06 March 2021: Articles 
Gastrointestinal Stromal Tumor in Monozygotic Twins Shows Distinct Mutational Status: A Case Report
Unusual clinical course, Challenging differential diagnosis, Rare coexistence of disease or pathology
Marcelo Sobral-Leite12ABDEF, Jesse Lopes da Silva34BDEF, Haynna Kimie Pimenta-Inada3BCF, Andrea Sobral Mendes Boisson4BF, Priscila de Almeida Romeiro5BF, Fernando Mallet Soares Parago4BF, Marcus Vinicius M. Valadão da Silva6BF, Andreia Cristina de Melo3ABDEFG*DOI: 10.12659/AJCR.929887
Am J Case Rep 2021; 22:e929887
Abstract
BACKGROUND: Gastrointestinal stromal tumors (GISTs) are rare mesenchymal cancers that affect the gastrointestinal tract and are most often located in the stomach and proximal small intestine. The most common molecular genetic abnormalities underlying GIST carcinogenesis are mutations in the tyrosine kinase gene (KIT) and in the platelet-derived growth factor receptor alpha (PDGFRA) gene. To the best of our knowledge, no cases have been reported so far of synchronous diagnosis of GIST in 2 monozygotic twins presenting with clinical and morphological features of sporadic disease.
CASE REPORT: This report presents the cases of 2 monozygotic twin sisters who were diagnosed with GIST at the same age and who had different KIT exon 11 tumor mutational statuses. In the current report, the screening examination that led to early detection of GIST in one of the sisters was not motivated by any symptom, but by a GIST diagnosis in her twin a few days before. The literature was reviewed for pathological and molecular features associated with prognosis and treatment response. Furthermore, we identified identical genotypes of KIT and PDGFRA polymorphisms in the DNA of both tumors that might be present in the germline DNA. The present case supports the implementation of specific cancer screening in the context of monozygotic twins, regardless of identification of the genetic components involved.
CONCLUSIONS: Our report suggests that monozygotic twins with GIST can have different mutational statuses for KIT and PDGFRA. Referral for special screening should be considered for individuals who have a monozygotic twin diagnosed with cancer.
Keywords: Diseases in Twins, gastrointestinal stromal tumors, Proto-Oncogene Proteins c-kit, DNA Mutational Analysis, Mutation, Receptor, Platelet-Derived Growth Factor alpha, Twins, Monozygotic
Background
Gastrointestinal stromal tumor (GIST) is a cancer with mesenchymal origin in the interstitial cells of Cajal, which belongs to the intestinal autonomic nervous system as a smooth muscle pacemaker [1]. GIST was introduced as a specific disease in 1983, and today it is the mesenchymal tumor most often seen in the gastrointestinal tract [2–4]. Most population-based studies report an incidence of approximately 10 to 15 cases of GISTs per million inhabitants [4].
In 75% to 80% of cases of GIST, the type III receptor tyrosine kinase protein, c-KIT (or CD117), is overexpressed [5,6]. Another receptor tyrosine kinase, the platelet-derived growth factor receptor alpha (PDGRF-α), is expressed in 5% to 10% of cases of GIST [7]. c-KIT and PDGRF-α appear to be the main oncogenic drivers involved in GIST carcinogenesis [8]. Thus, immunohistochemistry (IHC) for c-KIT and PDGRF-α needs to be performed as part of routine diagnosis [6].
Most GISTs are resistant to chemotherapy and the introduction of imatinib mesylate (IM), which targets and inhibits c-KIT, significantly improved overall patient survival [9,10]. Furthermore, the identification of mutations in the c-KIT (
A number of families with multiple inherited GIST syndromes (MIM #606764) have been described. These syndromes are rare and caused by inherited molecular defects that affect the entire organism [16]. The Carney triad is characterized by gastric GIST, paraganglioma, pulmonary chondroma, and loss of expression of succinate dehydrogenase subunit B but not mutations in
The present case report describes 2 monozygotic twin sisters who were diagnosed with GIST at the same age and who had differing tumor mutation statuses in exon 11 of the
Case Reports
CASE 1:
The patient in Case 1 was a 62-year-old woman. In 2011, she presented with a urinary tract infection after experiencing a fever intermittently for 1 week. On clinical examination, a pelvic mass was identified. The patient underwent pelvic and abdominal computed tomography (CT), which revealed a homogeneous soft tissue mass in the pelvis that measured 11.0×11.0×11.0 cm and liver lesions suggestive of metastases (Figure 1). An ultrasound-guided biopsy of the pelvis then was performed. Pathological analysis of the material from the patient’s small intestine revealed morphological features of GIST and IHC staining was positive for c-KIT.
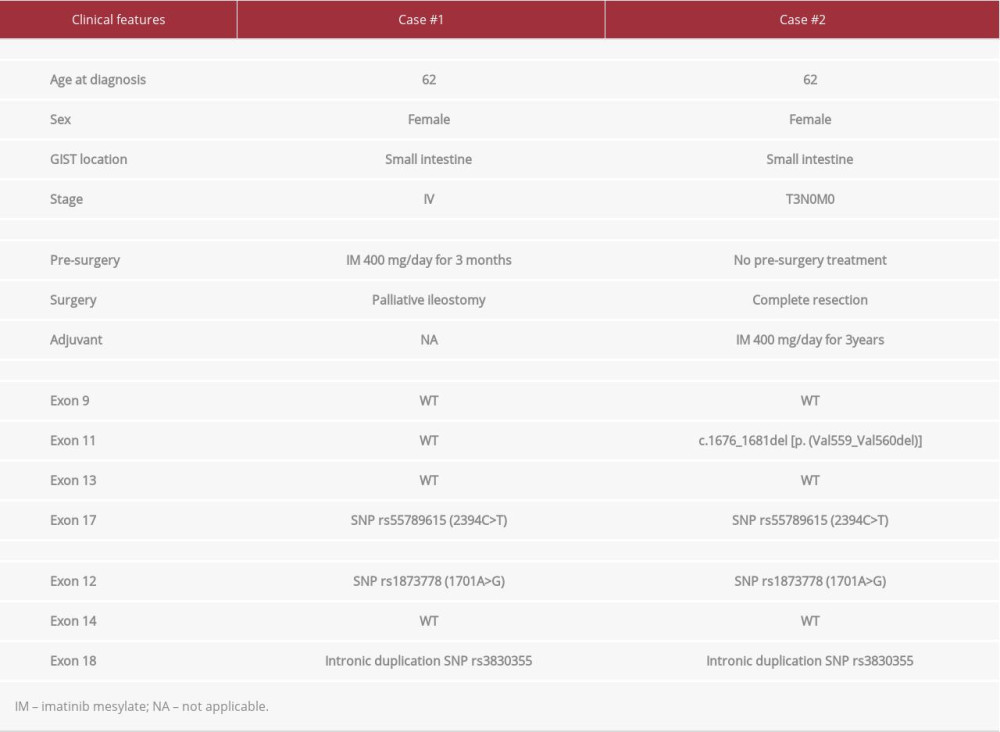
Initially, the patient was prescribed systemic therapy with 400 mg of IM daily. On Day 35 of treatment, she was admitted to the hospital with anemia and abdominal distension, and rapidly developed intestinal obstruction. New CT scans showed a significant increase in the pelvic mass with intestinal obstruction. An emergency ileostomy was performed. Fortunately, at that time, a sample was taken from the abdominal tumor and cryopreserved. Molecular analysis of this GIST sample showed no deletion or missense mutation in KIT or PDGFRA (Table 1). After the ileostomy, the patient was treated in the Intensive Care Unit for septic shock; mechanical ventilation and vasoactive amines were required. A few days later, her clinical condition deteriorated and she died.
CASE 2:
The patient in Case 2 was the monozygotic twin sister of the patient in Case 1. In 2001, she was diagnosed with breast cancer, subtype luminal B, and was treated with neoadjuvant chemotherapy, mastectomy, adjuvant radiotherapy, and hormonal therapy. During clinical surveillance in November 2011, she underwent pelvic and abdominal ultrasound as a screening examination; the motivation for it was the GIST diagnosis in her sister. The imaging revealed a pelvic mass measuring 8.5×6.5 cm, which prompted an exploratory laparotomy with optimal resection. The pathological findings from the surgical specimen were a GIST measuring 9×6×6 cm, with 19 mitoses/5 mm2. The tumor, which stained positive with IHC for c-KIT, was located in the small intestine. Genomic analysis of the specimen revealed a mutation in exon 11 of the KIT gene: c.1676_1681del [p. (Val559_Val560del)] (Table 1).
Beginning in March 2012, the patient was treated with adjuvant therapy with IM (400 mg daily). She completed 3 years of therapy with mild adverse effects, reporting only recurrent nausea.
Interestingly, in both Case 1 and Case 2, the genotyping of the tumors showed the same polymorphisms in
Six years after the GIST diagnosis, the patient in Case 2 reported abdominal pain. A CT scan demonstrated a solid nodule in her pelvis, attached to the sigmoid, which measured 3.3×2.8 cm.
In July 2018, laparotomy was performed and the lesion was completely resected. The pathology report confirmed recurrence of the GIST. IM was restarted in August 2018 and the patient is containing to take it and remains free of recurrence.
Discussion
This is the first report of GIST diagnosed concomitantly at a similar age in monozygotic twins. Analysis of the tumor molecular pathology, genetics, treatment response, and outcomes in the 2 related patients have resulted in insights about cancer screening and the carcinogenesis of GIST.
To the best of our knowledge, current guidelines for familial GIST screening [35] do not mention screening the other twin if there is no strong family history of the disease. Conclusions about cancer incidence in twins and first-degree relatives of individuals with GIST versus the general population, reported in familial studies, have proven controversial [36–38]. The observations are consistent enough, however, to have led to suspicion about the involvement of a heritable genetic component in the development of the malignancies [31, 39], whereas environmental factors are likely to play an important role in the carcinogenesis of sporadic cancers [40]. Nevertheless, long-term follow-up of a prospective study with Nordic twins (n=80 309 monozygotic twins) estimated an increase in absolute cumulative cancer risk of 14% (95%CI 12–16%) in monozygotic twins diagnosed with cancer as compared with the overall cohort, and 38% of twins were diagnosed with the same type of cancer [41]. In the present report, the screening with abdominal and pelvic ultrasound that led to diagnosis of GIST in Case 2 was motivated by the previous diagnosis in Case 1. Because it facilitated prompt diagnosis, surveillance may be warranted for early detection of asymptomatic disease. It should be noted that although the clinical records for Case 2 included a complete description of the findings from the first CT scans, which identified the primary GIST, the images themselves were unavailable.
Regarding Case 1, the later analysis of the tumor mutational profile showed no pathogenic mutations in KIT exons. That could explain the rapid progression of the advanced disease soon after the patient started treatment with IM. In most cases, patients with wild-type GIST have limited sensitivity to IM [42,43]. In the present case, knowing the patient’s mutational status earlier would not have changed treatment decisions because neoadjuvant therapy is recommended prior to extended resection [44].
A deletion in
Syndromic GISTs account for approximately 3% to 4% of all GIST cases and their clinicopathological characteristics are different from the sporadic cases [16]. The peculiar clinical and pathological features of the Carney triad and Carney-Stratakis syndrome – early age at diagnosis, mainly gastric GISTs, multiple neoplasms, and lack of c-KIT expression – do not correspond to the disease descriptions for Case 1 or Case 2, which involved older patients who had non-gastric GISTs and high expression of c-KIT [17,18]. It is important to note that the familial Carney syndrome has been described in 2 monozygotic twins brothers, who were aged 12 and 13 years old, respectively [53].
In recent years, some familial GISTs with germline mutations in
The involvement of
Appropriately designed genomic studies should be considered to search for germline polymorphisms associated with sporadic GISTs and investigate germline variations in
Conclusions
The description of the twin cases in the present report raises new questions about screening, diagnosis, and treatment of and prognosis in patients with cancer who are monozygotic twins. Even though different clinical stages could explain the different outcomes in our 2 patients, we have demonstrated that the mutational status of
References:
1.. Kindblom LG, Remotti HE, Aldenborg F, Gastrointestinal pacemaker cell tumor (GIPACT): Gastrointestinal stromal tumors show phenotypic characteristics of the interstitial cells of Cajal: Am J Pathol, 1998; 152(5); 1259-69
2.. Stout AP, Bizarre smooth muscle tumors of the stomach: Cancer, 1962; 15; 400-9
3.. Mazur MT, Clark HB, Gastric stromal tumors. Reappraisal of histogenesis: Am J Surg Pathol, 1983; 7(6); 507-19
4.. Søreide K, Sandvik OM, Søreide JA, Global epidemiology of gastrointestinal stromal tumours (GIST):A systematic review of population-based cohort studies: Cancer Epidemiol, 2016; 40; 39-46
5.. Sarlomo-Rikala M, Kovatich AJ, Barusevicius A, CD117: A sensitive marker for gastrointestinal stromal tumors that is more specific than CD34: Mod Pathol, 1998; 11(8); 728-34
6.. Rubin BP, Heinrich MC, Genotyping and immunohistochemistry of gastrointestinal stromal tumors: An update: Semin Diagn Pathol, 2015; 32(5); 392-99
7.. Heinrich MC, Corless CL, Duensing A, PDGFRA activating mutations in gastrointestinal stromal tumors: Science, 2003; 299(5607); 708-10
8.. Hirota S, Isozaki K, Moriyama Y, Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors: Science, 1998; 279(5350); 577-80
9.. Joensuu H, Roberts PJ, Sarlomo-Rikala M, Effect of the tyrosine kinase inhibitor STI571 in a patient with a metastatic gastrointestinal stromal tumor: N Engl J Med, 2001; 344(14); 1052-56
10.. Cioffi A, Maki RG, GI stromal tumors: 15 years of lessons from a rare cancer: J Clin Oncol, 2015; 33(16); 1849-54
11.. Lasota J, Miettinen M, KIT and PDGFRA mutations in gastrointestinal stromal tumors (GISTs): Semin Diagn Pathol, 2006; 23(2); 91-102
12.. Koumarianou A, Economopoulou P, Katsaounis P, Gastrointestinal Stromal tumors (GIST):A prospective analysis and an update on biomarkers and current treatment concepts: Biomark Cancer, 2015; 7(Suppl. 1); 1-7
13.. Valadão M, Braggio D, Santos AF, Involvement of signaling molecules in the prediction of response to imatinib treatment in metastatic GIST patients: J Surg Res, 2012; 178(1); 288-93
14.. Ahmad F, Lad P, Bhatia S, Molecular spectrum of c-KIT and PDGFRA gene mutations in gastro intestinal stromal tumor: Determination of frequency, distribution pattern and identification of novel mutations in Indian patients: Med Oncol Northwood, 2014; 32(1); 424
15.. Wada R, Arai H, Kure S, “Wild type” GIST: Clinicopathological features and clinical practice: Pathol Int, 2016; 66(8); 431-37
16.. Ricci R, Syndromic gastrointestinal stromal tumors: Hered Cancer Clin Pract, 2016; 14; 15
17.. Carney JA, Sheps SG, Go VL, The triad of gastric leiomyosarcoma, functioning extra-adrenal paraganglioma and pulmonary chondroma: N Engl J Med, 1977; 296(26); 1517-18
18.. Carney JA, Stratakis CA, Familial paraganglioma and gastric stromal sarcoma: A new syndrome distinct from the Carney triad: Am J Med Genet, 2002; 108(2); 132-39
19.. Nishida T, Hirota S, Taniguchi M, Familial gastrointestinal stromal tumours with germline mutation of the KIT gene: Nat Genet, 1998; 19(4); 323-24
20.. Hirota S, Okazaki T, Kitamura Y, Cause of familial and multiple gastrointestinal autonomic nerve tumors with hyperplasia of interstitial cells of Cajal is germline mutation of the c-kit gene: Am J Surg Pathol, 2000; 24(2); 326-27
21.. Isozaki K, Terris B, Belghiti J, Germline-activating mutation in the kinase domain of KIT gene in familial gastrointestinal stromal tumors: Am J Pathol, 2000; 157(5); 1581-85
22.. Beghini A, Tibiletti MG, Roversi G, Germline mutation in the juxta-membrane domain of the kit gene in a family with gastrointestinal stromal tumors and urticaria pigmentosa: Cancer, 2001; 92(3); 657-62
23.. O’Riain C, Corless CL, Heinrich MC, Gastrointestinal stromal tumors: Insights from a new familial GIST kindred with unusual genetic and pathologic features: Am J Surg Pathol, 2005; 29(12); 1680-83
24.. Carballo M, Roig I, Aguilar F, Novel c-KIT germline mutation in a family with gastrointestinal stromal tumors and cutaneous hyperpigmentation: Am J Med Genet A, 2005; 132A(4); 361-64
25.. Bachet J-B, Landi B, Laurent-Puig P, Diagnosis, prognosis and treatment of patients with gastrointestinal stromal tumour (GIST) and germ-line mutation of KIT exon 13: Eur J Cancer, 2013; 49(11); 2531-41
26.. Jones DH, Caracciolo JT, Hodul PJ, Familial gastrointestinal stromal tumor syndrome: Report of 2 cases with KIT exon 11 mutation: Cancer Control, 2015; 22(1); 102-8
27.. Chompret A, Kannengiesser C, Barrois M, PDGFRA germline mutation in a family with multiple cases of gastrointestinal stromal tumor: Gastroenterology, 2004; 126(1); 318-21
28.. Zöller ME, Rembeck B, Odén A, Malignant and benign tumors in patients with neurofibromatosis type 1 in a defined Swedish population: Cancer, 1997; 79(11); 2125-31
29.. Rubin BP, Heinrich MC, Corless CL, Gastrointestinal stromal tumour: Lancet, 2007; 369(9574); 1731-41
30.. Postow MA, Robson ME, Inherited gastrointestinal stromal tumor syndromes: Mutations, clinical features, and therapeutic implications: Clin Sarcoma Res, 2012; 2(1); 16
31.. Ahlbom A, Lichtenstein P, Malmström H, Cancer in twins: Genetic and nongenetic familial risk factors: J Natl Cancer Inst, 1997; 89(4); 287-93
32.. Zhang P, Lehmann BD, Shyr Y, The utilization of formalin fixed-paraffin-embedded specimens in high throughput genomic studies: Int J Genomics, 2017; 2017; 1926304
33.. McBride LJ, Koepf SM, Gibbs RA, Automated DNA sequencing methods involving polymerase chain reaction: Clin Chem, 1989; 35(11); 2196-201
34.. Schwartz LH, Litière S, de Vries E, RECIST 1.1 – Update and Clarification: From the RECIST Committee: Eur J Cancer, 2016; 62; 132-37
35.. von Mehren M, Randall RL, Benjamin RS, Gastrointestinal stromal tumors, version 2.2014: J Natl Compr Cancer Netw, 2014; 12(6); 853-62
36.. Chen L, Cnattingius S, Iliadou AN, Cancer risks in twins and singletons from twin and non-twin families: Int J Cancer, 2016; 138(5); 1102-10
37.. Goldgar DE, Easton DF, Cannon-Albright LA, Systematic population-based assessment of cancer risk in first-degree relatives of cancer pro-bands: J Natl Cancer Inst, 1994; 86(21); 1600-8
38.. Rawal R, Bertelsen L, Olsen JH, Cancer incidence in first-degree relatives of a population-based set of cases of early-onset breast cancer: Eur J Cancer, 2006; 42(17); 3034-40
39.. Hemminki K, Vaittinen P, Familial cancers in a nationwide family cancer database: Age distribution and prevalence: Eur J Cancer, 1999; 35(7); 1109-17
40.. Lichtenstein P, Holm NV, Verkasalo PK, Environmental and heritable factors in the causation of cancer – analyses of cohorts of twins from Sweden, Denmark, and Finland: N Engl J Med, 2000; 343(2); 78-85
41.. Mucci LA, Hjelmborg JB, Harris JR, Familial risk and heritability of cancer among twins in Nordic countries: JAMA, 2016; 315(1); 68-76
42.. Rutkowski P, Gronchi A, Hohenberger P, Neoadjuvant imatinib in locally advanced gastrointestinal stromal tumors (GIST):The EORTC STBSG experience: Ann Surg Oncol, 2013; 20(9); 2937-43
43.. Gronchi A, Risk stratification models and mutational analysis: Keys to optimising adjuvant therapy in patients with gastrointestinal stromal tumour: Eur J Cancer, 2013; 49(4); 884-92
44.. Ford SJ, Gronchi A, Indications for surgery in advanced/metastatic GIST: Eur J Cancer, 2016; 63; 154-67
45.. Corless CL, Barnett CM, Heinrich MC, Gastrointestinal stromal tumours: Origin and molecular oncology: Nat Rev Cancer, 2011; 11(12); 865-78
46.. Cerski MR, Pereira F, Matte US, Exon 11 mutations, Ki67, and p16(INK4A) as predictors of prognosis in patients with GIST: Pathol Res Pract, 2011; 207(11); 701-6
47.. Emile J-F, Théou N, Tabone S, Clinicopathologic, phenotypic, and genotypic characteristics of gastrointestinal mesenchymal tumors: Clin Gastroenterol Hepatol, 2004; 2(7); 597-605
48.. Zhi X, Zhou X, Wang W, The utilization of formalin fixed-paraffin-embedded specimens in high throughput genomic studies Practical role of mutation analysis for imatinib treatment in patients with advanced gastrointestinal stromal tumors: A meta-analysis: PLoS One, 2013; 8(11); e79275
49.. Joensuu H, Hohenberger P, Corless CL, Gastrointestinal stromal tumour: Lancet, 2013; 382(9896); 973-83
50.. Joensuu H, Risk stratification of patients diagnosed with gastrointestinal stromal tumor: Hum Pathol, 2008; 39(10); 1411-19
51.. Zhang H, Liu Q, Prognostic indicators for gastrointestinal stromal tumors: A review: Transl Oncol, 2020; 13(10); 100812
52.. Joensuu H, Eriksson M, Hall KS, Survival outcomes associated with 3 years vs 1 year of adjuvant imatinib for patients with high-risk gastrointestinal stromal tumors: An analysis of a randomized clinical trial after 10-year follow-up: JAMA Oncol, 2020; 6(8); 1241-46
53.. Boccon-Gibod L, Boman F, Boudjemaa S, Separate occurrence of extra-adrenal paraganglioma and gastrointestinal stromal tumor in monozygotic twins: Probable familial Carney syndrome: Pediatr Dev Pathol, 2004; 7(4); 380-84
54.. Thalheimer A, Schlemmer M, Bueter M, Familial gastrointestinal stromal tumors caused by the novel KIT exon 17 germline mutation N822Y: Am J Surg Pathol, 2008; 32(10); 1560-65
55.. Li FP, Fletcher JA, Heinrich MC, Familial gastrointestinal stromal tumor syndrome: Phenotypic and molecular features in a kindred: J Clin Oncol, 2005; 23(12); 2735-43
56.. Pasini B, Matyakhina L, Bei T, Multiple gastrointestinal stromal and other tumors caused by platelet-derived growth factor receptor alpha gene mutations: A case associated with a germline V561D defect: J Clin Endocrinol Metab, 2007; 92(9); 3728-32
57.. Ricci R, Martini M, Cenci T, PDGFRA-mutant syndrome: Mod Pathol, 2015; 28(7); 954-64
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133