24 November 2020: Articles 
A 45-Year-Old Man with Scleroderma Renal Crisis Associated with a History of Systemic Sclerosis Sine Scleroderma
Challenging differential diagnosis, Unusual setting of medical care, Rare disease
Panupong Hansrivijit1ABCDEF*, Kelechi F. Omeonu1BCDEF, Halimat O. Lawal1BCD, Mounika Gangireddy1BCD, Kinjal P. Gadhiya1BCD, Ravinder S. Dhatt1BCDDOI: 10.12659/AJCR.927030
Am J Case Rep 2020; 21:e927030
Abstract
BACKGROUND: The diagnosis of systemic sclerosis sine scleroderma (ssSSc) with renal crisis is difficult because of its unusual presentation and rarity.
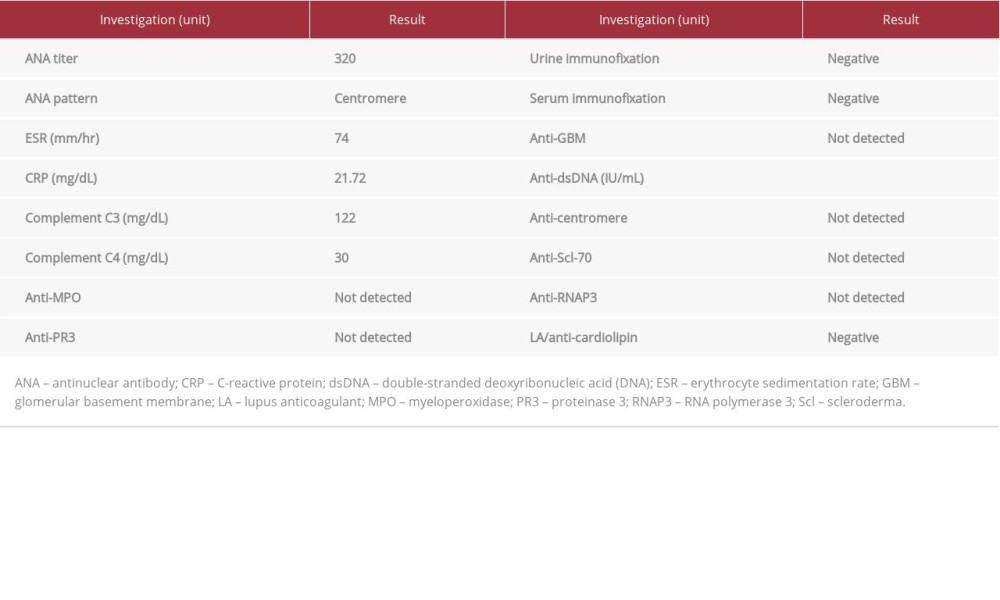
CASE REPORT: A 45-year-old man presented to the Emergency Department with worsening nausea, vomiting, and exertional dyspnea for 3 weeks. Initial examination showed blood pressure 182/108 mmHg without skin thickening or other skin manifestations. Laboratory investigations showed serum creatinine level 21.73 mg/dL and diffuse airspace opacities on chest radiography. He was admitted to the intensive care unit and started on emergent hemodialysis. He was anemic and became gradually hypoxic, requiring supplemental oxygen. Computed tomography of the chest showed bilateral infiltrates. Antinuclear antibodies (ANA) were positive for centromere pattern with titer of 320. Antineutrophil cytoplasmic antibodies and antiglomerular basement membrane antibodies were negative. He was started on therapeutic plasmapheresis (TP) and captopril, which resulted in significant improvement of respiratory symptoms. The kidney biopsy revealed thrombotic microangiopathy. Anticentromere, anti-Scl-70, and antiribonucleic acid polymerase III antibodies, drawn after 4 sessions of TP, were not detected.
CONCLUSIONS: Here we report a rare case of ssSSc with renal crisis in a patient who presented with acute renal failure requiring hemodialysis and suspected pulmonary hemorrhage. Clinical improvement was achieved by TP and angiotensin-converting enzyme inhibitor. The diagnosis of ssSSc was difficult and required an ANA pattern and kidney biopsy.
Keywords: Acute Kidney Injury, Scleroderma, Diffuse, Scleroderma, Limited, systemic vasculitis, Thrombotic Microangiopathies, Angiotensin-Converting Enzyme Inhibitors, Kidney, Scleroderma, Localized, Scleroderma, Systemic
Background
Systemic sclerosis (SSc) can be classified into limited systemic sclerosis (lcSSc), diffuse systemic sclerosis (dsSSc), and systemic sclerosis sine scleroderma (ssSSc), depending on the extension of skin involvement [1]. In general, lcSSc refers to patients with fibrosis of the skin in the acral parts of the body, such as the face and distal extremities. It carries the best prognosis, with 10-year survival over 90% [2]. Patients with dsSSc have poorer prognosis due to rapid progression of skin thickening and visceral involvement, with a 10-year survival rate of 65-82% [2]. The heart, lungs, liver, gastrointestinal tract, and kidneys, as well as central or peripheral nervous system, can be affected. ssSSc is defined by isolated organ involvement in the absence of skin fibrosis. It is a rare form, accounting for less than 5% of SSc [1].
Renal involvement in ssSSc can present in 2 different forms: scleroderma renal crisis (SRC) and chronic form. SRC is characterized by acute-onset hypertension and rapid progression of acute kidney injury (AKI). It is a serious complication of SSc and carries significant morbidity and mortality [3]. There are increasing numbers of reported patients with renal crisis without skin involvement in the literature [4–7]. In the absence of skin manifestations and autoantibodies, ssSSc with renal crisis is difficult to diagnose on initial presentation. Here, we present a case-based review of the literature of ssSSc with renal crisis and a patient who presented with a clinical picture of pulmonary-renal syndrome. The aim of this article is to remind clinicians of ssSSc when considering the differential diagnoses of pulmonary-renal syndrome.
Case Report
A 45-year-old man with a past medical history of essential hypertension for 3 years, not requiring any medications, presented to the Emergency Department with worsening nausea and vomiting for 3 weeks. The patient stated feeling nauseous every day, and vomited after drinking juices or sodas. He had been tolerating solid meals without complaints. Additionally, the patient had progressive exertional dyspnea when walking around the house or walking upstairs. He reported orthopnea and bilateral pedal edema. He denied smoking, alcohol use, or illicit drug use. Family history was notable for systemic lupus erythematosus in his aunt, and diabetes in his mother. Initial vital signs showed body temperature 36.6°C, heart rate 95 beats/min, respiratory rate 19 breaths/min, blood pressure 182/108 mmHg, and oxygen saturation 99% on room air. Physical examination was significant for mild jugular venous dilation, fine crackles in both lungs, and 1+ pitting edema bilaterally. The skin appeared normal with modified Rodnan score [8] of zero without nail changes, sclerodactyly, telangiectasia, or Raynaud’s phenomena.
Complete blood count showed white blood cell count 16 700/μL, hemoglobin 9.2 g/dL, hematocrit 26.5%, platelet count 194 000/μL. Comprehensive metabolic panel showed serum sodium 117 mEq/L, serum potassium 3.3 mEq/L, serum chloride 68 mEq/L, serum bicarbonate 17.7 mEq/L, serum creatinine 21.73 mg/dL (repeated), blood urea nitrogen (BUN) 169 mg/dL, serum magnesium 2.1 mg/dL, serum calcium 7.9 mg/dL, serum phosphate 11.7 mg/dL, aspartate aminotransferase 23 U/L, alanine aminotransferase 20 U/L, serum lipase 8 U/L, B-type natriuretic peptide 9016 pg/mL, troponin I 0.54 ng/mL, lactate dehydrogenase 536 U/L. Urinalysis showed clear, light-yellow urine, specific gravity 1.006, pH 6.0, 1+ occult blood, 2+ protein, urine white blood cells 0–5, urine red blood cell 0–5. Urine drug screen was unremarkable. Chest radiograph showed diffuse airspace opacities (Figure 1). Serum procalcitonin was elevated at 3.22 ng/mL. Real-time polymerase chain reaction (RT-PCR) for severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) from a nasopharyngeal swab was negative. Electrocardiography revealed no abnormalities. An ultrasono-graph of the kidneys showed normal sizes (right 9.6×4.9×5.2 cm; left 8.9×3.6×4.3 cm) with bilateral increased echogenicity.
He was admitted to the intensive care unit because of hypertensive emergency with AKI for blood pressure control with nitroglycerin infusion. He remained anuric without improvement in serum creatinine level and BUN despite adequate intravenous fluid hydration. Thus, the patient was started on hemodialysis. Intravenous antibiotics were also initiated to cover community-acquired pneumonia. His echocardiography showed moderate concentric left ventricular hypertrophy with an ejection fraction 55–60%. No regional wall motion abnormality or valvular dysfunction was seen. He underwent 5 sessions of hemodialysis, each with negative ultrafiltration and was deemed hemodialysis dependent by the nephrologist. His hemodialysis was resumed 3 times a week. His blood pressure was difficult to control with 5 different oral medications. His oxygenation, however, did not improve with treatment. Intravenous antibiotics were escalated to cover hospital-acquired pneumonia, although the blood cultures showed no growth. The patient became gradually hypoxic with increased oxygen requirement from room air to 6 liters via nasal cannula. Computed tomography of the chest showed bilateral pulmonary infiltrates, most notable in the mid- to upper lung fields without evidence of acute pulmonary embolism (Figure 2). The need for bronchos-copy was discussed; however, the patient did not consent to proceed because of its risk of intubation. His hemoglobin level dropped from 9.2 g/dL to 6.5 g/dL over a few days without overt external bleeding. Peripheral blood smear revealed 1+schistocytosis, 1+poikilocytosis, 2+polychromasia, 2+anisocytosis and 2+hypochromasia. He was transfused with a total of 2 units of packed red blood cells. A repeated RT-PCR for SARS-CoV-2 was negative for the second time.
Table 1 summarizes the findings of rheumatological investigations. Antinuclear antibody (ANA) staining at a titer of 320 is notable for centromere pattern. Antineutrophil cytoplasmic antibodies were negative. Human immunodeficiency virus-1 and -2 antigen/antibody, hepatitis B virus surface antigen/ core antibody, and hepatis C virus antibody were all negative.
The patient underwent a kidney biopsy that revealed severe arterial intimal fibrosis, mucoid intimal edema, intraluminal thrombi, arteriolar hyalinosis, moderate tubular atrophy, and interstitial fibrosis. Only 1 glomerulus was globally sclerotic. Other glomeruli displayed ischemic changes and mesangiolysis with focal sclerosing features. There was no evidence of fibrinoid necrosis or acute/cellular crescent formation. Immunofluorescent staining for immunoglobulin (Ig)G, IgM, IgA, C3, C1q, albumin, fibrinogen, and kappa and lambda light chains were all negative. Overall, the final result of kidney biopsy suggested a pathological picture of thrombotic microangiopathy (TMA) commonly seen in accelerated hypertension, SRC, antiphospholipid syndrome, and drug-induced etiologies (Figure 3).
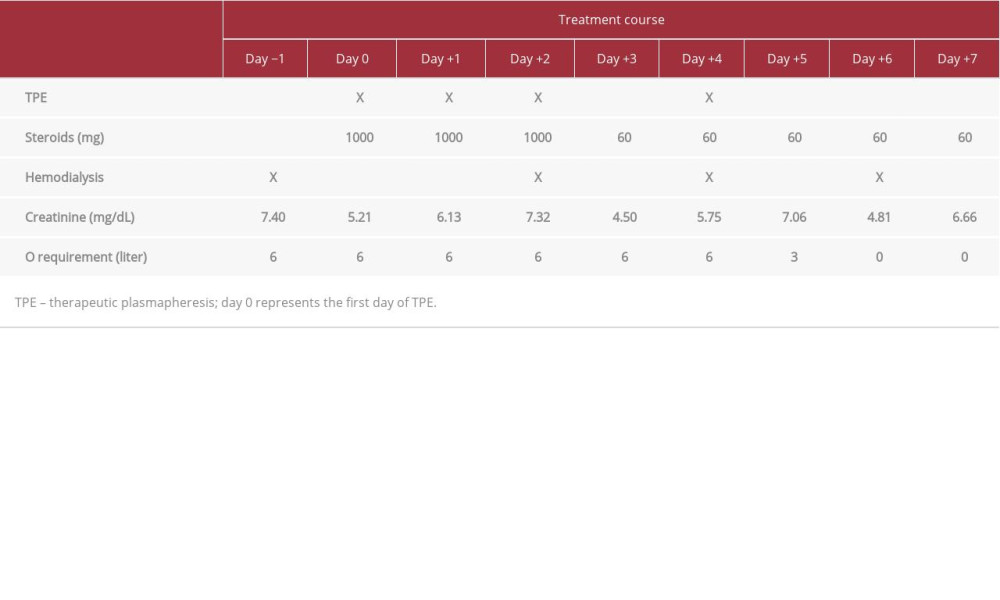
Given the suspicion of diffuse alveolar hemorrhage and glomerulonephritis, starting therapeutic plasmapheresis (TP) and high-dose steroids were justified even when the vasculitis work-up and the kidney biopsy were pending. He received a total of 4 sessions of TP on days 0, +1, +2, and +4, where day 0 represented the first session. Anticentromere B, anti-Scl70, and antiribonucleic acid polymerase III (RNAP3) antibodies were collected after the patient completed 4 sessions of TP. Methylprednisolone was given on the first session of TP at 1000 mg for 3 days, followed by 60 mg of prednisone for 7 days, then tapered by 10 mg every week until discontinued. Hemodialysis was continued on a Tuesday, Thursday, and Saturday schedule throughout the treatment course. Captopril was continued along with 4 other antihypertensive agents.
The patient completed a 14-day course of intravenous antibiotics for possible hospital-acquired pneumonia, although all cultures had no growth. Table 2 summarizes the time line of treatment and clinical response in this patient. Oxygen requirement significantly improved with treatment. The patient did not require oxygen support on day +6 onward. The chest radiography on discharge showed significant improvement of bilateral infiltrates. Serum creatinine levels remained stable throughout the course of treatment. Unfortunately, he was hemodialysis dependent and was discharged with an outpatient hemodialysis schedule 3 times a week.
Discussion
Here, we presented a suspected case of ssSSc with renal crisis in a patient who presented with 1) gastrointestinal symptoms (nausea, vomiting); 2) accelerated, difficult-to-control hypertension; 3) acute renal failure requiring hemodialysis; and 4) suspected pulmonary hemorrhage from computed tomography imaging and acute anemia. Rheumatological investigations revealed a markedly positive centromere pattern on ANA staining (titer of 320). The kidney biopsy was notable for TMA. His clinical improvement was achieved by adding captopril and TP. The diagnosis of this case was difficult because of certain limitations. First, the diagnosis of diffuse alveolar hemorrhage was not confirmed by bronchoscopy or lung biopsy because of patient’s dissent given a risk of intubation. Second, specific autoantibodies were negative. It is possible that these specific autoantibodies were falsely negative as the patient underwent 4 sessions of TP before blood draw. However, as concluded by Hunzelmann et al., negative specific autoantibodies should not exclude ssSSc if there is high clinical suspicion and ANA positivity [9]. Third, although 1+schistocytosis was noted, a complete microangiopathic hemolytic anemia picture was not seen because of lack of thrombocytopenia. Fourth, follow-up duration as skin manifestations may occur several months after internal organ involvement. Moreover, one could argue that the findings of TMA could be secondary to accelerated hyper-tension, and TMA in SRC patients is mostly due to hypertension as well. However, we found it unusual for this patient to be hypertensive without a cause since his blood pressure had been well controlled without the use of antihypertensive agents before being hospitalized.
ssSSc can be further classified into 3 different subsets: complete (type I), referring to absence of any sclerodermatous skin involvement (skin thickening, sclerodactyly, telangiectasia); incomplete (type II), when there is lack of skin thickening in the presence of other cutaneous features; and delayed (type III), when cutaneous manifestations become detectable later after visceral involvement [10].
Are autoantibodies always positive? No. In 2008, the clinical characteristics and positivity of autoantibodies in ssSSc were described among 1500 patients from the registry of the German Network for Systemic Scleroderma [9]. Women account for 90% of total ssSSc cases. Only 40% of ssSSc patients had positive antitopoisomerase I (anti-Scl-70) antibodies, whereas 35% had anticentromere antibodies. In this study, nearly all patients (95%) had positive ANA. Although anti-RNAP3 antibodies can be found in 3–20% of patients with SSc, depending on the population [11,12], its positivity has been associated with renal crisis in SSc (odds ratio 3.8; 95% confidence interval 1.2–11.5) [13]. However, the prevalence of anti-RNAP3 antibodies in ssSSc has never been established. Thus, negative autoantibodies should not exclude ssSSc if there is high suspicion from internal organ involvement and positive ANA.
How common is renal involvement in ssSSc? More common than the diffuse form. The frequency of internal organ involvement in ssSSc has been consistent across studies [9,14,15]. Gastrointestinal or esophageal involvement is most commonly seen in ssSSc (73–83%), followed by pulmonary involvement (64–73%). Renal involvement occurred in approximately 22% of ssSSc [9]. Hunzelmann et al. suggested that renal involvement may be twice as common in ssSSc compared with dsSSc [9]. Swallowing problems, abdominal pain, nausea, or vomiting are common gastrointestinal symptoms [10]. Pulmonary hypertension followed by interstitial lung disease (ILD) is a common pulmonary complication of SSc [1]. A large majority of patients with ILD presents with a pattern called nonspecific interstitial pneumonia, in which there are ground glass opacities in the peripheries, especially at the subpleural regions of the lung bases [1]. Diffuse alveolar hemorrhage has been demonstrated in SSc as well, but is mostly described in case reports and case series [16–18].
What is the most common lesion in SRC? Thrombotic micro-angiopathy. SRC is a severe complication of SSc,, accounting for 10% of dSSc and only 0.5% in lcSSc [3]. The pathogenesis of SRC is unclear, but recent studies suggested some vasculopathic changes in the kidneys, resulting in thickening of the vessel walls [1]. Renal biopsies are not routinely warranted in all cases but may help guide the diagnosis in difficult cases. The pathologic findings of SRC were first described by Fisher and Rodnan in 1958 [19]. In their study, the thrombotic microangiopathic process is the most common presenting microscopic picture. In SRC, it is possible that TMA lesions could be the result of accelerated hypertension. Of interest, although TMA can be found in other conditions such as hemolytic uremic syndrome (HUS), malignant hypertension, and thrombotic thrombocytopenic purpura (TTP), there are some histologic features that are pathognomonic to SRC. A large proportion of small-vessel thrombi in relation to glomerular thrombi would favor SRC, whereas the opposite is seen in HUS or TTP [20,21]. In our patient, the thrombi were seen mainly in small vessels and none in the glomeruli, which would favor SRC in origin.
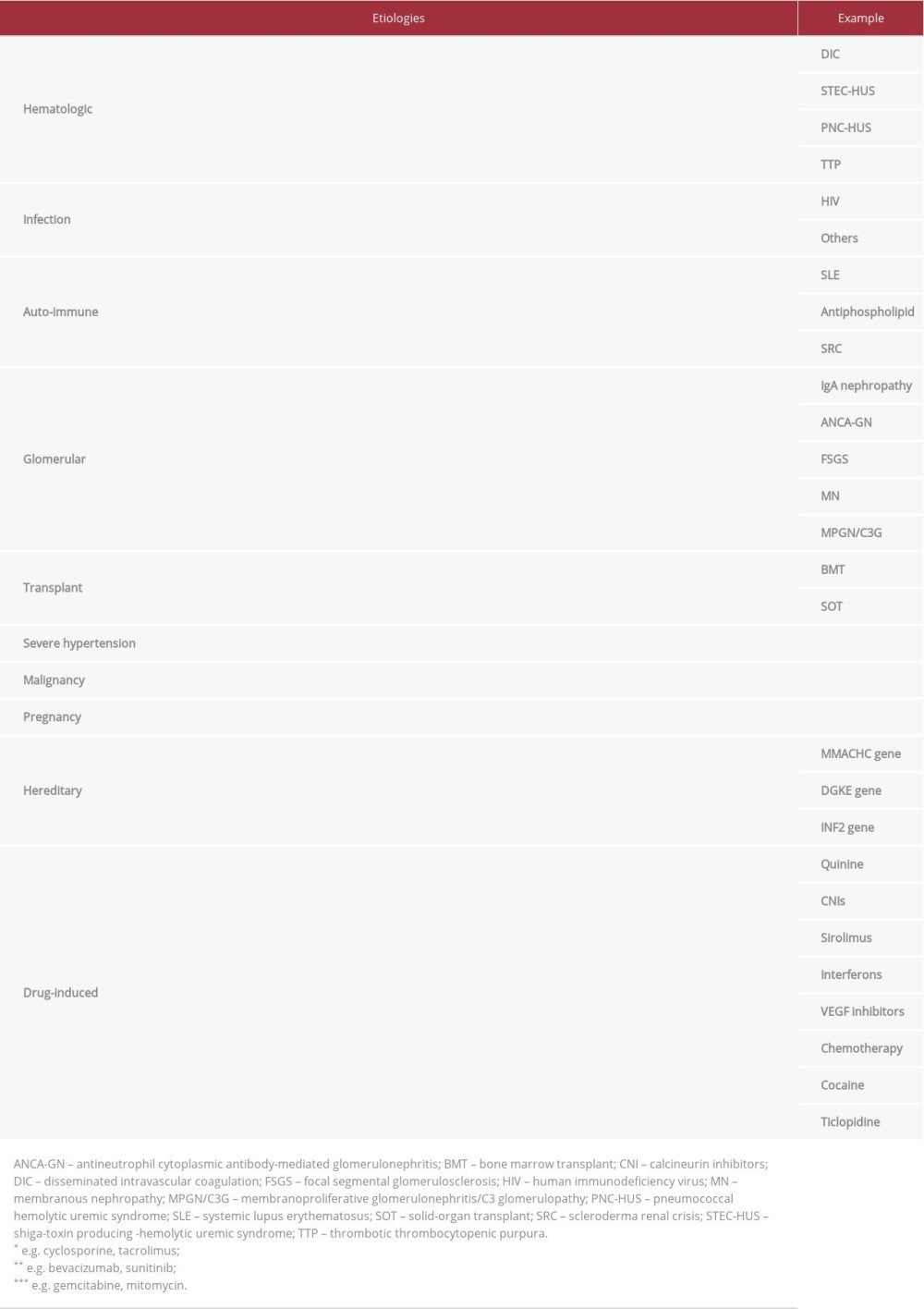
What is TMA? It is a pathologic description of clinical presentation of an organ injury [22]. Histologically, presence of von Willebrand factor- and platelet-rich thrombi in the capillaries and arterioles, with findings reflecting tissue responses to endothelial injury, such as endothelial swelling and mesangiolysis and double contours of the basement membrane, are suggestive for TMA. An etiologic approach to TMA is depicted in Table 3. This table is adjusted from Brocklebank et al. [23]. In our case, SRC and severe hypertension were suspected. By adding the positive pattern from ANA staining and history of easily controlled hypertension before admission, SRC was the most likely cause of TMA.
What is the treatment of SRC? Angiotensin-converting enzyme (ACE) inhibitors are the mainstay antihypertensive medication of choice. Several observational studies have shown that ACE inhibitors are associated with increased survival, preserved kidney function, and better blood pressure control in SRC patients [24–27]. Captopril was commonly used in these studies. Serum creatinine level should be monitored after ACE inhibitor initiation. Although angiotensin II receptor blockers are expected to be effective in SRC patients, the efficacy has not been sufficiently investigated.
Other treatment modalities of SRC? Hemodialysis should be continued if there are indications. Plasma exchange therapy in combination with ACE inhibitors may be considered. In a single-center retrospective cohort study of 20 patients; therapeutic plasma exchange in addition to ACE inhibitor significantly improved 1-year renal survival rate compared with ACE inhibitors alone (80%
In this report, the patient was diagnosed with SRC and suspected diffuse alveolar hemorrhage in the setting of ssSSc. Our patient is interesting in many respects. First, the patient responded to a combination of TP and ACE inhibitor. Although he was started on pre-emptive high-dose steroids when antibodies and kidney biopsy results were pending, steroids were slowly tapered and discontinued once the diagnosis of SRC was made. Second, kidney biopsy is helpful in making the diagnosis but not always necessary. TMA from the kidney biopsy is high yield. Brocklebank et al. have described an algorithm approach to TMA that can lead to the definite diagnosis if followed appropriately [23].
Conclusions
ssSSc with renal crisis is rare and difficult to diagnose clinically. Negative specific autoantibodies should not exclude ssSSc if there is high clinical suspicion and ANA positivity. Kidney biopsy is helpful, as TMA is the most common lesion in SRC. However, TMA alone is not suggestive of SRC if other additional features or ANA are not present. The main treatment of SRC is ACE inhibitors. TP may also be used but corticosteroids should be strongly avoided.
Figures
References:
1.. Sobolewski P, Maślińska M, Wieczorek M, Systemic sclerosis – multi-disciplinary disease: Clinical features and treatment: Reumatologia, 2019; 57(4); 221-33
2.. Al-Dhaher FF, Pope JE, Ouimet JM, Determinants of morbidity and mortality of systemic sclerosis in Canada: Semin Arthritis Rheum, 2010; 39(4); 269-77
3.. Bose N, Chiesa-Vottero A, Chatterjee S, Scleroderma renal crisis: Semin Arthritis Rheum, 2015; 44(6); 687-94
4.. Gore JE, Brown AN, Scleroderma renal crisis sine scleroderma in pregnancy: A case report: Clin Exp Rheumatol, 2006; 24(1); 87-88
5.. Zwettler U, Andrassy K, Waldherr R, Ritz E, Scleroderma renal crisis as a presenting feature in the absence of skin involvement: Am J Kidney Dis, 1993; 22(1); 53-56
6.. Molina JF, Anaya JM, Cabrera GE, Systemic sclerosis sine scleroderma: An unusual presentation in scleroderma renal crisis: J Rheumatol, 1995; 22(3); 557-60
7.. Canet JJ, Castañé J, Alvarez M, Nava JM, Llibre J, Scleroderma renal crisis sine scleroderma: Nephron, 2002; 90(1); 119-20
8.. Clements P, Lachenbruch P, Siebold J, Inter and intraobserver variability of total skin thickness score (modified Rodnan TSS) in systemic sclerosis: J Rheumatol, 1995; 22(7); 1281-85
9.. Hunzelmann N, Genth E, Krieg T, The registry of the German Network for Systemic Scleroderma: Frequency of disease subsets and patterns of organ involvement: Rheumatology (Oxford), 2008; 47(8); 1185-92
10.. Kucharz EJ, Kopeć-Mędrek M, Systemic sclerosis sine scleroderma: Adv Clin Exp Med, 2017; 26(5); 875-80
11.. Santiago M, Baron M, Hudson M, Antibodies to RNA polymerase III in systemic sclerosis detected by ELISA: J Rheumatol, 2007; 34(7); 1528-34
12.. Foocharoen C, Watcharenwong P, Netwijitpan S, Relevance of clinical and autoantibody profiles in systemic sclerosis among Thais: Int J Rheum Dis, 2017; 20(10); 1572-81
13.. Nikpour M, Hissaria P, Byron J, Prevalence, correlates and clinical usefulness of antibodies to RNA polymerase III in systemic sclerosis: A cross-sectional analysis of data from an Australian cohort: Arthritis Res Ther, 2011; 13(6); R211
14.. Marangoni RG, Rocha LF, Del Rio AP, Systemic sclerosis sine scleroderma: Distinct features in a large Brazilian cohort: Rheumatology (Oxford), 2013; 52(8); 1520-24
15.. Poormoghim H, Lucas M, Fertig N, Medsger TA, Systemic sclerosis sine scleroderma: Demographic, clinical, and serologic features and survival in forty-eight patients: Arthritis Rheum, 2000; 43(2); 444-51
16.. Naniwa T, Banno S, Sugiura Y, Pulmonary-renal syndrome in systemic sclerosis: A report of three cases and review of the literature: Mod Rheumatol, 2007; 17(1); 37-44
17.. Chaer RA, Massad MG, Evans A, Systemic sclerosis complicated by diffuse alveolar hemorrhage: Med Sci Monit, 2001; 7(5); 1013-15
18.. Herndon TM, Kim TT, Goeckeritz BE, Alveolar hemorrhage and pulmonary hypertension in systemic sclerosis: A continuum of scleroderma renal crisis?: J Clin Rheumatol, 2001; 7(2); 115-19
19.. Fisher ER, Rodnan GP, Pathologic observations concerning the kidney in progressive systemic sclerosis: AMA Arch Pathol, 1958; 65(1); 29-39
20.. Batal I, Domsic RT, Shafer A, Renal biopsy findings predicting outcome in scleroderma renal crisis: Hum Pathol, 2009; 40(3); 332-40
21.. Tostivint I, Mougenot B, Flahault A, Adult haemolytic and uraemic syndrome: Causes and prognostic factors in the last decade: Nephrol Dial Transplant, 2002; 17(7); 1228-34
22.. Moake JL, Thrombotic microangiopathies: N Engl J Med, 2002; 347(8); 589-600
23.. Brocklebank V, Wood KM, Kavanagh D, Thrombotic microangiopathy and the kidney: Clin J Am Soc Nephrol, 2018; 13(2); 300-17
24.. Penn H, Howie AJ, Kingdon EJ, Scleroderma renal crisis: Patient characteristics and long-term outcomes: Q J Med, 2007; 100(8); 485-94
25.. Steen VD, Medsger TA, Long-term outcomes of scleroderma renal crisis: Ann Intern Med, 2000; 133(8); 600-3
26.. Steen VD, Costantino JP, Shapiro AP, Medsger TA, Outcome of renal crisis in systemic sclerosis: Relation to availability of angiotensin converting enzyme (ACE) inhibitors: Ann Intern Med, 1990; 113(5); 352-57
27.. Beckett VL, Donadio JV, Brennan LA, Use of captopril as early therapy for renal scleroderma: A prospective study: Mayo Clin Proc, 1985; 60(11); 763-71
28.. Cozzi F, Marson P, Cardarelli S, Prognosis of scleroderma renal crisis: A long-term observational study: Nephrol Dial Transplant, 2012; 27(12); 4398-403
29.. Steen VD, Medsger TA, Case-control study of corticosteroids and other drugs that either precipitate or protect from the development of scleroderma renal crisis: Arthritis Rheum, 1998; 41(9); 1613-19
30.. Teixeira L, Mouthon L, Mahr A, Mortality and risk factors of scleroderma renal crisis: A French retrospective study of 50 patients: Ann Rheum Dis, 2008; 67(1); 110-16
Figures
Tables
In Press
16 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943687
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943070
17 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943370
18 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943803
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250