02 November 2020: Articles 
Brodifacoum-Laced Synthetic Marijuana Toxicity: A Fight Against Time
Challenging differential diagnosis, Unusual setting of medical care
Sanjay Kumar1AEF*, Geeta Bhagia1AEFDOI: 10.12659/AJCR.927111
Am J Case Rep 2020; 21:e927111
Abstract
BACKGROUND: As marijuana is being legalized in some states in the United States, there is a growing need for physicians to be aware of potential complications related to various forms of marijuana used in the community. Historically, marijuana has been laced with potentially toxic substances to increase its efficacy, and brodifacoum is one of them. Here, we present the case of a patient with toxicity related to use of brodifacoum-laced synthetic marijuana.
CASE REPORT: A 30-year-old man with history of polysubstance abuse presented with 5 days of flank pain and hematuria. He reported current use of synthetic marijuana. Vital signs were unremarkable. On physical examination, he had petechiae on bilateral upper extremities. Pertinent lab findings included: leukocytosis of 14 000 K/UL, international normalized ratio (INR) 13, prothrombin time (PT) 134.6 s, activated partial thromboplastin time (aPTT) 58.3 s, and only hematuria on urinalysis. CT scans of the abdomen and pelvis were unremarkable. The initial toxicology screen was negative. Brodifacoum toxicity was suspected. The patient was managed in collaboration with poison control, and he was treated with oral vitamin K and close monitoring of INR.
CONCLUSIONS: Brodifacoum-laced synthetic marijuana toxicity can lead to potentially lethal complications if not recognized and treated in a timely manner. Hence, physicians should have a high index of suspicion in patients presenting with unexplained coagulation abnormalities.
Keywords: Blood Coagulation, Marijuana Abuse, Vitamin K, 4-Hydroxycoumarins, Cannabis, Rodenticides
Background
As of April 25, 2018, around 155 cases of coagulopathy from synthetic marijuana laced with brodifacoum have been reported in Illinois [1]. Indiana, Missouri, and Maryland have also reported cases of this new and worrisome drug. The Centers for Disease Control and Prevention (CDC) and hospitals nationwide are recognizing the threat from this growing epidemic, and 4 deaths have been reported [1]. Unlike other FDA-approved anticoagulants, the clinical course and management of coagulopathy associated with brodifacoum are unfamiliar and challenging to most physicians. The aim of this case report is to highlight the need for timely diagnosis of brodifacoum-associated coagulopathy and the uniqueness of its treatment.
Case Report
A 30-year-old man came to the Emergency Department (ED) with right lower-quadrant (RLQ) abdominal pain for 5 days. The pain felt sharp and burning, constant, 5/10 in intensity, radiating to the right flank, and associated with nausea and dark brown urine. The patient denied urethral discharge, fevers, chills, melena/bright red bleeding per rectum, or vomiting. He reported no recent trauma. His past medical history was positive for polysubstance abuse including cocaine, K-2/spice, and alcohol abuse. He was not aware of any bleeding/clotting disorders or any other comorbid conditions in the family. The patient was homeless, unemployed, and living with his girlfriend. He had no known drug or food allergies.
On physical examination, vital signs were temperature 36.8°C, pulse 68 beats per min, respiratory rate 22 breaths per min, oxygen saturation 100% on room air, height 178 cm, weight 52.6 kg, body mass index 16.6, and blood pressure 134/69 mmHg. He was anxious and appeared cachectic. There was a cigarette burn on his right cheek that was not actively bleeding. His pupils were equal bilaterally and reactive, and extraocular movements were normal. There was mild gingival bleeding. There was no jugular venous distention. Heart sounds were normal. Lungs were clear to auscultation. The abdomen was soft, non-tender, and non-distended. Bowel sounds were normal. Results of a genitourinary exam were normal and there was no discharge or blood at the urethral meatus. There was no peripheral edema. The results of a neurology exam were unremarkable. There were petechiae on the bilateral upper extremities and ecchymoses on the right lower extremity.
Initial laboratory studies revealed hemoglobin 16.5g/dL, white blood cell count 14 200/µL, platelet count 229 000/µL, sodium 139 mEq/L, potassium 4.5 mmol/L, chloride 104 mEq/L, bicarbonate 23 mEq/L, blood urea nitrogen 11 mg/dL, creatinine 0.78 mg/dL, calcium 9.8 mg/dL, glucose 111 mg/dL, albumin 4.2 g/dL, total protein 7 g/dL, alkaline phosphate 78 IU/L, aspartate transaminase 24 IU/L, alanine transaminase 19 IU/L, and bilirubin 0.5 mg/dL. On urinalysis, the urine was brown and cloudy, with moderate bilirubin, ketones 40 mg/dL, hematuria, and protein 100 mg/dL. Urine microscopy was positive only for too many red blood cells to count. Urine toxicology was negative. His international normalized ratio (INR) on admission was >13, prothrombin time (PT) 134.6 s, and activated partial thromboplastin time (PTT) was 58.3 s. CT scans of the abdomen and pelvis without contrast were only remarkable for a 1-mm non-obstructing calcification seen in the right lower pole of the kidney. A CT scan head without contrast was negative for acute intracranial bleeding.
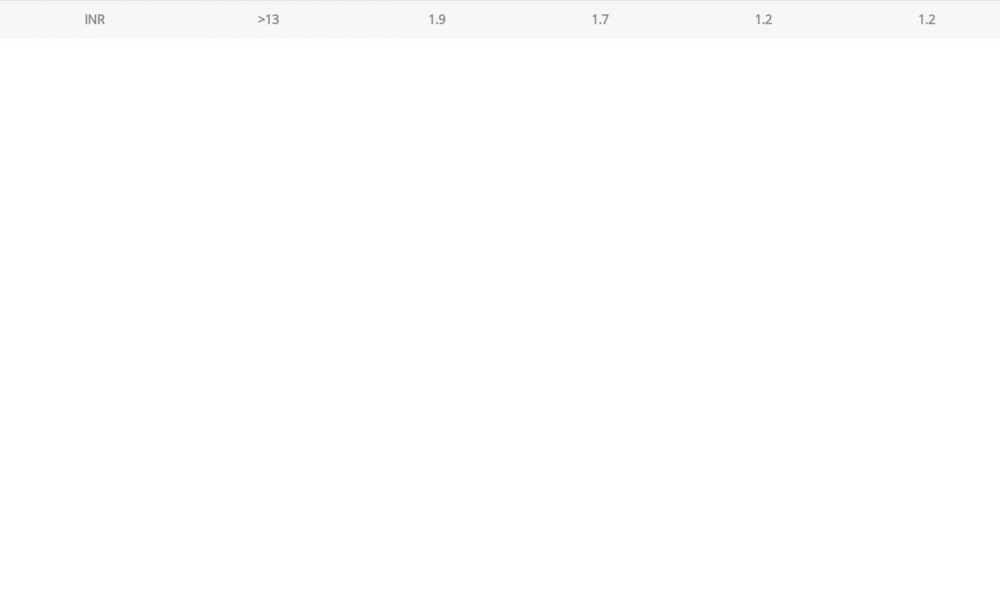
On further history-taking after lab results were obtained, the patient reported that he started using K2/spice from a new buyer, and one of his friends who used the same product was also admitted in a different hospital with similar complaints. Based on the patient’s history, brodifacoum toxicity was suspected due to possible use of laced synthetic marijuana. Poison control was contacted and they recommended 50 mg of oral vitamin K 3 times a day and INR monitoring every 12 h. As the patient was not actively bleeding, fresh frozen plasma (FFP) or K-centra was not given. The patient’s INR trend is noted in Table 1.
The patient’s symptoms improved during the hospital stay but he decided to leave against medical advice 2 days later, despite our counselling; he verbalized understanding the risks.
After leaving the hospital, he continued to use K2/spice, and 14 days later he was admitted to the hospital again with similar symptoms. His initial INR was >13 with normal liver function test results. Ten mg Intravenous vitamin K was given and along with 50 mg oral vitamin K 3 times a day as per recommendations from poison control. His INR trend is noted in Table 2.
On day 3, he was transferred to the inpatient psychiatry unit for management of depression and polysubstance use disorder.
Case Report
Synthetic marijuana/K2 spice is laced with many agents, including brodifacoum, which increases the euphoric effect by slowing its metabolism (through saturating liver enzymes) [2,3]. Brodifacoum was first introduced in the late 1970s to poison warfarin-resistant rodents [4]. It was synthesized by replacing a methyl group of methyl 4-hydroxycoumarin with a phenyl group, which made it highly lipid-soluble, leading to its longer duration of action and greater potency [5,6]. Brodifacoum is about 100 times more potent than warfarin [7] and its half-life ranges from 16 to 34 days [4]. It has been observed to be persistent for 16 to 270 days [4]. It decreases hepatic production of vitamin K-dependent factors (e.g., II, VII, IX, X, protein C, and protein S) by inhibiting 2,3-vitamin K epoxide reductase, leading to increased PT/INR. It also increases permeability of capillaries, which further increases risk of bleeding. A fatal dose for a 60-kg man is 15 mg without treatment. The onset of increased INR can be delayed by up to 48 h after ingestion, so it is very important to monitor patients with brodifacoum ingestion by regularly checking the INR [3,4,8].
This diagnosis should be suspected in any patient with a suspicious history and high PT/INR and/or high PTT. Some experts have recommended high-performance liquid chromatography to check its level [4]. Mixing studies would demonstrate correction of PT and PTT to normal, hence, suggesting deficiency of vitamin K-dependent factors due to decreased hepatic synthesis [3].
Current guidelines recommend that asymptomatic patients should continue to be observed without treatment for any coagulation abnormalities at 48–72 h after exposure [9,10]. If a patient has coagulation abnormalities and active bleeding, FFPs are recommended along with IV vitamin K, frequently given at doses of 20 to 50 mg, and some patients have received higher doses [4]. Human studies have indicated that vitamin K-dependent factor levels begin to rise 6 to 8 h after treatment, but recurrent symptoms and coagulation abnormalities have been noted at 12 to 16 h after initial treatment. Therefore, maintenance therapy with oral vitamin K should be started promptly [4]. Maintenance doses vary depending upon the condition, ranging from 15 mg to 600 mg per day in divided doses. The most common dosage is 100 mg per day (given in divided doses), usually given for 30 to 200 days (the duration of treatment can vary) [3,4,8].
Other therapies that can be used in acute cases include recombinant factor VIIa and prothrombin complex concentrate [4]. Phenobarbital has been shown to increase microsomal activity, thus increasing drug metabolism/clearance, but it has not been shown to increase survival or hasten recovery, so its use is not recommended [11].
Conclusions
Our case report emphasizes that physicians should maintain a high index of suspicion for toxicity related to brodifacoum-laced synthetic marijuana, especially in patients presenting with unexplained coagulation abnormalities with or without bleeding. The initial urine toxicology screen may be negative, but serum drug level/mixing studies and further evaluation can reveal the appropriate diagnosis. Prolonged treatment with vitamin K and regular INR monitoring remain the cornerstone of management and can prevent potential lethal complications.
References:
1.. Moritz E, Austin C, Wahl M, Notes from the field: Outbreak of severe illness linked to the vitamin K antagonist brodifacoum and use of synthetic cannabinoids – Illinois, March–April 2018: Morb Mortal Wkly Rep, 2018; 67; 607-8
2.. Wallin R, Hutson SM, Warfarin and the vitamin K-dependent γ-carboxylation system: Trends Mol Med, 2004; 10(7); 299-302
3.. Spahr JE, Maul JS, Rodgers GM, Superwarfarin poisoning: A report of two cases and review of the literature: Am J Hematol, 2007; 82(7); 656-60
4.. King N, Tran M-H, Long-acting anticoagulant rodenticide (superwarfarin) poisoning: A review of its historical development, epidemiology, and clinical management: Transfusion Med Rev, 2015; 29(4); 250-58
5.. Hadler MR, Buckle AP, Forty-five years of anticoagulant rodenticides – past, present and future trends: Fifteenth Vertebrate Pest Conference, 1992; 1992, Davis, University of California
6.. Hadler MR, Shadbolt RS, Novel 4-hydroxycoumarin anticoagulants active against resistant rats: Nature, 1975; 253(5489); 275-77
7.. Lipton RA, Klass EM, Human ingestion of a ‘super warfarin’ rodenticide resulting in a prolonged anticoagulant effect: JAMA, 1984; 252(21); 3004-5
8.. Tran M-H, King NC, Epidemiology and symptomatology of long acting anticoagulant rodenticide poisoning: Journal of Epidemiological Research, 2015; 2(2); 1
9.. Caravati EM, Erdman AR, Scharman EJ, Longacting anticoagulant rodenticide poisoning: An evidence-based consensus guideline for out-of-hospital management: Clin Toxicol (Phila), 2007; 45(1); 1-22
10.. Lepine MLI, Gonzalo FE, Ferrer FC, Tous EG, Superwarfarin rodenticide intoxication in adults: An update on bromadiolone, brodifacoum, and difethialone: Emergencias, 2013; 25; 201-3
11.. Oliveri CV, Stubbs JR, Chernova T, Short-term use of phenobarbital in the treatment of “superwarfarin” poisoning: Blood, 2004; 104(11); 4024
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133