02 March 2021: Articles 
Infection-Associated Loss of Vision: A Case Report in a Patient with a History of Intravenous Drug Use
Rare disease
Shirisha Pasula1ABCDEFG*, Vichar Trivedi2BEF, Elvisa Loshe3BD, Pranatharthi Chandrasekar2DEGDOI: 10.12659/AJCR.929116
Am J Case Rep 2021; 22:e929116
Abstract
BACKGROUND: Serratia marcescens infections have historic association with injection drug use. The present report is about a 53-year-old man with a history of intravenous (IV) drug use who presented with acute loss of vision due to endophthalmitis associated with disseminated S. marcescens infection.
CASE REPORT: A 53-year-old man with a history of active illicit IV drug use presented with a chief complaint of loss of vision in his left eye for 5 days. He also reported having a fever, chills, and shortness of breath. While in the Emergency Department, he became hypotensive and hypoxic. He needed to be intubated and was started on vasopressor support. An ophthalmological examination was suspicious for endophthalmitis. The patient underwent a vitreous tap with injection of intravitreal antibiotics on the day of admission. An echocardiogram showed severe tricuspid endocarditis requiring valve replacement. He also was found to have a left lung/pleural abscess, which was surgically drained. Later, a left eye vitrectomy was performed and the intravitreal antibiotics were repeated; the treatment was unsuccessful and enucleation eventually was required. In addition, the patient had gastric bleeding and underwent esophagogastroduodenoscopy, which showed ischemic stomach ulcers suggestive of septic emboli. Cultures of the patient’s blood, tricuspid valve, lung abscess, and vitreous fluid revealed S. marcescens. He was treated long term with a 2-drug antibiotic regimen and discharged in stable condition.
CONCLUSIONS: We have presented a rare case of acute loss of vision due to endophthalmitis in a patient with a history of IV drug use, which was associated with disseminated infection with the Gram-negative saprophyte S. marcescens.
Keywords: Endocarditis, Bacterial, Endophthalmitis, Serratia Infections, Street Drugs, Pharmaceutical Preparations, Serratia marcescens, Vitrectomy
Background
In intravenous (IV) drug users, the most common microorganisms isolated are
Fungal pathogens are the most common causes of endogenous endophthalmitis. The bacterial organisms most often responsible for endogenous endophthalmitis are Gram-positive organism,
The present report is about a 53-year-old man with a history of IV drug use who presented with acute loss of vision due to endophthalmitis associated with disseminated
Case Report
A 53-year-old man presented to the Emergency Department (ED) with a chief complaint of sudden loss of vision in his left eye, which had begun 5 days previously. He also complained of feeling unwell, shortness of breath, generalized body pains, fever, and chills. The patient denied headache, extremity weakness or numbness, chest pain, cough, abdominal pain, nausea, or vomiting. His medical history included chronic obstructive lung disease; an exploratory laparotomy had been done 12 years previously for a gunshot wound. He smoked tobacco daily and had a history of active, illicit IV drug use.
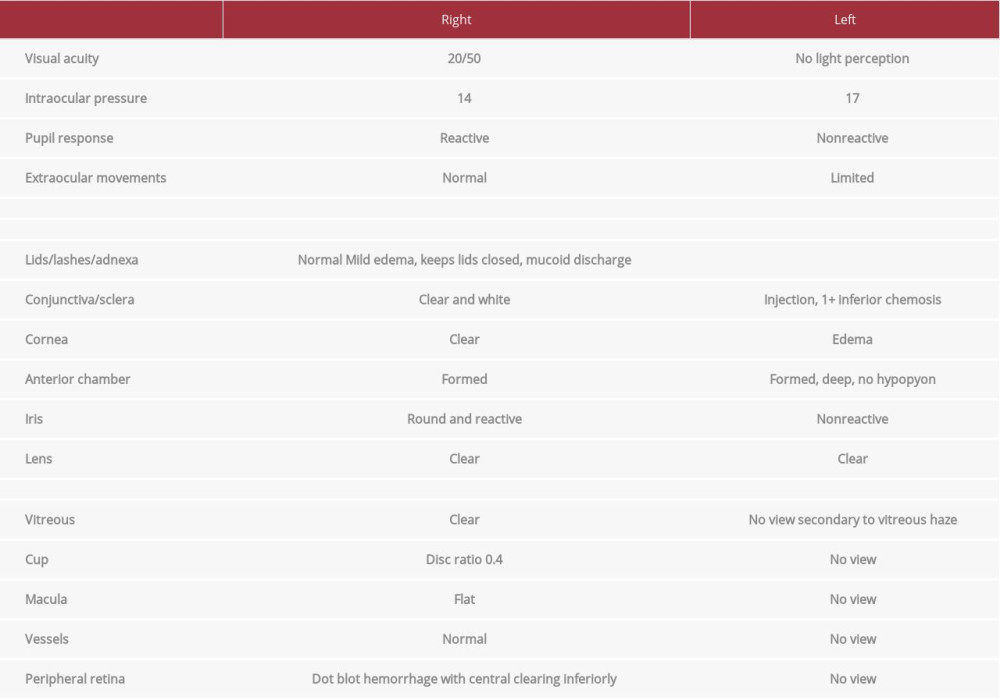
The patient’s vital signs were as follows: temperature 36.8ºC, heart rate 92 bpm, blood pressure 91/55 mmHg, respiratory rate 18 breaths/min, and oxygen saturation 92% on 2 L of oxygen. On examination, he appeared to be in moderate respiratory distress. On lung auscultation, there were diffuse bilateral rhonchi. The patient had a regular heart rate and rhythm with an ejection murmur in the tricuspid area. His abdomen was soft and nontender. An ophthalmological examination was concerning for left eye endophthalmitis. The results of the patient’s ocular examination are listed in Table 1. A photo of his left eye is shown in Figure 1.
Soon after arriving in the ED, the patient became increasingly short of breath and hypotensive. He was intubated and fluid resuscitation and vasopressor therapy were initiated. A vitreous tap was performed with injection of 0.1 mL of vancomycin (1 mg/0.1 mL) and 0.1 mL of ceftazidime (2 mg/0.1 mL). Empiric antibiotic therapy was started with IV vancomycin, cefepime, and moxifloxacin eye drops.
Laboratory studies showed an elevated white blood cell count of 28 000/mm3 (reference range, 3500–10 600/mm3), hemoglobin 8.4 g/dL (reference range, 13.3–17.1 g/dL), platelet count 210 000/mm3 (reference range, 150 000–450 000/mm3), and creatinine 1.45 mg/dL (reference range, 0.7–1.3 mg/dL); a urine drug screen was positive for benzodiazepine and opiates. Blood cultures were obtained. HIV antigen/antibody testing was nonreactive.
A chest X-ray showed left upper lobe reticulonodular infiltrates, prominent pulmonary arteries, and coarse left basilar and right upper lung zone markings. Further imaging of the thorax with a computed tomography (CT) scan showed multiple scattered cystic-like areas of opacity surrounded by consolidation, mainly in the upper lungs and periphery. The findings were suspicious for multifocal pneumonia. A CT scan of the orbit showed minimal edema of the left preseptal soft tissues with subtle thickening of the sclera of the left globe. Slight intraconal fat stranding also was identified. These findings were suspicious for concomitant preseptal and postseptal cellulitis. A transthoracic echocardiogram (TTE) showed a large, mobile echo density measuring 2.3×2.4 cm on the anterior tricuspid leaflet and a small echo density on the septal leaflet, both consistent with vegetation.
The timeline for the course of the patient’s infection is shown in Figure 2. On Day 2, blood cultures revealed
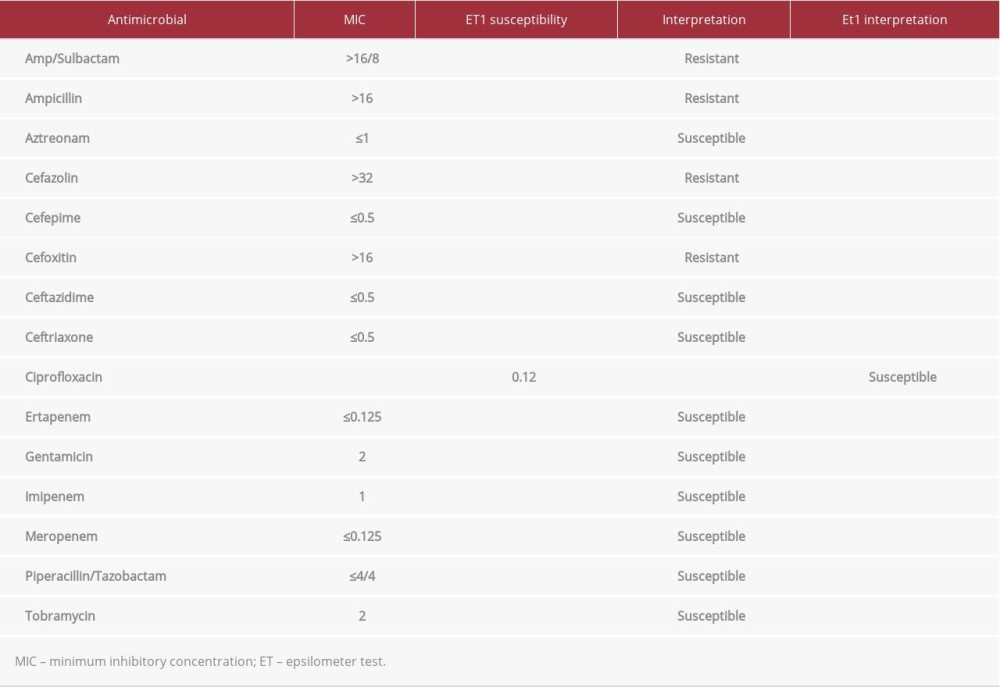
Table 2 lists the antibiotics to which the organism was susceptible. He was switched from cefepime to meropenem because
On Day 3, the patient was found to have dark red blood coming out of his nasogastric tube and his hemoglobin level was low. Gastrointestinal bleeding was suspected. An esophagogastroduodenoscopy showed esophagitis and a large amount of clotted blood in the fundus of the stomach. Four ischemic ulcers were seen, which were believed to be due to septic emboli. Two of the ulcers measured 3 to 4 cm in diameter and had black-yellow necrotic bases. In these ulcers, visible vessels were identified and hemoclips were successfully placed.
On Day 4, the patient was still critically ill in the Intensive Care Unit. A culture of vitreous taken from the patient’s left eye on the day of admission also grew
On Day 8, follow-up blood cultures became negative. An Epsilometer test showed that the
A follow-up ophthalmological examination of the patient’s left eye showed persistent chemosis and the anterior chamber was cloudy, suggesting possible progression of his infection. A repeat CT scan of the orbit showed minimal asymmetric preseptal swelling in the left periorbital soft tissue and no abnormal fluid collection. A repeat intravitreal injection of ceftazidime was performed. A B-scan ultrasound of the left eye, performed on Day 14, showed increased vitreous debris, suggesting posterior hyaloid detachment or retinal detachment, and small loculations. The patient underwent a pars plana vitrectomy on Day 15. Three ports were placed behind the limbus. Because subconjunctival pus was noted, a 360-degree conjunctival peritomy was performed. His lens was completely hazy and it was removed by phacofragmentation. The infection had eroded through the sclera and there was purulent intraocular drainage. Vitreous biopsy and anterior chamber paracentesis were performed. The iris membrane was removed with forceps and a vitrectomy was performed. Intravitreal vancomycin, 1 mg, and ceftazidime, 2.25 mg, were injected. A posterior perforation was found during the procedure; because it was not repairable, a left eye enucleation was performed on Day 19. Vitreous cultures and left eye cultures also grew
Antibiotic therapy with IV meropenem, 2 g every 8 h, and oral ciprofloxacin, 750 mg twice a day, was continued for a total of 6 weeks. The patient was discharged to a nursing home on Day 35. Unfortunately, he was lost to follow-up, but 6 months later, he was admitted to the hospital with
Discussion
Caring for patients with a history of IV drug use requires special attention because it poses unique challenges. These individuals are at high risk for developing infections with uncommon, drug-resistant organisms and septic complications. In a recent study of the epidemiology and outcomes of non-HACEK (
In the few cases of septic emboli from
Regarding management of
Conclusions
The present case was a rare instance of acute loss of vision due to endophthalmitis in a patient with a history of IV drug use which was associated with disseminated infection with the Gram-negative saprophyte
References:
1.. Phadke VK, Jacob JT: Infect Dis Clin Pract (Baltim Md), 2016; 24; 143-50
2.. Bennett JE, Dolin R, Blaser MJ: Mandell, Douglas, and Bennett’s principles and practice of infectious diseases, 2019, Elsevier
3.. Sadiq MA, Hassan M, Agarwal A, Endogenous endophthalmitis. Dagnosis, management, and prognosis: J Ophthalmic Inflamm Infect, 2015; 5; 32
4.. Breazzano MP, Jonna G, Nathan NR: Am J Ophthalmol Case Rep, 2019; 16; 100531
5.. Veve MP, McCurry ED, Cooksey GE, Epidemiology and outcomes of non-HACEK infective endocarditis in the southeast United States: PLoS One, 2020; 15; e0230199
6.. Hlaing KM, Gaballa S, Saini J: Cureus, 2020; 12; e9419
7.. Grinberg S, Bishburg E, Nagarakanti SR, Embolic serratia aortic valve endocarditis: IDCases, 2020; 22; e00953
8.. Yeung HM, Chavarria B, Shahsavari D: Case Rep Infect Dis, 2018; 2018; 5903589
9.. Shah SB, Bansal AS, Rabinowitz MP: Retin Cases Brief Rep, 2014; 8; 7-9
10.. Memon M, Raman V, Serratia marcescens endogenous endophthalmitis in an immunocompetent host: BMJ Case Rep, 2016; 2016; bcr2015209887
11.. Guo HP, Wang TJ: Am J Med Sci, 2017; 353; e7
12.. Makusha LP, Young CR, Agarwal DR: Clin Nucl Med, 2020; 45; e141-43
13.. Nath JD, Kashem A, Osman AM, Bilateral endophthalmitis and symmetrical peripheral gangrene in a patient with chronic kidney disease on maintenance hemodialysis: Saudi J Kidney Dis Transpl, 2019; 30; 715-18
14.. Baddour LM, Wilson WR, Bayer AS, Infective endocarditis in adults: Diagnosis, antimicrobial therapy, and management of complications: A scientific statement for healthcare professionals from the American Heart Association: Circulation, 2015; 132; 1435-86
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133