16 October 2020: Articles 
Spontaneous Biloma Resulting from Intrahepatic Bile Duct Perforation Coexisting with Intrahepatic Cholelithiasis and Cholangiocarcinoma: A Case Report and Literature Review
Rare coexistence of disease or pathology
Keiichi Suzuki1ABCDEF*, Takeo Hashimoto1D, Syoko Osugi1B, Naoyuki Toyota1A, Kenshi Omagari1ABC, Akihiko Tamura1DFDOI: 10.12659/AJCR.926270
Am J Case Rep 2020; 21:e926270
Abstract
BACKGROUND: Spontaneous biloma is a rare non-traumatic disease in which an extrahepatic or intrahepatic bile duct perforates spontaneously with no discernable cause. We present the details of a patient with spontaneous biloma resulting from intrahepatic bile duct perforation with concurrent intrahepatic cholelithiasis and cholangiocarcinoma.
CASE REPORT: A 74-year-old woman was admitted to our hospital with symptoms of abrupt epigastralgia, nausea, and fever. Physical examination revealed epigastric tenderness, guarding, and rebound tenderness. Laboratory test results were normal, except for elevated leukocytes, and C-reactive protein, total bilirubin, and blood urea nitrogen concentrations. Carcinoembryonic antigen and carbohydrate antigen 19-9 concentrations were also elevated. Abdominal computed tomography revealed perihepatic fluid and ascites, with common bile duct dilatation and localized cholangiectasia of B2 with areas of slight high density, which indicated an intraabdominal abscess and intrahepatic cholelithiasis. Spontaneous intrahepatic bile duct perforation was subsequently diagnosed by cholangiography via endoscopic nasobiliary drainage. Left hepatic lobectomy was performed to treat the intrahepatic cholelithiasis and spontaneous biloma. Intraoperatively, a perforation was identified at the edge of the lateral segment of the left triangular ligament, through which bile had been leaking. Histopathology revealed intraductal cholangiocellular carcinoma with intrahepatic cholangiolithiasis. The patient’s postoperative course was excellent, and she was discharged on postoperative day 16. However, cancer dissemination to the peritoneum was identified 8 months after surgery.
CONCLUSIONS: Treatment for patients with intrahepatic cholelithiasis should involve aggressive surgery because of the associated carcinogenicity. This approach reduces the risk of dissemination secondary to intrahepatic bile duct perforation.
Keywords: Abdomen, Acute, Bile Duct Neoplasms, Calculi, liver abscess, Bile Duct Diseases, Bile Ducts, Intrahepatic, Cholangiocarcinoma, Cholelithiasis
Background
Biloma is defined as a localized collection of bile with iatrogenic or traumatic causes. Spontaneous biloma is defined by extrahepatic or intrahepatic bile ducts spontaneously perfo-rating with no definable cause. This non-traumatic bile duct injury is rare in adults.
Intrahepatic cholelithiasis is defined as the presence of stones within the intrahepatic bile ducts, specifically before the confluence of the right and left hepatic ducts. Intrahepatic cholelithiasis is endemic in the Asia–Pacific region, where its prevalence can be as high as 30–50% [1]. The clinical relevance of this condition lies in its frequently progressive course, which is characterized by recurrent cholangitis, inflammatory biliary strictures, parenchymal atrophy, and secondary biliary cirrhosis. The condition has a well-known etiological relationship with cholangiocarcinoma [2]; therefore, the treatment strategy for intrahepatic cholelithiasis should be selected carefully.
We present our experience treating a patient with spontaneous biloma that resulted from intrahepatic bile duct perforation, with concurrent intrahepatic cholelithiasis and cholangio-carcinoma. In this interesting case, we suggest a treatment for intrahepatic cholelithiasis.
Case Report
A 74-year-old woman was emergently admitted to our hospital with symptoms of abrupt epigastralgia, nausea, and fever of 38.1°C. She had a history of hypertension and choledocholithiasis, which was treated 8 months previously by endoscopic sphincterotomy. Physical examination revealed epigastric tenderness, guarding, and rebound tenderness. Laboratory test results showed increased leukocytes (14 400 cells/μL) and C-reactive protein (CRP) (13.36 mg/dL) concentrations. Total bilirubin (T-Bil) and blood urea nitrogen (BUN) concentrations were slightly elevated (1.84 mg/dL and 26.9 mg/dL, respectively), and concentrations of the tumor markers carcinoembryonic antigen (CEA) and carbohydrate antigen (CA) 19-9 were also elevated (7.5 ng/mL and 144.3 U/mL, respectively). Hemoglobin, serum electrolyte, and other liver and biliary enzyme concentrations were within normal limits. Abdominal computed tomography (CT) (Figure 1A, 1B; white arrows) revealed fluid in adjacent lateral hepatic segments and a small amount of as-cites surrounding the right liver lobe. Abdominal CT and magnetic resonance cholangio-pancreatography (MRCP) revealed common bile duct (CBD) dilatation and localized cholangiectasia of B2 with slight high-density components, which suggested intrahepatic choledocholithiasis or mucus (Figure 1C, 1D and Figure 2, arrowheads). Percutaneous abscess drainage (PAD) was performed under ultrasound guidance to treat the intraabdominal abscess. Fluid obtained from the abscess was viscous and pale yellow, which suggests that it originated from bile. Endoscopic retrograde cholangio-pancreatography (ERCP) was performed to drain and decompress the dilated bile duct. There were several filling defects in the left hepatic duct and B2 with dilatation (Figure 3, white arrow) and choledocholithiasis. Intraductal ultrasonography (IDUS) revealed cholangiolithiasis and mucus at the same site (Figure 4A, 4B). We placed an indwelling endoscopic nasobiliary drain (ENBD) in segment B2 because we suspected bile duct stenosis at the confluence of segments B2 and B3. Brush cytology diagnosed class II changes. Cholangiography performed via the ENBD tube showed bile leaking from the periphery of segment B2 outside the liver (Figure 5, white arrow).
The patient was temporarily discharged from our hospital because drainage from the PAD tube decreased markedly after placing the indwelling ENBD drain. The ENBD drain was subsequently changed to an endoscopic retrograde biliary drain (ERBD). Left hepatic lobectomy was performed on day 62 after onset, with a pre-operative diagnosis of intrahepatic cholelithiasis with bile duct perforation and spontaneous biloma. The intraoperative findings revealed no residual abscess in the abdomen. However, a tiny hole with bile leakage was found at the edge of the lateral segment when the left triangular ligament was detached (Figure 6), which was identified as the bile duct perforation point. The histopathological diagnosis was intraductal cholangiocellular carcinoma with intrahepatic cholangiolithiasis. The carcinoma measured 45×15 mm and was diagnosed as periductal-infiltrating type, stage T1a (microinvasion without hepatic parenchymal invasion) N0M0, stage IA according to the Union for International Cancer Control staging system (Figure 7A–7D). The patient’s postoperative course was excellent and she was discharged on postoperative day 16. She was followed up at the outpatient clinic until peritoneal dissemination was identified 8 months after surgery.
Discussion
To our knowledge, this is the first reported case of spontaneous biloma resulting from intrahepatic bile duct perforation with concurrent intrahepatic cholelithiasis and cholangiocarcinoma. This report is important because we discuss treatment for patients with intrahepatic cholangiolithiasis to avoid a negative outcome.
The first case of biloma was described by Whipple in 1898, but the term “biloma” was coined by Gould et al. in 1979 to describe extrahepatic post-traumatic bile collection [3]. Currently, biloma is defined as an encapsulated collection of bile located in the abdomen. Biloma can be located either intra- or extrahepatically and may occur spontaneously or secondary to a traumatic or iatrogenic lesion in the biliary system. The most common causes of iatrogenic biloma are surgery-related, and injury may occur during cholecystectomy, diagnostic and therapeutic procedures (e.g., percutaneous transhepatic cholangiography, liver biopsy, biliary drainage procedures, transcatheter arterial embolization, and ablation), and ERCP. Iatrogenic and traumatic bilomas are common, while spontaneous biloma is rare. Most bile collections are subphrenic and subhepatic; subcapsular bilomas are less common and have been reported following surgery and trauma [4–6].
The most frequent cause of spontaneous biloma is choledocholithiasis [5,7]. Less commonly reported causes are malignant biliary tree tumors, acute cholecystitis, hepatic infarction and abscess, obstructive jaundice, and tuberculosis [8,9]. The precise mechanism underlying spontaneous biloma development remains unclear. The suggested contributing factors are increasing intraductal pressure caused by an obstruction that results from a stone, tumor, or sphincter of Oddi spasm. Additionally, a vulnerable bile duct wall due to necrosis resulting from a confinement stone, cyst formation, bile duct diver-ticulum, and focal liver infarction are also a possible causes of biloma formation [5,7].
Intrahepatic bile duct ruptures occur in the left lobe of the liver in more than 80% of the reported cases. This may be because the left lobe parenchyma is thinner than in the right lobe; therefore, the bile duct directly subjacent to the hepatic capsule is susceptible to rupture following a sharp intraductal pressure increase related to intrahepatic cholelithiasis or intrahepatic cholangiocarcinoma (iCCA) [10,11]. The other reason for the high frequency of intrahepatic bile duct ruptures in the left lobe is the presence of an aberrant bile duct in the left triangular ligament [12]. Gao et al. reported that 80–90% of 92 autopsies had aberrant bile ducts in the left triangular ligament [13]. This aberrant bile duct is generally fragile and can rupture easily; therefore, the left triangular ligament is potentially the most common site of rupture.
Spontaneous reabsorption of bilomas ≥4 cm in diameter is rare. The presence of a biloma may cause bile peritonitis or lead to severe complications (e.g., sepsis, abscess, pancreatitis, respiratory failure, gastrointestinal bleeding, necrotizing fasciitis, pulmonary embolism, and myocardial infarction); therefore, prompt treatment is recommended. If there is no communication between the biloma and the biliary system and no concomitant local disease, percutaneous drainage and antibiotic therapy is sufficient [14]. In the remaining cases, specific procedures, including surgery, are required [11,15–19]. The mainstays of treating a spontaneous biloma are abdominal drainage and decompressing the biliary tree by ERCP with endoscopic sphincterotomy.
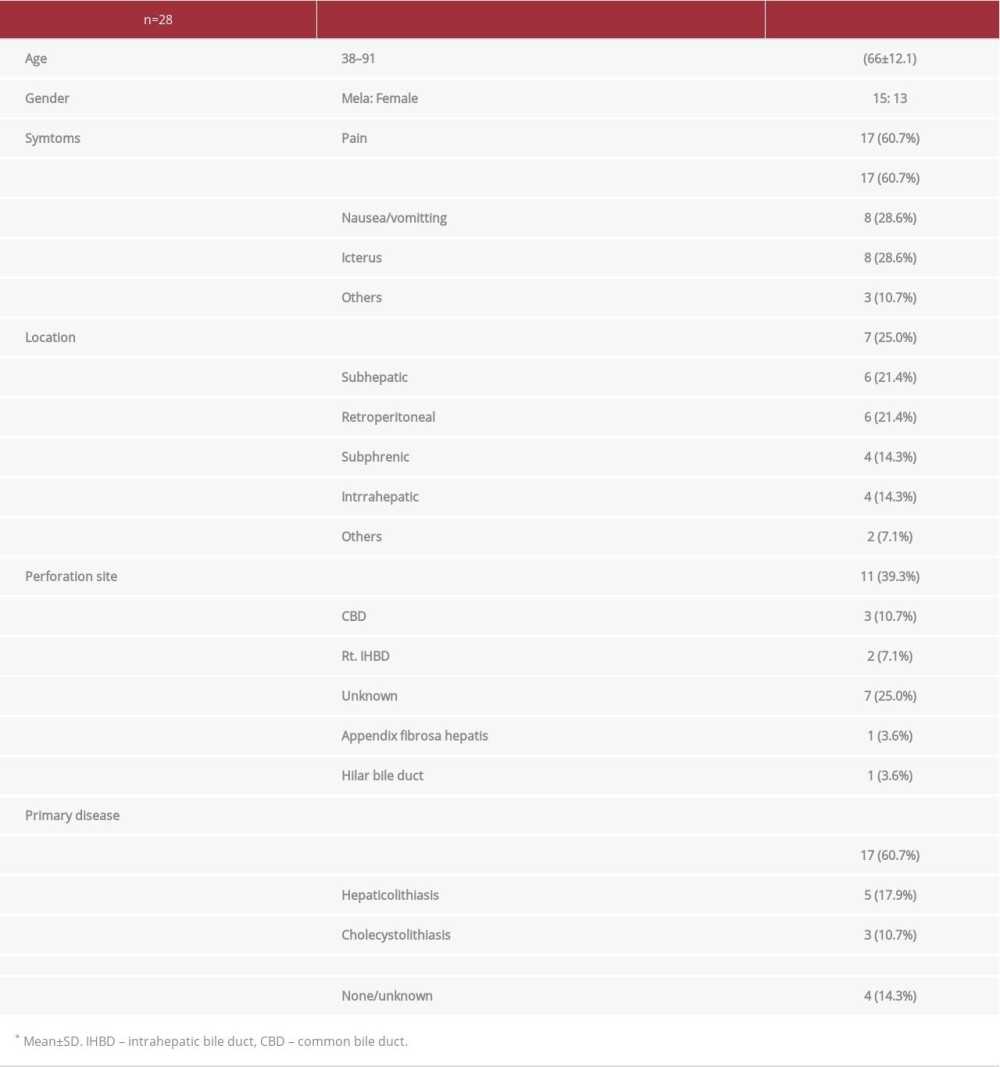
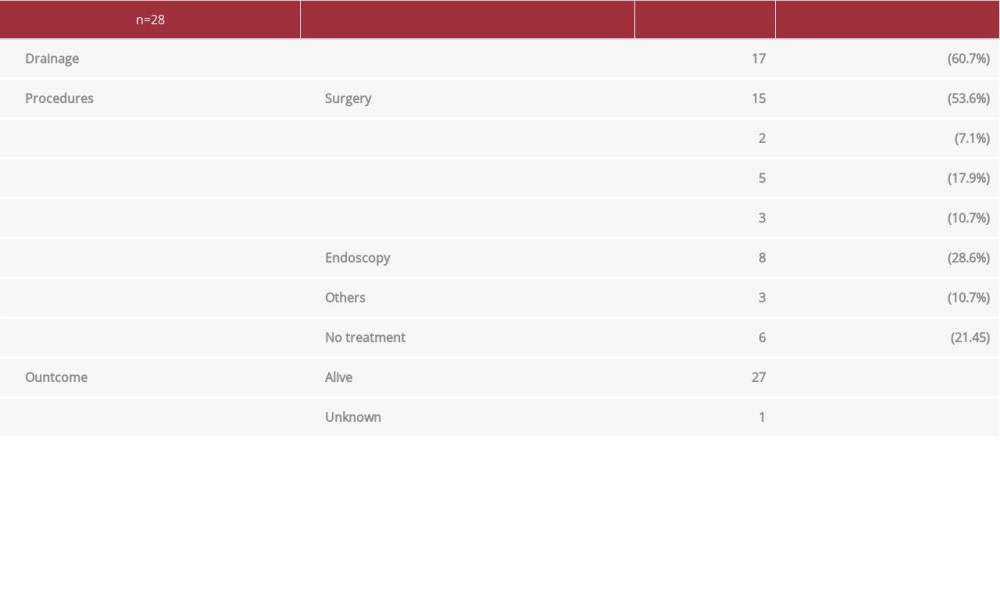
A search of case reports in the MEDLINE database from 2001 to 2019 using the term “spontaneous biloma” identified 28 adult patients (from 19 authors) with a spontaneous biloma [5,8,9,11,15–28], including our case (Table 1). The age at onset ranged from 28 to 91 years (mean±standard deviation: 66.7±12.1 years). Fifteen patients were men, and 13 were women. There were no specific symptoms, but the patients had common symptoms such as abdominal pain, fever, nausea, vomiting, and jaundice. The most common site of biloma formation was subcapsular (25.0%) followed by subhepatic (21.4%). The most frequent perforation site was the left hepatic duct (39.3%), whereas the right hepatic duct accounted for only 7.1% of perforations, which was similar to previous reports. The perforation site was unknown in 25.0% of the cases. The most common primary disease was benign disease, such as cholelithiasis (21/28 cases, 75.0%), whereas obstruction because of a malignant tumor occurred in 14.3% of cases. Most patients with spontaneous biloma required surgery (53.6%), whereas 21.4% of the patients received only follow-up observation. Additionally, 8.6% of the cases underwent endoscopic interventions such as ERCP. The outcomes were extremely good, in that all cases recovered (Table 2).
Intrahepatic cholelithiasis is defined as a gallstone disorder in the intrahepatic bile ducts. Patients with intrahepatic cholelithiasis typically present with concurrent biliary stasis, cholangitis, strictures, and bacterial infections, which subsequently lead to prolonged inflammation and biliary injury, which triggers malignant cholangiocyte growth in some cases. Long-term intrahepatic cholelithiasis can cause secondary biliary stricture, liver cirrhosis, and even cholangiocarcinoma. The indications for hepatectomy in intrahepatic cholelithiasis are: (i) unilobar intrahepatic cholelithiasis, particularly left-sided; (ii) atrophy, fibrosis, and multiple abscesses secondary to cholangitis; (iii) suspicion of concomitant intrahepatic cholangiocarcinoma; and (iv) multiple intrahepatic stones with biliary strictures that cannot be treated percutaneously or endoscopically [29].
iCCA is the second most common primary hepatic tumor following hepatocellular carcinoma. iCCA is a primary adenocarcinoma of the liver arising from the intrahepatic bile ducts [30], and is estimated to account for 5–15% of primary liver cancers [31]. No specific etiological factors have been identified for iCCA, and established risk factors include hepatitis B virus- and hepatitis C virus-related liver diseases [32,33], primary sclerosing cholangitis [34], intrahepatic cholelithiasis [35], choledochal cysts [36], and exposure to certain toxins. In East Asian countries, intrahepatic cholelithiasis and biliary parasitic infection are more common risk factors. Stones anywhere in the biliary tree are generally associated with increased iCCA risk, and the risk increases substantially with intrahepatic cholelithiasis, where it is estimated that 10% of patients develop iCCA [37]. Cholecystolithiasis and choledocholithiasis were associated with increased intrahepatic cholangiocarcinogenesis, with odds ratios of 4.0 and 23.97, respectively, in a large-scale Danish case-control study [38]. Because the symptoms and signs of intrahepatic cholelithiasis-associated iCCA (HL-iCCA) are vague and nonspecific, most patients are treated at an advanced stage and have a poor prognosis. However, recently, aggressive surgical treatment has improved long-term survival in select patients with HL-iCCA.
The present case is the first reported case in which spontaneous intrahepatic bile duct rupture resulted from iCCA with concurrent intrahepatic cholangiolithiasis. Consequently, our patient developed recurrence as peritoneal dissemination. The first choice of treatment for intrahepatic cholelithiasis without biliary stricture is extracting the stones under radiological (percutaneous transhepatic cholangiography with or without lithotripsy or access through a T-tube) or endoscopic (ERCP with or without lithotripsy) guidance. However, when complete extraction cannot be accomplished by non-surgical treatment, or when patients have biliary stricture, surgery should not be delayed because the ultimate treatment goal for intrahepatic cholangiolithiasis is to exclude the possibility of cholangiocarcinoma, which is a possible cause of bile duct rupture. Additionally, even with incidental intrahepatic cholangiolithiasis, hepatectomy should be actively considered when there is high suspicion of intrahepatic malignancy.
Conclusions
In conclusion, surgery should not be delayed in patients with intrahepatic cholelithiasis because of the carcinogenic concern and to avoid the risk of dissemination that results from intrahepatic bile duct perforation.
Figures
References:
1.. Catena M, Aldrighetti L, Finazzi R, Treatment of non-endemic hepatolithiasis in a Western country. The role of hepatic resection: Ann R Coll Surg Engl, 2006; 88(4); 383-89
2.. Koga A, Ichimiya H, Yamaguchi K, Hepatolithiasis associated with cholangiocarcinoma. Possible etiologic significance: Cancer, 1985; 55(12); 2826-29
3.. Gould L, Patel A, Ultrasound detection of extrahepatic encapsulated bile: “Biloma”: Am J Roentgenol, 1979; 132(6); 1014-15
4.. Dupas JL, Mancheron H, Sevenet F, Hepatic subcapsular biloma. An unusual complication of endoscopic retrograde cholangiopancreatography: Gastroenterology, 1988; 94(5 Pt 1); 1225-27
5.. Akhtar MA, Bandyopadhyay D, Montgomery HD, Mahomed A, Spontaneous idiopathic subcapsular biloma: J Hepatobiliary Pancreat Surg, 2007; 14(6); 579-81
6.. Braithwaite BM, Cabanilla LT, Lilly M, Hepatic subcapsular biloma: A rare complication of laparoscopic cholecystectomy and common bile duct exploration: Curr Surg, 2003; 60(2); 196-98
7.. Fujiwara H, Yamamoto M, Takahashi M, Spontaneous rupture of an intrahepatic bile duct with biloma treated by percutaneous drainage and endoscopic sphincterotomy: Am J Gastroenterol, 1998; 93(11); 2282-84
8.. Lee JH, Suh JI, A case of infected biloma due to spontaneous intrahepatic biliary rupture: Korean J Intern Med, 2007; 22(3); 220-24
9.. Bas G, Okan I, Sahin M, Spontaneous biloma managed with endoscopic retrograde cholangiopancreatography and percutaneous drainage: A case report: J Med Case Rep, 2011; 5; 3
10.. Smith W, Rupture of an intrahepatic bile duct with fatal peritonitis: Ann Surg, 1926; 83(1); 55-68
11.. Kang SB, Han HS, Min SK, Lee HK, Nontraumatic perforation of the bile duct in adults: Arch Surg, 2004; 139(10); 1083-87
12.. Champetier J, Davin JL, Letoublon C, Aberrant biliary ducts (vasa aberrantia): Surgical implications: Anatomia Clinica, 1982; 4(2); 137-45
13.. Gao XH, Roberts A, The left triangular ligament of the liver and the structures in its free edge (appendix fibrosa hepatis) in Chinese and Canadian cadavers: Am Surg, 1986; 52(5); 246-52
14.. Tipaldi MA, Rossi M, Pisano A, Percutaneous treatment of intrahepatic biloma using a ureteral double J stent: Cardiovasc Intervent Radiol, 2020; 43(4); 662-64
15.. Yasar NF, Yasar B, Kebapci M, Spontaneous common bile duct perforation due to chronic pancreatitis, presenting as a huge cystic retroperitoneal mass: A case report: Cases J, 2009; 2; 6273
16.. Blake-Siemsen JC, Kortright-Farias M, [Spontaneous retroperitoneal biloma: A case report]: Cir Cir, 2017; 85(6); 552-56 [in Spanish]
17.. Georgiou GK, Tsili A, Batistatou A, Spontaneous biloma due to an intrahepatic cholangiocarcinoma: An extremely rare case report with long term survival and literature review: Ann Med Surg (Lond), 2017; 14; 36-39
18.. Ishii K, Matsuo K, Seki H, Retroperitoneal biloma due to spontaneous perforation of the left hepatic duct: Am J Case Rep, 2016; 17; 264-67
19.. Mushtaque M, Farooq Mir M, Nazir P, Spontaneous hepatic subcapsular biloma: Report of three cases with review of the literature: Turk J Gastroenterol, 2012; 23(3); 284-89
20.. Hsieh TJ, Chiang IC, Kuo KK, Spontaneous retroperitoneal biloma: A case report: Kaohsiung J Med Sci, 2002; 18(8); 412-16
21.. Brady RR, McAteer E, Weir CD, Biliscrotum and retroperitoneal biloma: Spontaneous rupture of the biliary system presenting as an incarcerated inguinal hernia: Ulster Med J, 2006; 75(1); 85-87
22.. Ahn YH, Kim TH, Yang BJ, [A case of spontaneous biloma complicated with choledocholithiasis and chronic cholecystitis]: Korean J Gastroenterol, 2005; 46(2); 133-36 [in Ko]
23.. Trivedi PJ, Gupta P, Phillips-Hughes J, Ellis A, Biloma: An unusual complication in a patient with pancreatic cancer: World J Gastroenterol, 2009; 15(41); 5218-20
24.. Takahashi K, Okabe Y, Orino A, Spontaneous rupture of a biliary diverticulum in the distal common bile duct, with formation of a retroperitoneal biloma: Gastrointest Endosc, 2005; 61(6); 783-87
25.. Della Valle V, Eshja E, Bassi EM, Spontaneous biloma: A case report: J Ultrasound, 2015; 18(3); 293-96
26.. Chao CT, Sclerotherapy as a palliative treatment for spontaneous huge biloma: Intern Emerg Med, 2014; 9(5); 597-98
27.. Gomez-Torres GA, Rodriguez-Navarro FM, Lopez-Lizarraga CR, Acute abdomen secondary to a spontaneous perforation of the biliary tract, a rare complication of choledocholithiasis: Int J Surg Case Rep, 2017; 41; 255-58
28.. Tana C, D’Alessandro P, Tartaro A, Tana M, Mezzetti A, Schiavone C, Sonographic assessment of a suspected biloma: A case report and review of the literature: World J Radiol, 2013; 5(5); 220-25
29.. Sakpal SV, Babel N, Chamberlain RS, Surgical management of hepatolithiasis: HPB (Oxford), 2009; 11(3); 194-202
30.. Bridgewater J, Galle PR, Khan SA, Guidelines for the diagnosis and management of intrahepatic cholangiocarcinoma: J Hepatol, 2014; 60(6); 1268-89
31.. Shaib Y, El-Serag HB, The epidemiology of cholangiocarcinoma: Semin Liver Dis, 2004; 24(2); 115-25
32.. El-Serag HB, Engels EA, Landgren O, Risk of hepatobiliary and pancreatic cancers after hepatitis C virus infection: A population-based study of U.S. veterans: Hepatology, 2009; 49(1); 116-23
33.. Lee CH, Chang CJ, Lin YJ, Viral hepatitis-associated intrahepatic cholangiocarcinoma shares common disease processes with hepatocellular carcinoma: Br J Cancer, 2009; 100(11); 1765-70
34.. Lindor KD, Characteristics of primary sclerosing cholangitis in the USA: Hepatol Res, 2007; 37(Suppl. 3); S474-77
35.. Nakanuma Y, Sato Y, Harada K, Pathological classification of intrahepatic cholangiocarcinoma based on a new concept: World J Hepatol, 2010; 2(12); 419-27
36.. Tyson GL, El-Serag HB, Risk factors for cholangiocarcinoma: Hepatology, 2011; 54(1); 173-84
37.. Kim HJ, Kim JS, Joo MK, Hepatolithiasis and intrahepatic cholangio-carcinoma: A review: World J Gastroenterol, 2015; 21(48); 13418-31
38.. Welzel TM, Mellemkjaer L, Gloria G, Risk factors for intrahepatic cholangiocarcinoma in a low-risk population: A nationwide case-control study: Int J Cancer, 2007; 120(3); 638-41
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133