19 November 2020: Articles 
Symptomatic Giant Primary Nonparasitic Splenic Cyst Treated with Laparoscopic Decapsulation: A Case Report and Literature Review
Challenging differential diagnosis, Rare disease
Salah Termos1AF*, Feras Othman1BEFG, Ali Aljewaied1E, Afaq Mahmoud Alkhalil1F, Mohammad Alhunaidi1A, Socrates Mathew Parayil2D, Fahad Alabdulghani3CDDOI: 10.12659/AJCR.927893
Am J Case Rep 2020; 21:e927893
Abstract
BACKGROUND: Primary nonparasitic splenic cysts (PNSC) are unusual epithelial fluid lesions of the spleen. They are considered congenital cysts and are often discovered incidentally in young people. Larger cysts can be symptomatic and are traditionally managed with splenectomy. This report is of a woman with a large symptomatic PNSC that was managed surgically by laparoscopic decapsulation.
CASE REPORT: A 22-year-old Lebanese woman presented with left upper-quadrant pain, left pleuritic pain, food intolerance, and significant weight loss. Investigations showed a 20×17×15 cm cystic lesion in the spleen. Secondary causes were ruled out and tumor marker and hydatid serology were unremarkable. Laparoscopic decapsulation of the cyst with spleen preservation was performed with no perioperative complications. The patient’s 3-year follow-up visit revealed no clinical or radiological recurrence.
CONCLUSIONS: True congenital splenic cysts are rare clinical findings. Generally, they do not have malignant potential. The development of minimally invasive techniques has shifted the trend toward splenic salvaging procedures. Literature review revealed an acceptable recurrence rate with near-total rather than partial unroofing. Laparoscopic decapsulation can be a safe and adequate therapeutic option in selected cases.
Keywords: Laparoscopy, Epithelial Cyst, Decapsulation, PNSC, Primary Nonparasitic Splenic Cyst, Adolescent, Cysts, Splenectomy, Splenic Diseases, young adult
Background
Splenic cysts are categorized etiologically into parasitic and nonparasitic cysts. Nonparasotic cysts can be primary or congenital cysts and secondary cysts, which are most commonly pseudocysts due to trauma [1]. Primary nonparasitic splenic cysts (PNSCs), also referred to as splenic epidermoid or splenic epithelial cysts, are unilocular fluid lesions with an epithelial lining. They have thin smooth nonenhancing wall [2] and are usually innocuous incidental imaging findings encountered mostly in children and young adults, which comprise 20% of all cases of nonparasitic cysts [3].
Large congenital splenic cysts are usually symptomatic and are a rare finding in routine practice. They were previously treated with splenectomy. Decapsulation is an unroofing technique characterized by near-total excision of the cyst, with splenic preservation. We describe the case of a giant PNSC treated surgically by laparoscopic decapsulation.
Case Report
A 22-year-old Lebanese woman, who was otherwise healthy, presented with a persistent dull aching left upper-quadrant pain which was associated with left pleuritic pain, early satiety, and subsequent significant weight loss. She denied any contact with animals and noted no history of trauma. Physical exam revealed she was pale and thin with a notable bulge on her left upper quadrant causing asymmetry of her abdominal wall. Laboratory tests were remarkable only for iron deficiency anemia.
Ultrasound revealed a well-defined encapsulated hypoechoic cyst arising within the spleen measuring 19×16 cm with posterior acoustic enhancement indicating a thin-walled fluid-filled lesion and absence of a solid component (Figure 1). Contrast-enhanced computed tomography (CECT) showed a 20×16 cm homogenous hypodense lesion originating from the spleen, lacking septations and enhancing or a solid component. A mass effect was noted because of the size of the cyst and was represented by an atrophy of the left lateral liver segments and shifting of the stomach to the right side. Radiologic features were consistent with a simple splenic cyst (Figure 2).
The decision from a multidisciplinary team meeting was to exclude malignancy and secondary causes with an attempt to salvage the spleen. Tumor markers, carcinoembryonic antigen (CEA), and cancer antigen (CA) 19.9, were in the normal range, and a hydatid serology test, indirect hemagglutination test, and enzyme-linked immunosorbent assay (ELISA) were negative. The patient was provided with post-splenectomy triple vaccinations. She signed consent for a minimally invasive
The laparoscopic exploration was performed with 3 trocars: one 10-mm port used for the camera and two 5-mm ports used as functioning trocars. The operative findings revealed a giant cyst arising from the upper pole and occupying almost the entire surface of the spleen. Compression and displacement of the gastric wall were also noted (Figure 3). The cyst lesion was carefully dissected from the adherent adjacent structures and entirely exposed. Since the patient was Lebanese (Lebanon is an endemic hydatid region), the field was prepped and surrounded with hypertonic saline-impregnated gauzes to prevent any spillage, and all safety measures were taken for possible hydatid anaphylaxis. The cyst was then punctured using a laparoscopic needle and about 2 L of turbid greyish-yellowish fluid was aspirated. A frozen section was performed and denoted a simple congenital cyst with no malignant cells. Decapsulation (near-total unroofing) was done using a harmonic scalpel device (Ethicon, Johnson & Johnson) by excising approximately 90% of the cyst wall (Figure 4). The specimen was removed in an endobag to avoid contamination. Hemostasis was achieved and the greater omentum was fixed to the incision surface using clips. A vacuum drain was placed near the incision surface.
The macroscopic aspect showed a glistening inner surface having a heavily trabeculated appearance resembling endocardium (Figure 5). Microscopic examination demonstrated a nonkeratinizing squamous epithelial lining with the presence of a Gamna-Gandy body and absence of malignancy (Figure 6). The histopathologic findings were consistent with a congenital splenic cyst. The patient’s hospital stay was uneventful, she was discharged on postoperative day 2, and prompt improvement of her symptoms was observed. The patient’s 3-year follow-up visit revealed no clinical or radiological recurrence.
Discussion
Cystic lesion of the spleen is a rare medical diagnosis. Classification of these cysts has evolved since the first case was reported by Andral in 1929. Splenic cysts are divided etio-logically into parasitic and nonparasitic cysts. Parasitic cysts are seen in endemic areas and are often caused by
Splenic cysts were categorized histologically by Martin into false cysts and true cysts, based on the presence or absence of a cellular lining of the cystic wall [7]. False splenic cysts are secondary cysts; they are deprived of an epithelium and are called pseudocysts. False splenic cysts account for 80% of all splenic cysts and usually occur as a result of trauma, disorganized hematoma, splenic infarction, intrasplenic pancreatic pseudocyst, and splenic abscess. True splenic cysts are primary cysts that have an epithelial lining. They represent only 20% of splenic cysts and can be neoplastic in origin (hemangioma and lymphangioma), infectious (mostly hydatid disease), or congenital (primary epithelial cysts) [8]. They are usually thin-walled, contain clear or turbid yellowish fluid, and have a heavily trabeculated appearance in the inner layer covered by a shiny lining resembling the endocardium. Their epithelium lining may be squamous, columnar, or cuboidal. These cysts are immunoreactive for markers CEA and CA 19.9, which may be elevated in blood serum and usually return to baseline after removal of the cyst. Gamna-Gandy bodies are iron encrustations of the connective tissue and sclerosiderotic nodules. They are often associated with reactions to a foreign body but may be found in PNSC as a result of focal hemorrhage. Therefore, histopathological evaluation is necessary to sub-type splenic cysts and rule out malignancy [9].
Splenic congenital cysts, or epidermoid cysts, are also referred to as PNSCs and are uncommon fluid lesions which constitute 10% of all nonparasitic cysts of the spleen [10]. The pathogenesis of PNSCs is not clear. Many hypotheses were proposed for congenital cysts. One states they are usually formed due to the unfolding of the peritoneal mesothelium or by collections of peritoneal mesothelial cells trapped within the splenic sulci. The mesothelium undergoes metaplasia to squamous epithelium secondary to chronic irritation [11]. Another theory states that the cyst may arise from lymph spaces of the spleen. While most cases of PNSC are sporadic, in rare cases, there is a familial occurrence [12].
PNSCs occur predominantly in the second and third decades of life but can occur in the pediatric age group. They are more common in the female than male sex [13]. Small cysts are often asymptomatic but they can grow slowly and become large and symptomatic by the time of discovery. The typical symptom in 30% to 40% of cases, however, is a painless mass in the left hypochondrium [14]. PNSCs can also present as a localized abdominal or referred pain, which can be due to splenomegaly and the mass effect on the surrounding structures. Initial manifestations are primarily gastrointestinal and include early satiety, nausea, vomiting, and dysphagia [15]. PNSCs are solitary in 80% of cases and can also present with complications like infection, hemorrhage, and rupture. On rare occasions, they can cause thrombocytopenia or elevated systemic blood pressure due to renal artery compression or even pleuritic cough [2,3].
Definitive diagnosis is made after surgery when the epithelial lining is tested by histopathology and immunochemistry stains [16]. But a comprehensive differential diagnosis for cystic lesions of the spleen is mandatory. Physical examination and routine laboratory studies are usually unremarkable. Tumor markers CEA and CA 19.9 may be elevated. Ultrasonography is a reliable noninvasive tool for differentiating between solid and cystic lesions in most cases. Ultrasound characteristics include an anechoic mass with thin walls and septations or irregular walls. Peripheral or focal calcifications are uncommon, occurring in 10% to15% of cases; however, they can be seen in complex cases [17]. CT scanning and magnetic resonance imaging can give detailed information regarding the morphology of the cyst as well as its exact dimensions and relation to the hilum and surrounding structures [18,19].
The proper management of congenital splenic cysts is still controversial because of the absence of a standardized protocol. The debate regarding treatment is in deciding which cysts should be surgically managed and by what method. According to the limited data in the literature, the size and location of splenic cysts are the major factors determining the therapeutic decision. Small cysts, less than 4 cm, are usually asymptomatic and can be observed only. Larger cysts are potentially symptomatic and require surgical intervention. Also, the site of the cyst has a significant value in decision making for spleen preservation. Upper pole and lateral cysts can be managed with a splenic salvaging procedure; however, splenectomy is recommended for disease multiplicity and for lesions located in the lower pole or near the hilum [20].
Different surgical procedures were designed in the literature according to the age of the patient and the size, location, type, and presentation of the cyst. These options include open
Recently, in the era of minimally invasive techniques, laparoscopic splenectomy, simple unroofing, and decapsulation can be performed with shorter hospital stays, less morbidity, and better cosmetic results [21]. Registered data show a higher rate of recurrence with partial unroofing compared to with decapsulation (near-total unroofing), which is excising as much as possible from the cyst wall and its epithelial lining to prevent reclosure and reaccumulation of the cyst [23,24]. Mackenzie et al. reported the laparoscopic decapsulation of congenital splenic cysts using a harmonic scalpel and diathermy in 3 patients and recommended this technique as an effective, safe method [25].
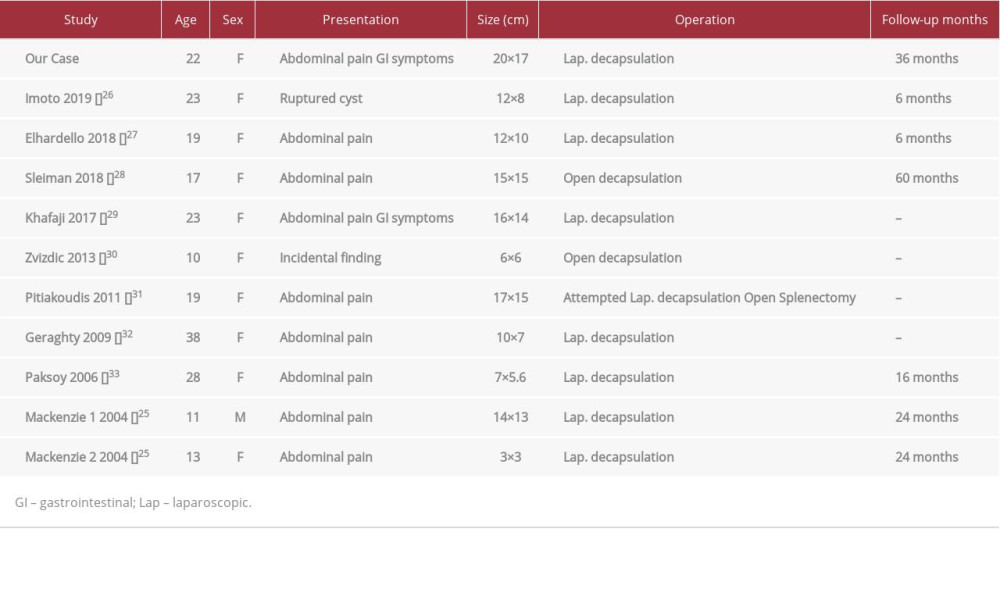
Our literature search in PubMed and Google Scholar included relevant keywords and data between 1980 and 2020. We included only cases of congenital splenic cysts and excluded surgeries for other splenic pathologies. We included cases managed with decapsulation and excluded studies reporting treatment with splenectomy or only partial unroofing. The search for spleen preserving decapsulation in PNSCs revealed only 11 clearly reported cases (Table 1). Most cases were symptomatic young female patients having cysts larger than 10 cm. Of these 11 reported cases, 9 were managed successfully via laparoscopy and 2 by open approach. Only 1 of the 9 laparoscopy cases underwent completion splenectomy due to a postoperative complication. In the present case, the patient was a 22-year-old woman from Lebanon who presented with a severely symptomatic large cyst, which was a clear indication for surgery. The endemic area of the patient directed us toward hydatid disease; however, we excluded the parasitic infection because of the absence of radiologic findings and negative serology results. Given the favorable location of the cyst and the absence of any potentially malignant findings, splenic preserving laparoscopic decapsulation was done to relieve her symptoms and salvage her spleen.
Conclusions
Cystic lesion of the spleen is a rare entity and its management continues to evolve. The available literature on the treatment of a huge PNSC advocates total splenectomy; however, a minimally invasive approach is usually preferred. The laparoscopic decapsulation of giant congenital cysts, instead of splenectomy, can be an adequate and safe surgical option in selected patients. More studies are needed with larger sample sizes and longer follow-up periods.
Figures
References:
1.. Boybeyi O, Karnak I, Tanyel FC, The management of primary nonparasitic splenic cysts: Turkish J Pediatr, 2010; 52; 500-4
2.. Dachman AH, Ros PR, Murari PJ, Nonparasitic splenic cysts: A report of 52 cases with radiologic-pathologic correlation: Am J Roentgenol, 1986; 147(3); 537-42
3.. Morgenstern L, Nonparasitic splenic cysts: Pathogenesis, classification, and treatment: J Am Coll Surg, 2002; 194; 306-14
4.. Safioleas M, Misiakos E, Manti C, Surgical treatment for splenic hydatidosis: World J Surg, 1997; 21; 374-78
5.. Morgenstern L, Non-parasitic splenic cysts: Pathogenesis, classification and treatment: J Am Coll Surg, 2002; 194; 306-14
6.. Abi Saad GS, Hussein M, El Saghir NS, Isolated splenic metastasis from colorectal cancer: Int J Clin Oncol, 2011; 16(4); 306-13
7.. Martin JW, Congenital splenic cysts: Am J Surg, 1958; 96; 302-8
8.. Macheras A, Misiakos E, Liakakos T, Non-parasitic cysts: A report of three cases: World J Gastroenterol, 2005; 11; 6884-87
9.. Palmieri I, Natale E, Crafa F, Epithelial splenic cysts: Anticancer Res, 2005; 25; 515-22
10.. Belekar D, Desai A, Dewoolkar A, Splenic epithelial cyst: A rare entity: Int J Surg, 2010; 22; 1-18
11.. Robbins FG, Splenic epidermoid cysts: Ann Surg, 1978; 187; 231-35
12.. Williams RJ, Glazer G, Splenic cysts: Changes in diagnosis, treatment and aetiological concepts: Ann R Coll Surg Engl, 1993; 75; 87-89
13.. Sakamoto Y, Yunotani S, Edakuni G, Laparoscopic splenectomy for a giant splenic epidermoid cyst: Report of a case: Surg Today, 1999; 29; 1268-72
14.. Santos HL, Sodré FC, De Macedo HW, Blastocystis sp. in splenic cysts: Causative agent or accidental association? A unique case report: Parasit Vectors, 2014; 7; 207
15.. Madia C, Lumachi F, Veroux M, Giant splenic epithelial cyst with elevated tumor markers CEA and CA 19.9: An incidental association?: Anticancer Res, 2003; 23; 773-76
16.. Ingle SB, Hinge CR, Jatal SN, An interesting case of primary epithelial cyst of spleen: Indian J Pathol Microbiol, 2013; 56; 181-82
17.. Siniluoto TM, Päivänsalo MJ, Lähde ST, Nonparasitic splenic cysts. Ultrasonographic features and follow-up: Acta Radiol, 1994; 35; 447-51
18.. Robertson F, Leander P, Ekberg O, Radiology of the spleen: Eur Radiol, 2001; 11; 80-95
19.. Termos S, Redha A, Zbibo R, Torsion of huge wandering accessory spleen. Case report and literature review: Int J Sci Res, 2017; 38; 131-35
20.. Yoh T, Wada S, Kobayashi A, Laparoscopic splenectomy for a large multilocular splenic cyst with elevated CA19-9: Report of a case: Int J Surg Case Rep, 2013; 4(3); 319-21
21.. Szczepanik AB, Meissner AJ, Partial splenectomy in the management of nonparasitic splenic cysts: World J Surg, 2009; 33; 852-56
22.. Grinblat J, Gilboa Y, Overwhelming pneumococcal sepsis 25 years after splenectomy: Am J Med Sci, 1975; 270; 523-24
23.. Touloukian RJ, Seashore JH, Partial splenic decapsulation: A simplified operation for splenic pseudocyst: J Pediatr Surg, 1987; 22; 135-37
24.. Hansen MB, Moller AC, Splenic cysts: Surg Laparosc Endosc Percutan Tech, 2004; 14(6); 316-22
25.. Mackenzie RK, Youngson GG, Mahomed AA, Laparoscopic decapsulation of congenital splenic cysts: a step forward in splenic preservation: J Pediat Surg, 2004; 39; 88-90
26.. Imoto Y, Einama T, Fukumura M, Laparoscopic fenestration for a large ruptured splenic cyst with an elevated CA19.9 level: A case report: BMC Surg, 2019; 19; 57
27.. Elhardello O, Ammori B, Splenic pedicle control during laparoscopic decapsulation of a giant splenic cyst: J Surg Rep, 2018; 2018(1); rjx255
28.. Sleiman Y, Bohlok A, Elkhoury M, Splenic epithelial cyst mistaken with hydatid cyst: A case report: Int J Surg Case Rep, 2018; 53; 21-24
29.. AlKhafaji B, Younis MU, Laparoscopic splenic cyst fenestration – a viable spleen preserving option: J Surg Case Rep, 2017; 2017(8); rjx154
30.. Zvizdic Z, Karavdic K, Splenic preserving surgery in treatment of large mesothelial splenic cyst in children: Bons J Basic Med Sci, 2013; 13(2); 126-28
31.. Pitiakoudis M, Zezos P, Oikonomou A, Total splenectomy due to an unexpected “complication” after successful extended laparoscopic partial decapsulation of a giant epidermoid splenic cyst: A case report: Case Rep Med, 2011; 2011; 318208
32.. Geraghty M, Khan IZ, Konlon KC, Large primary splenic cyst: A laparoscopic technique: J Minim Access Surg, 2009; 5(1); 14-16
33.. Paksoy M, Karabicak I, Ramazan K, Laparoscopic splenic total cystectomy in a patient with elevated CA 19-9: JSLS, 2006; 10(4); 507-10
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133