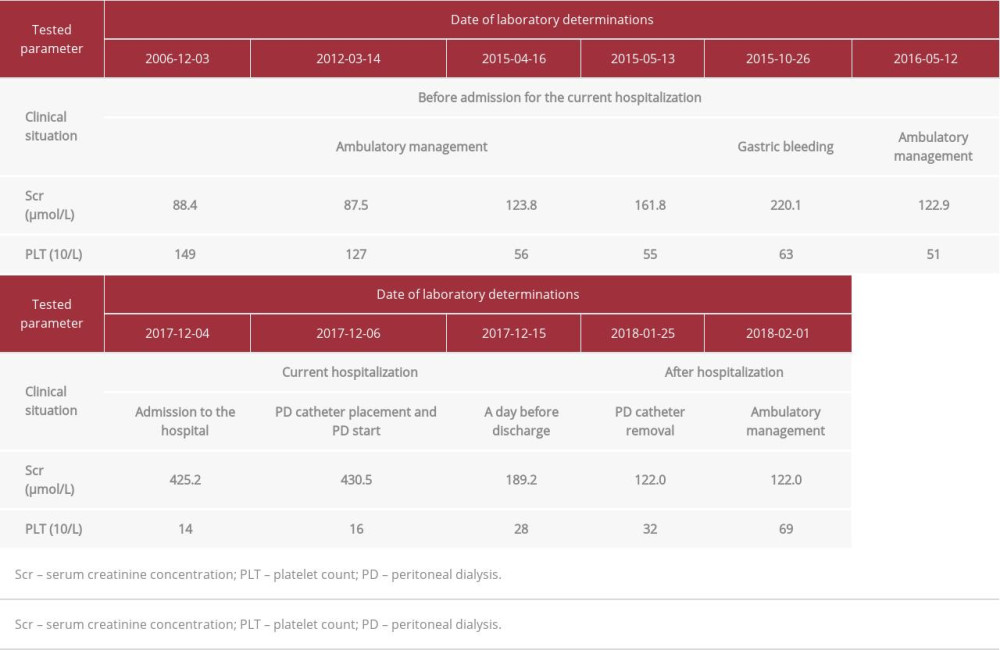
A 60-year-old woman was admitted to the hospital after experiencing syncope at work. She had blood pressure of 70/40 mmHg, tachycardia, and impaired logical contact. Her medical history included liver cirrhosis (Child-Pugh class A score, no previous decompensation events due to liver cirrhosis), thrombocytopenia, type 2 diabetes, hypertension, hypothyroidism, and CKD category G3b of unknown origin. Before admission, urine analyses did not reveal proteinuria or erythrocyturia. Hepatorenal syndrome type 2 and the abuse of painkillers, mainly nonsteroidal anti-inflammatory drugs, were suspected as contributors to CKD. In October 2015, she had gastrointestinal bleeding due to a vascular malformation detected by gastroscopy in the stomach drain region. Esophageal varices were not found. At that time, the hemoglobin level decreased to 4.59 mmol/L, and blood was transfused. Serum creatinine increased to 220.1 µmol/L (Table 1).
The patient was not a tobacco smoker and did not take psychoactive substances. Her alcohol consumption was below 30 g/week.
At the time of admission, we excluded acute ischemic cardiac disease and assessed the status of her already-known diseases. For elimination of infection, septicemia, and shock due to sepsis, C-reactive protein and procalcitonin levels were measured in serum, and urine and blood samples were cultured. We did not suspect thromboembolic diseases or disseminated intravascular coagulopathy (DIC); therefore, fibrinogen and D-dimer levels were not assessed.
Laboratory tests revealed: C-reactive protein 208 mg/L, procalcitonin 8 ng/mL, albumin 30 g/L, alanine aminotransferase 25 U/L, creatinine 430.5 µmol/L, urea 44.0 mmol/L, sodium 138 mmol/L, potassium 5.7 mmol/L, hemoglobin 5.83 mmol/L, leucocytes 14×109/L, platelets 14×109/L, total bilirubin 26.51 µmol/L, international normalized ratio (INR) 1.3, prothrombin index 78%, activated partial thromboplastin time (APTT) 38 s, urine analysis: yellow color, cloudy, specific gravidity 1.012, protein 64 mg/dL, glucose and ketone bodies not present, sediment packed with numerous leukocytes and bacteria, dysmorphic erythrocytes 6-10/high-power field, and fresh erythrocytes 2–3/high-power field. The Child-Pugh classification [4] indicated class B and the traditional Model for End-Stage Liver Disease (MELD) [5] showed a score of 24. As this score was >12, we adjusted it by incorporating the serum sodium concentration [6] and also obtained a score of 24.
An abdominal ultrasound scan showed enlarged nodular heterogeneous liver (sonographic features of cirrhosis), a trace free fluid around the liver, enlarged spleen (the bipolar dimension of 170 mm), correct position and size of both kidneys, increased echogenicity of the renal cortex, cortical cysts up to 53 mm bilaterally, and collective systems without hydronephrosis and calculi above 4 mm. Grade 1 ascites (mild ascites only detectable by ultrasound) was diagnosed [7].
Based on the results mentioned above, we diagnosed AKI stage 3 [8], liver cirrhosis with mild ascites, which progressed from class A to B by the Child-Pugh classification, and very severe thrombocytopenia (platelet count ≤20×109/L [9]). We also suspected sepsis of urinary tract origin (procalcitonin over 2 ng/mL, pyuria). Positive blood and urine cultures for
Complex treatment was initiated (see below). The most urgent medical problem was deciding what kind of RRT to apply. The patient was qualified for PD due to a risk of bleeding during surgery for obtaining vascular access for extracorporeal dialysis, and unstable blood pressure.
A straight, double-cuffed PD catheter (Tenckhoff type) was surgically implanted under local anesthesia. During the catheter insertion, about 1000 mL of amber-colored peritoneal fluid was evacuated. Platelets were transfused before and during the procedure. Bleeding to the peritoneal cavity was not detected, but an abdominal wall hematoma occurred (Figure 1). Automated PD (APD) using a tidal system was performed. We used parameters of the daily APD tidal procedure: treatment time about 10 h; retention time: 60 min; primary inflow: 1250 ml; tidal inflow: 700 ml; total volume: 6160 ml. Fluids with glucose concentration 1×1.5% 5000 ml and 1×2.3% 5000 ml were used. Ultrafiltration obtained from the procedure was 800–1200 ml/day. The abdominal wall hematoma was completely absorbed during hospitalization.
We applied intravenous therapy with antibiotics (levofloxacin, linezolid), catecholamines, 20% albumins, irradiated platelet concentrate (40 units transfused during hospitalization), and irradiated red blood cell concentrate (2 units in total). Medicines associated with comorbidities were administered orally: levothyroxine sodium (75 µg once daily, linagliptin (5 mg once daily), esomeprazole (40 mg once daily), potassium chloride (two 600 mg tablets three times daily), and carvedilol (6.25 mg twice a day).
As a result of the complex multistep treatment used, there were significant improvements in the patient’s condition and laboratory test results. Despite the substantial deterioration of medical status due to sepsis and AKI, liver function remained stable. The patient was discharged from the hospital with a serum creatinine concentration of 189.2 μmol/L. APD was continued on an ambulatory basis for 20 days. The PD catheter was removed 50 days after the start of PD, without any complications, at a serum creatinine level of 122.0 µmol/L and platelet count of 32×109/L.
Discussion
Acute kidney injury in subjects with multiorgan failure should be treated with a method individually tailored to the patient. The use of PD in an emergency is not common, possibly due to the lack of availability in a given facility or the staff’s inability to use this RRT [1,10].
Patients with liver cirrhosis, especially those with ascites, requiring urgent RRT, are a unique challenge. This population has relative contraindications for both extracorporeal RRT and PD [2,3]. Therefore, for AKI treatment in our patient, we initially discussed extracorporeal dialysis techniques and PD.
Extracorporeal methods include conventional intermittent hemodialysis (HD) and continuous RRT (CRRT), as well as slow low-efficiency dialysis (SLED) and slow low-efficiency daily dialysis (SLEDD). The advantage of extracorporeal dialysis techniques over PD is the more efficient and quicker elimination of uremic toxins, potassium, serum ammonia, and water. Therefore, life-threatening hyperkaliemia, severe hypercatabolism, or pulmonary edema require highly efficient extracorporeal techniques. However, the related risk of hypotension and bleeding limit their use in cirrhotic subjects [11,12].
Hemodynamic disorders occurring during traditional HD may be poorly tolerated in patients with hepatorenal syndrome due to concomitant systemic vasodilatation, increased cardiac output, and hypotension [3]. SLED or SLEDD appear to be acceptable RRT methods for use in hemodynamically unstable AKI subjects [13–15], and they could be performed without anticoagulation in 86% of dialysis sessions [15]. Patients with liver cirrhosis often develop acquired coagulopathy due to decreased hepatic clotting factor synthesis, increased fibrinolysis, and quantitative platelet deficiency associated with hypersplenism. These abnormalities increase the risk of bleeding during large-vessel cannulation to the access for extracorporeal RRT. The overall frequency of hemorrhage/hematoma due to image-guided central venous catheter insertion ranges from below 2% to 4% [16,17], and it is the most common early complication of this procedure. Blind catheter insertion accounts for 10% of hematomas [17]. Lethal hemorrhagic complications of central venous catheter placement were also described [18]. The data do not show that patients subjected to these procedures were at risk of bleeding due to thrombocytopenia or coagulopathy.
There are 2 main types of PD: continuous ambulatory PD (CAPD) and APD, which is cycler-assisted. APD is more effective in waste product removal, which optimizes dialysis adequacy. One of the APD modalities is tidal APD, which is the preferred treatment modality in subjects with ascites, as it allows a controlled outflow of fluid from the peritoneal cavity. Tidal PD also reduces drainage pain because the peritoneal cavity always contains part of the fill volume [19]. Ascites drainage and management improve patients’ well-being and quality of life [3,11].
Among the shortcomings of using PD in patients with liver cirrhosis and ascites is the possibility of an increased risk of peritonitis and worsening of malnutrition due to protein loss with dialysate. On the other hand, there are reports that an increase in hydrostatic pressure in the abdomen during PD therapy can slow the appearance of protein-rich ascites, leading to better nutrition [20]. In septic AKI patients without liver cirrhosis, and showing mild or moderate catabolism, the higher intensity of PD did not lower the mortality rate or improve kidney function recovery or metabolic control [21]. Therefore, a favorable effect of PD treatment might be obtained with lower dialysate volumes per session (32–38 L
Abdominal wall hematomas are seldom reported as complications of PD catheter insertion if a surgical method without trocar is used for implantation of classic catheters. In the previous studies on use of PD for AKI in patients with chronic liver diseases, no abdominal hematomas were shown [22–25], although bleedings to the peritoneal cavity with the use of trocar technique for catheter placement were demonstrated [22]. When routine methods were not applied, like the insertion of embedded PD catheters, abdominal wall hematomas were reported in from 0% [26] to 46% [27] of cases. Surgical revision was needed in 33% of subjects with subcutaneous hematomas due to implantation of embedded PD catheters [27]. However, embedded catheters are not used for AKI treatment. A simplified one-port laparoscopic technique of PD catheter placement was not found to be complicated with postoperative abdominal wall bleeding [28]. A comparison of permanent PD catheters (surgically-implanted Tenckhoff, medically-implanted Tenckhoff, and the 2 types of the Toronto Western Hospital catheters) did not reveal any differences concerning abdominal hematomas [29].
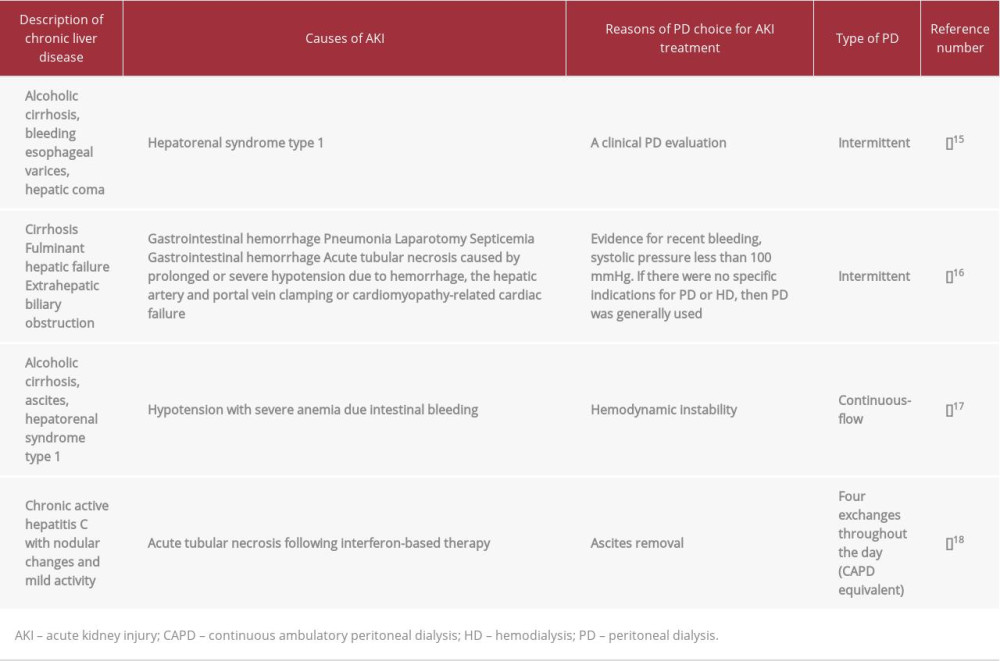
PD is already used for AKI in patients with chronic liver diseases (Table 2) [22–25]. Evidence of recent bleeding [23], hemodynamic instability [23,24], and ascites removal [25] were among the reasons for use of PD for AKI treatment. Studies evaluating use of PD in a population presenting various etiologies of end-stage renal disease and liver disease concluded that PD has the advantages of direct removal of ascitic fluid, avoidance of anticoagulation, and slow and continuous ultrafiltration during equilibration PD techniques, which causes less hemodynamic instability [30,31].
In our patient, we used PD for AKI treatment. Compared with extracorporeal RRT methods, the advantages of using PD in our patient included effective restoration and maintenance of hemodynamic stability, less risk of life-threatening bleeding during the performance of dialysis access, and removal of ascitic fluid, if needed, without additional abdominal punctures. CRRT, SLED, or SLEDD were excluded for use in our patient due to the need for good vascular access [14], the performance of which we gauged as a potential burden with thrombocytopenia-related bleeding. The main PD disadvantage in our patient would be a risk of peritonitis. Protein malnutrition was also considered. We expected that the patient’s state would improve due to the complex treatment, quickly obtaining hemo-dynamic stability and amelioration of thrombocytopenia severity. If AKI was reversible in our patient, PD treatment could be discontinued. If the patient needed permanent RRT, PD could be continued without any additional surgery for dialysis access. The initiation of RRT with PD did not preclude its conversion to other dialysis modalities if such a treatment was determined to be better given the new health conditions of our patient.
We predicted a bleeding tendency in our patient mainly from hypersplenic thrombocytopenia exacerbated by septic shock. Although we did not consider liver cirrhosis as a cause of AKI, hepatic-related abnormalities could worsen in a septic state that concomitantly occurred [11]. The patient’s prothrombin index being ≤80%–≥70% (78%) indicated severe liver fibrosis but still not cirrhosis [32]. The role played by coagulation defects in the occurrence of bleeding in liver cirrhosis is unclear [33]. Severe thrombocytopenia, like that in our patient, occurs in 1% of cirrhotic subjects [34]. A platelet count below 15×109/L can cause risk of bleeding with minimal trauma [35]. In non-cirrhotic patients with AKI, thrombocytopenia was also an indication for PD. In an 89-year-old woman with acute pyelonephritis, septic shock, atrial fibrillation, and worsening renal failure, HD had to be changed to PD due to type II heparin-induced thrombocytopenia [36]. PD was intensified to avoid installing an HD catheter in a patient with severe thrombocytopenia after methotrexate administration [37]. In gram-negative sepsis and AKI, DIC can contribute to a decrease in platelet count [38]. However, we did not find any evidence of this mechanism in our patient. We did not suspect DIC at admission or later, when the patient’s status was quickly improving.
In our patient, a substantial abdominal wall hematoma occurred due to the classical PD catheter insertion using the surgical method, but it must be remembered that she had severe thrombocytopenia. This suggested that bleeding during vein cannulation for extracorporeal RRT and anticoagulation during RRT would be risky. Eventual bleeding during surgical insertion of the PD catheter is usually less dangerous and easier to overcome than potential bleeding during large-vessel cannulation.
Our case shows that tidal PD can be an effective RRT method in cirrhotic AKI patients with hemodynamic instability during sepsis and thrombocytopenia. In the described case, a significant improvement in the patient’s condition was obtained after the complex treatment, including the use of PD. Return of renal function to the status before AKI was achieved; therefore, RRT could be withdrawn. The treatment turned out to be efficient and free from significant complications. However, it needs to be mentioned that liver cirrhosis in our patient was associated with mild ascites (never previously evacuated), no hyponatremia, a thrombotic index showing rather severe fibrosis than cirrhosis [32], and a MELD score of 24 (this score ranges from 6 to 40) [5,6].
Although urosepsis and AKI resolved and the platelet count increased, liver cirrhosis and CKD were still significant health problems in our patient. As stated in the Case Report section, hepatorenal syndrome type 2 was suspected in our patient before admission, among other contributing factors. One of the main criteria for the diagnosis of hepatorenal syndrome type 2 is serum creatinine level over 133 µmol/L in the absence of signs of parenchymal renal disease (proteinuria of over 500 mg/d or hematuria of over 0.50 red blood cells/high-power field) in a cirrhotic patient with ascites [39]. Before AKI and after recovery from AKI, our patient did not present with serum creatinine over 133 µmol/L when she was in stable clinical condition. However, the risk of hepatorenal syndrome existed in this patient: the probability of hepatorenal syndrome occur-rence was evaluated for 18% at 1 year and 39% at 5 years in nonazotemic patients with cirrhosis and ascites [40]. At present, hepatorenal syndrome can be improved by vasoconstrictors, such as terlipressin, in combination with albumin [41,42]. Terlipressin therapy reversed hepatorenal syndrome types 1 and 2 in 48% and 46% of patients, respectively [42]. Our patient could be a candidate for such a treatment in the future.
Conclusions
PD appears to be an effective method of AKI treatment in selected liver cirrhotic patients with sepsis of renal origin. Thrombocytopenia, with a high risk of hemorrhagic diathesis, and hemodynamic instability, suggest choosing PD as a method of RRT in such patients. Further research and reports of experience from other medical centers might help to select PD in similar cases. Despite this, in our opinion, PD is underused in emergency patients with multiple comorbidities.
References:
1.. Ponce D, Brabo AM, Balbi AL, Urgent start peritoneal dialysis: Curr Opin Nephrol Hypertens, 2018; 27; 478-86
2.. Howard CS, Teitelbaum I, Renal replacement therapy in patients with chronic liver disease: Semin Dial, 2005; 18; 212-16
3.. Guest S, Peritoneal dialysis in patients with cirrhosis and ascites: Adv Perit Dial, 2010; 26; 82-87
4.. Pugh RN, Murray-Lyon IM, Dawson JL, Transection of the oesophagus for bleeding esophageal varices: Br J Surg, 1973; 60; 646-49
5.. Kamath PS, Kim WR, The model for end-stage liver disease (MELD): Hepatol, 2007; 45(3); 797-805
6.. Kim WR, Biggins SW, Kremers WK, Hyponatremia and mortality among patients on the liver-transplant waiting list: N Engl J Med, 2008; 359(10); 1018-26
7.. , EASL clinical practice guidelines on the management of ascites, spontaneous bacterial peritonitis, and hepatorenal syndrome in cirrhosis: J Hepatol, 2010; 53j; 397-417
8.. Fliser D, Laville M, Covic A, A The ad-hoc working group of European Renal Best Practice (ERBP). A ERBP position statement on the Kidney Disease Improving Global Outcomes (KDIGO) Clinical Practice Guidelines on Acute Kidney Injury: Part 1: definitions, conservative management and contrast – induced nephropathy: Nephrol Dial Transplant, 2012; 27; 4263-72
9.. Sharma B, Sharma M, Majumder M, Thrombocytopenia in septic shock patients – a prospective observational study of incidence, risk factors and correlation with clinical outcome: Anaesth Intensive Care, 2007; 35(6); 874-80
10.. Wańkowicz Z, Peritoneal dialysis – the role in the integrated renal care: Pitfalls and benefits of therapy: Adv Clin Exp Med, 2003; 12; 243-49
11.. Hartleb M, Gutkowski K, Kidneys in chronic liver diseases: World J Gastroenterol, 2012; 18; 3035-49
12.. Pipili C, Polydorou A, Pantelias K, Improvement of hepatic encephalopathy by application of peritoneal dialysis in a patient with non-end-stage renal disease: Perit Dial Int, 2013; 33(2); 213-16
13.. Molina-Andújar A, Blasco M, Poch E, Role of sustained low-efficiency dialysis in the intensive care unit: Nefrologia, 2019; 39(1); 1-110
14.. Nickson C, 2019 https://litfl.com/ihd-vs-crrt-vs-sled/
15.. Kitchlu A, Adhikari N, Burns KEA, Outcomes of sustained low efficiency dialysis versus continuous renal replacement therapy in critically ill adults with acute kidney injury: A cohort study: BMC Nephrol, 2015; 16; 127
16.. Bream PR, Update on insertion and complications of central venous catheters for hemodialysis: Semin Intervent Radiol, 2016; 33(1); 31-38
17.. Sazdov D, Jovanovski Srceva M, Nikolova Todorova Z, Comparative analysis of ultrasound guided central venous catheterization compared to blind catheterization: Pril (Makedon Akad Nauk Umet Odd Med Nauki), 2017; 38(2); 107-14
18.. Bagwell CE, Salzberg AM, Sonnino RE, Haynes JH, Potentially lethal complications of central venous catheter placement: J Pediatr Surg, 2000; 35(5); 709-13
19.. Vychytil A, Hörl WH, The role of tidal peritoneal dialysis in modern practice: A european perspective: Kidney Int Suppl, 2006(103); 96-103
20.. Selgas R, Bajo MA, Del Peso G, Peritoneal dialysis in the comprehensive management of end-stage renal disease patients with liver cirrhosis and ascites: practical aspects and review of the literature: Perit Dial Int, 2008; 28; 118-22
21.. Ponce D, Brito GA, Abrão JG, Balb AL, Different prescribed doses of high-volume peritoneal dialysis and outcome of patients with acute kidney injury: Adv Perit Dial, 2011; 27; 118-24
22.. Cohen H, A clinical evaluation of peritoneal dialysis: Can Med Assoc J, 1963; 88(18); 932-38
23.. Wilkinson SP, Weston MJ, Parsons V, Williams R, Dialysis in the treatment of renal failure in patients with liver disease: Clin Nephrol, 1977; 8(1); 287-92
24.. Charen E, Dadzie K, Sheth N, Hepatorenal syndrome treated for eight months with continuous-flow peritoneal dialysis: Adv Perit Dial, 2013; 29; 38-42
25.. Fabrizi F, Aghemo A, Fogazzi GB, Acute tubular necrosis following interferon – based therapy for hepatitis C: Case study with literature review: Kidney Blood Press Res, 2013; 38; 52-60
26.. McCormick BB, Brown PA, Knoll G, use of the embedded peritoneal dialysis catheter: Experience and results from a North American Center: Kidney Int, 2006; 70(Suppl. 103); 38-43
27.. Prischl FC, Wallner M, Kalchmair H, Initial subcutaneous embedding of the peritoneal dialysis catheter – a critical appraisal of this new implantation technique: Nephrol Dial Tranplant, 1997; 12; 1661-67
28.. Harissis HV, Katsios CS, Koliousi EL, A new simplified one-port laparoscopic technique of peritoneal dialysis catheter placement with intra-abdominal fixation: Am J Surg, 2006; 192(1); 125-29
29.. Ponce PS, Pierratos A, Izatt S, Comparison of the survival and complications of three permanent peritoneal dialysis cetheters: Perit Dial Int, 1982; 2; 82-85
30.. Marcus RG, Messana J, Swartz R, Peritoneal dialysis in end-stage renal disease patients with preexisting chronic liver disease and ascites: Am J Med, 1992; 93(1); 35-40
31.. Vion VB, Sosa Barrios RH, Cabrera MC, Peritoneal dialysis (PD) in end-stage renal disease (ESRD) patients with liver cirrhosis
32.. Croquet V, Vuillemin E, Ternisien C, Prothrombin index is an indirect marker of severe liver fibrosis: Eur J Gastroenterol Hepatol, 2002; 14(10); 1133-41
33.. Senzolo M, Burra P, Cholongitas E, Burroughs AK, New insights into the coagulopathy of liver disease and liver transplantation: World J Gastroenterol, 2006; 12(48); 7725-36
34.. Afdhal N, McHutchison J, Brown R, Thrombocytopenia associated with chronic liver disease: J Hepatol, 2008; 48(6); 1000-7
35.. Gauer RL, Braun MM, Thrombocytopenia: Am Fam Physician, 2012; 85(6); 612-22
36.. Nakamura H, Sato T, Anayama M, [A case of atypical-onset heparin-induced thrombocytopenia at the initiation of hemodialysis in which it was necessary to alter the patient’s peritoneal dialysis protocol]: Nihon Toseki Igakkai Zasshi, 2016; 49(5); 363-67 [in Japanese]
37.. Aristizabal-Alzate A, Nieto-Rios JF, Ocampo-Kohn C, Successful multiple-exchange peritoneal dialysis in a patient with severe hematological toxicity by methotrexate: Case report and literature review: Braz J Nephrol, 2019; 41(3); 427-32
38.. LaMonica CR, Blackston M, Dawson RB, Cowley RA, Acute renal failure associated with the thrombocytopenia of septicemia: Adv Shock Res, 1981; 6; 75-79
39.. Gonwa TA, Wadei HM, Kidney disease in the setting of liver failure: Core curriculum 2013: Am J Kidney Dis, 2013; 62(6); 1198-212
40.. Ginès A, Escorsell A, Ginès P, Incidence, predictive factors, and prognosis of the hepatorenal syndrome in cirrhosis with ascites: Gastroenterology, 1993; 105(1); 229-36
41.. Zhang J, Rössle M, Zhou X, Terlipressin for the treatment of hepatorenal syndrome: An overview of current evidence: Curr Med Res Opin, 2019; 35(5); 859-68
42.. Nguyen-Tat M, Jäger J, Rey JW, Terlipressin and albumin combination treatment in patients with hepatorenal syndrome type 2: United European Gastroenterol J, 2019; 7(4); 529-37
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133