30 March 2021: Articles 
Bacteremia without a Focus in a Non-Immunosuppressed Patient
Rare disease
Talal Shaikhain1E, Faisal Al-Husayni12F, Sarah Al-Fawaz1ABCDEFG, Erada M. Alghamdi3EF, Abdulfattah Al-Amri345BD, Mona Alfares367AE*DOI: 10.12659/AJCR.929537
Am J Case Rep 2021; 22:e929537
Abstract
BACKGROUND: Leclercia adecarboxylata is a gram-negative rod, which is normally found in water and food. It is an emerging pathogen that affects immunocompromised patients, including patients with hematological malignancies or those receiving chemotherapy. Generally, L. adecarboxylata is considered a low-virulence pathogen with an excellent susceptibility profile, but some strains may be resistant to multiple antibiotics, such as b-lactams. Moreover, L. adecarboxylata is usually isolated as a part of polymicrobial cultures in immunocompetent individuals, but there have been cases where it was the only isolate.
CASE REPORT: A 74-year-old woman who was non-immunosuppressed and had multiple comorbidities was admitted with acute decompensated heart failure due to pneumonia. She was treated with multiple courses of antibiotics including amoxicillin-clavulanate and ciprofloxacin for pneumonia, but her infection worsened, and she had cardiopulmonary arrest. After resuscitation, she was stable for several days but suddenly became confused and hypotensive. The septic screen showed L. adecarboxylata bacteremia without a clear source, which was treated successfully with meropenem for 14 days. After the meropenem course, the patient developed diarrhea and was found to have severe Clostridium difficile infection. She did not respond to oral vancomycin and intravenous metronidazole and died.
CONCLUSIONS: This case illustrated an infection in a non-immunosuppressed individual by an organism that is considered an opportunistic pathogen, mainly affecting immunocompromised patients. The patient’s blood culture grew L. adecarboxylata, which was sensitive to all antibiotics but resolved with meropenem treatment. Owing to increasing L. adecarboxylata infections, we recommend further studies to understand the organism’s pathogenesis, risk factors, and resistance pattern.
Keywords: Bacteremia, Bacterial Infections, Carbapenems, Clostridium difficile, Immunocompetence, Anti-Bacterial Agents, Enterobacteriaceae, Enterobacteriaceae Infections, Immunocompromised Host
Background
There are a wide variety of treatment options for
Here, we are reporting a case of isolated
Case Report
A 74-year-old woman presented to the emergency department with progressive shortness of breath and productive cough for 1 week, along with increasing abdominal distention. She had a history of diabetes mellitus (with latest A1C 7.1%), hyper-tension, chronic kidney disease, heart failure with preserved ejection fraction, coronary artery bypass graft, peripheral vascular disease, and right above-knee amputation.
The patient had progressive dyspnea, associated with orthopnea, paroxysmal nocturnal dyspnea, and lower-extremity swelling. Her cough was productive with yellowish sputum, but she had no fever. The patient had recurrent abdominal as-cites secondary to heart failure (serum ascites albumin gradient of 1.5 g/L and ascitic protein of 26 g/L) for 6 months prior to presentation, which required weekly abdominal paracentesis. She was conscious and oriented, and her vital signs were stable. The examination did not show any stigmata of chronic liver disease. A cardiac examination revealed a pansystolic murmur in the left lower sternal border, while the lung examination showed fine bilateral lung crepitations. She had generalized abdominal tenderness, with tense ascites and bilateral lower-extremity pitting edema. The patient also had a stage 2 sacral ulcer, which was not infected.
The initial laboratory test results showed a normal white blood cell (WBC) count of 5.9×109 g/L. The chest X-ray at admission showed bilateral perihilar and right basilar airspace opacities, with right pleural effusion. She was admitted for acute decompensated heart failure secondary to pneumonia.
The patient was admitted to the cardiac care unit and was administered furosemide 40 mg IV twice per day and amoxicillinclavulanate. Six days after admission, the patient had a temperature of 38.9°C, with diffuse abdominal pain. Blood laboratory results showed an elevated WBC count of 13.7×109 g/L (reference range, 4.0–11.0×109), neutrophil count of 11.0×109 (reference range, 2.0–7.5×109), lymph count of 0.96×109 (reference range, 1.5–4.0×109), C-reactive protein level of 31.3 mg/L (reference range, 0.0–5.0 mg/L), and procalcitonin level of 0.42 ug/L (reference range, <0.25 ug/L). The patient was still febrile and not responding to amoxicillin-clavulanate, and was therefore switched to IV ciprofloxacin to treat the sepsis. A few hours later, the patient went into cardiac arrest. Cardiopulmonary resuscitation was performed for 3 minutes, a peripherally inserted central catheter (PICC) was inserted, and the patient was intubated.
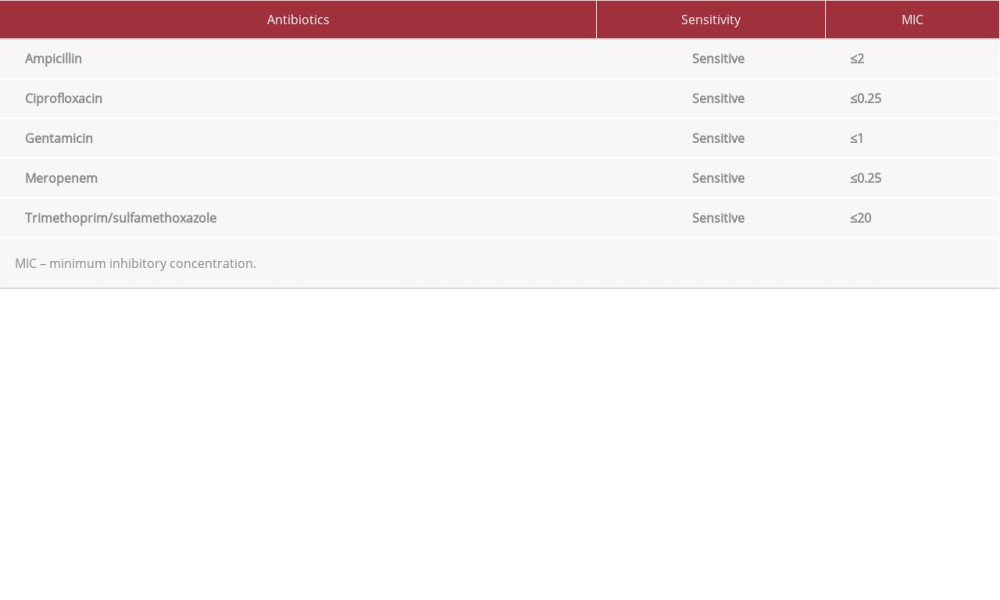
After 3 days, the patient was extubated and paracentesis was performed. Ascitic fluid culture results were negative but had a cell count of 196 leukocytes/µL. Two days later, the patient became confused and hypotensive and a laboratory examination showed that the inflammatory markers were elevated. A full septic screen was done and exhibited no organisms in the urine, sputum, or cerebrospinal fluid; however, a superficial swab from the sacral ulcer, which did not show signs of infection (Figure 1), grew
After 48 h of meropenem, her fever abated, inflammatory markers improved, and
Discussion
In case reports and series,
Bacteremia caused by
There are no guidelines discussing treatment, but good response to β-lactams and fluoroquinolones was observed in multiple reports [7,12]. Our patient cleared her bacteremia with a brief course of meropenem, suggesting this antibiotic might be a good option to treat
Conclusions
Figures
References:
1.. Armentrout R, Brown R, Molecular cloning of genes for cellobiose utilization and their expression in Escherichia coli: Appl Environ Microbiol, 1981; 41; 1355-62
2.. Tamura K, Sakazaki R, Kosako Y, Yoshizaki E: Curr Microbiol, 1986; 13; 179-98
3.. Sarma P, Bhattacharya D, Krishnan S, Lal B: Appl Environ Microbiol, 2004; 70; 3163-66
4.. Stock I, Burak S, Wiedemann B: Clin Microbiol Infect Dis, 2004; 10; 724-33
5.. Mazzariol A, Zuliani J, Fontana R, Cornaglia G: J Clin Microbiol, 2003; 41; 1738-39
6.. Alosaimi R, Muhmmed Kaaki M, Catheter-related ESBL-producing Leclercia adecarboxylata septicemia in hemodialysis patient: An emerging pathogen?: Case Rep Infect Dis, 2020; 2020; 7403152
7.. Spiegelhauer M, Andersen P, Frandsen T: Infect Dis, 2018; 51; 179-88
8.. Temesgen Z, Toal D, Cockerill F: Clin Infect Dis, 1997; 25; 79-81
9.. Forrester J, Adams J, Sawyer R: Surg Infect (Larchmt), 2012; 13; 63-66
10.. Rodriguez J, Sanchez F, Gutierrez N, Garcia J, Garcia-Rodriguez J: Enferm Infecc Microbiol Clin, 2001; 19; 237-38
11.. Sawamura H, Kawamura Y, Yasuda M: Kansenshogaku Zasshi, 2005; 79; 831-35 [in Japanese]
12.. Kim H, Chon C, Ahn S: Int J Clin Pract, 2008; 68; 1294-98
13.. Hess B, Burchett A, Huntington M: J Med Microbiol, 2008; 57(7); 896-98
14.. Yehia H, Antimicrobial resistance patterns of Enterobacteriaceae and non-Enterobacteriaceae isolated from poultry intestinal: Life Sci J, 2013; 10; 3438-46
15.. Al-Holy M, Osaili T, El-Sayed S, Microbiological quality of leafy green vegetables sold in the local market of Saudi Arabia: Ital J Food Sci, 2013; 25; 446-52
16.. Osaili T, Alaboudi A, Al-Quran H, Al-Nabulsi A, Decontamination and survival of Enterobacteriaceae on shredded iceberg lettuce during storage: Food Microbiol, 2018; 73; 129-36
17.. Anuradha M: J Clin Diagn Res, 2014; 8(12); DD03-4
18.. Fattal O, Deville J: PediatrNephrol, 2014; 15(3–4); 186-87
19.. Ghosh R, Misra R, Prasad K, Prasad N: Int J Res Med Sci, 2016; 4(4); 1254-56
20.. Adapa S, Konala V, Nawaz F: Clin Case Rep, 2019; 7; 829-31
21.. Rayfield E, Ault M, Keusch G, Infection and diabetes: The case for glucose control: Am J Med, 1982; 72(3); 439-50
22.. Lee N, Ki C, Kang W: Korean J Infect Dis, 1999; 31; 167-70
23.. De La Obra P, Domingo D, Casaseca R: Clin Microbiol Newsl, 1999; 21; 142-43
24.. Botero-Garcia C, Gomez C, Bravo J: Infect, 2018; 22; 223-26
25.. Beltran A, Vicente A, Capilla S: Med Clin (Barc), 2004; 122; 159 [in Spanish]
26.. Ando A, Majewski L, Kajioka E: Open Forum Infect Dis, 2016; 3(Suppl.1); 1096
27.. de Baere T, Wauters G, Huylenbroeck A: J Clin Microbiol, 2001; 39(4); 1674-75
28.. Papagiannitsis C, Studentová V, Hrabák J: Antimicrob Agents Chemother, 2013; 57(6); 2896-97
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133