08 May 2021: Articles 
Rhabdomyolysis Following Recovery from Severe COVID-19: A Case Report
Rare coexistence of disease or pathology
Julie Byler1E*, Rebecca Harrison2E, Lindsey L. Fell3BDOI: 10.12659/AJCR.931616
Am J Case Rep 2021; 22:e931616
Abstract
BACKGROUND: Rhabdomyolysis occurs when muscle injury leads to the release of muscle cell constituents into circulation, often leading to significant systemic complications. There are many causes of rhabdomyolysis, and the etiology is often multifactorial or unclear. Current data suggest that acute COVID-19 may cause muscle injury that can lead to rhabdomyolysis, particularly in cases of severe illness requiring prolonged hospitalization; however, data on the long-term effects of COVID-19 on the musculoskeletal system are lacking.
CASE REPORT: We present a case of a woman with generalized weakness 1 week following discharge from the hospital after a prolonged admission for severe COVID-19. She was found to have acute kidney injury and elevated creatine kinase (CK) of 1775 U/L (normal 36-234 U/L). Her home medications, including her statin, were held, but her CK continued to rise, peaking at 15 085 U/L, and she developed renal failure necessitating renal replacement therapy. A thorough work-up for the underlying etiology of her rhabdomyolysis was pursued, including testing for autoimmune myositis, statin-associated necrotizing autoimmune myositis, and a muscle biopsy, which were all unrevealing. Ultimately, the patient’s rhabdomyolysis was determined to likely be secondary to a post-viral myopathy from COVID-19. A toxic myopathy from medication use or a delayed critical illness myopathy from her recent prolonged hospitalization could have also contributed.
CONCLUSIONS: This case highlights the wide differential diagnosis of rhabdomyolysis in the setting of recent COVID-19 and prolonged hospitalization. It demonstrates the possibility that muscle injury and resultant rhabdomyolysis may be a late complication of COVID-19 that is not yet fully described in the literature.
Keywords: COVID-19, myositis, rhabdomyolysis, COVID-19, Muscular Diseases, SARS-CoV-2
Background
Rhabdomyolysis occurs when muscle injury leads to the release of muscle cell constituents into circulation, often leading to significant systemic complications including acute kidney injury. It is characterized by an acute elevation of creatine kinase (CK), and often concurrently leads to electrolyte abnormalities, acute kidney injury, and myoglobinuria.
There are many different causes of rhabdomyolysis, and the etiology is often multifactorial or unclear. The most common cause of rhabdomyolysis is trauma and these cases are often easy to diagnose, but non-exertional, non-traumatic etiologies, which include medications, endocrinopathies, infections, inflammatory myopathies, and critical illness myopathies, are often more difficult to detect. Determining the cause of rhabdomyolysis and identifying factors associated with it is vital for early recognition and treatment of this important clinical entity.
It is known that many viral illnesses, including influenza, para-influenza, adenovirus, and HIV, can cause a viral myositis that in severe cases can lead to rhabdomyolysis [1,2]. Preliminary case report and retrospective data suggests that COVID-19 may also be associated with an acute viral myositis that has the potential to cause rhabdomyolysis, but more data on the long-term effects of COVID-19 on the musculoskeletal and neurologic system are needed.
Several case reports have been published that describe rhabdomyolysis or acute myositis as an initial manifestation of COVID-19 [3–10] and numerous studies have noted that myalgias are a common presentation of COVID-19 [11]. Orsucci et al described the various neurologic effects of COVID-19 and explored skeletal muscle injury as a neurologic effect of the disease. They described a study of 214 patients from Wuhan, China, where 36% of SARS-CoV-2-positive patients had neurologic symptoms, with 11% of these being related to skeletal muscle injury; however, more research is needed to replicate and confirm these findings [9,10]. There have also been numerous case reports on myopathies associated with severe COVID-19 and prolonged ICU stays, which are likely related to at least an element of critical illness myopathy, with a potential additional role of direct toxic viral effects, or inflammation [11–14]. However, there are very limited data on the long-term effects of COVID-19, and only a few case reports of rhabdomyolysis as a late complication of COVID-19 have been published [15,16]. Here, we present a case of a woman with rhabdomyolysis following recovery from severe COVID-19.
Case Report
A 67-year-old woman with a past medical history of type II diabetes, hypertension, and hyperlipidemia on statin therapy presented to an outside hospital reporting generalized weakness and nausea 1 week after discharge from a 1-month admission for severe COVID-19.
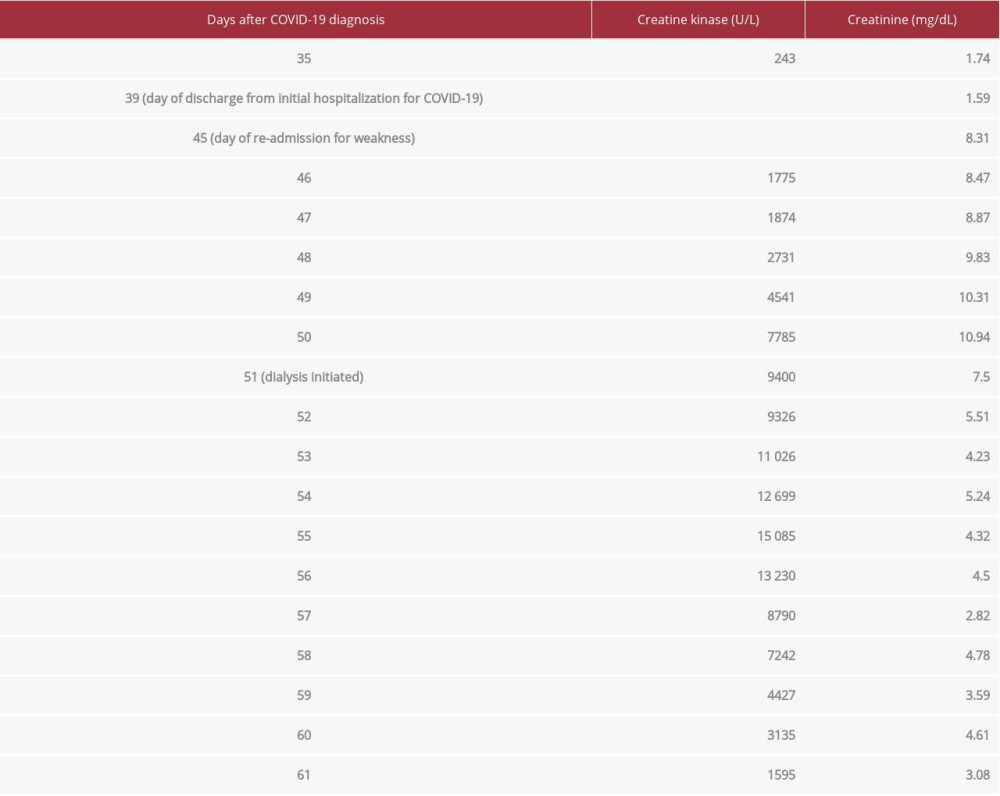
The patient initially presented to an outside urgent care department reporting 3 days of fatigue, chills, and chest tightness, found to be positive for SARS-CoV-2 (COVID-19) by nucleic acid amplification. She was noted to be febrile at the time, but was otherwise hemodynamically stable and oxygenating well on room air, so she was discharged home. She then re-presented to the Emergency Department approximately 1 week later with progressive COVID-19 symptoms, including worsening shortness of breath and cough. A chest X-ray revealed extensive bilateral pulmonary opacities and she developed worsening hypoxemic respiratory failure, necessitating intubation and admission to the Intensive Care Unit. She was given dexamethasone 6 mg daily for 7 days (stopped early due to an up-trending blood urea nitrogen [BUN]) as well as convalescent plasma for her severe COVID-19. Remdesivir was held due to liver function abnormalities. She had an acute kidney injury on arrival, attributed to poor oral intake and acute illness, which subsequently normalized. However, later in her hospital stay, she again had worsening of her renal function and a broad work-up was initiated, including a CK that was only mildly elevated at 243 U/L (normal range 38–234 U/L) (Table 1, Figure 1). Ultimately, her kidney injury was thought to be likely secondary to acute tubular necrosis (ATN), and it gradually improved with supportive care. She required prolonged intubation and a tracheostomy, but her respiratory status improved and she was decannulated 3 weeks after her initial intubation and discharged home. At the time of discharge, her kidney injury had improved but not completely resolved, with a creatinine of 1.59 mg/dL.
One week following her discharge, she developed nausea, vomiting, and generalized weakness, leading her to re-present to the Emergency Department. Her physical exam was unre-markable except for mild anterior thigh tenderness and symmetric, proximal weakness of her bilateral lower extremities. Sensation and reflexes were intact. Laboratory work-up demonstrated an acute kidney injury with a creatinine of 8.31 mg/dL (normal 0.60–1.10 mg/dL), an elevated CK of 1775 U/L, elevated AST at 749 U/L (normal 8–39 U/L), and ALT of 337 U/L (normal 10–35 U/L). TSH was normal. A urinalysis showed 3+ blood with 10–20 RBCs per high-power field. Urine myoglobin was unfortunately not obtained; however, with the patient’s elevated CK and kidney injury in the setting of myalgias and weakness, she met the criteria for severe rhabdomyolysis by most available definitions, despite lack of definitive evidence of myoglobulinuria. There was no history of trauma or of an exertional etiology of her rhabdomyolysis. She had no signs or symptoms of systemic bacterial infection, and was retested for SARS-CoV-2, which was negative. Recent medication and drug exposures that could have contributed were explored.
Her ethanol level was negative. She had received dexamethasone during her prior hospitalization, but had been off steroids for approximately 1 month at the time of her presentation. Her home medications included a statin, and this was held out of concern for a statin-induced myopathy. She was started on intravenous fluid for treatment of her rhabdomyolysis. Unfortunately, despite this, her CK up-trended and her acute kidney injury progressed, eventually requiring renal replacement therapy (Table 1, Figure 1). Given this, a broader work-up for alternative causes of her rhabdomyolysis was initiated, including testing for an autoimmune myositis. Her ANA was positive at 1: 320 in a speckled pattern. Ro, La, RNP, anti-Sm, Scl-70, Jo-1, and dsDNA antibodies were negative and complement levels were mildly elevated. She was treated empirically for autoimmune myositis while her autoimmune myositis panel was pending with 60 mg of prednisone daily, followed by 1000 mg i.v. methylprednisolone. CK remained elevated, peaking at 15 085 U/L (Table 1, Figure 1). Given her lack of improvement and the elusive etiology of her rhabdomyolysis, the patient was transferred for higher level of care.
Additional work-up demonstrated a negative autoimmune myositis panel and a negative HMGCR antibody, ruling out statin-associated necrotizing autoimmune myositis. Bilateral quadriceps MRI was obtained and demonstrated diffuse, bilateral myositis (Figure 2A, 2B). A muscle biopsy of her right vastus lateralis was completed and showed a non-specific, pauci-immune necrotizing myopathy. There was no definitive evidence for an immune-mediated component and, given the biopsy findings, a necrotizing autoimmune myopathy was thought to be less likely. She was treated with supportive care and aggressive Physical Therapy and her CK eventually down-trended (Table 1, Figure 1). Unfortunately, she did not have renal recovery and continued to require hemodialysis at time of discharge.
Ultimately, the patient’s rhabdomyolysis was determined to be likely secondary to a post-viral myopathy from COVID-19, potentially exacerbated by a delayed critical illness myopathy from her recent prolonged hospitalization, although the delayed timing would be unusual. Toxic myopathy from statin use was considered but deemed less likely given the time course of her presentation, as well as negative HMGCR antibodies and muscle biopsy findings.
Discussion
The most common cause of rhabdomyolysis is trauma, but it is also common in critically ill medical patients. In the critically ill population, however, the diagnosis is often less clear and requires a high index of suspicion. Once diagnosed, determining the underlying etiology is also challenging, as many critically ill patients have multiple factors that may be contributing, or no clear unifying diagnosis.
One of the more common causes of rhabdomyolysis in medical patients is a critical illness myopathy (CIM) that occurs during prolonged hospitalization and immobility. Approximately 25–33% of patients who are ventilated for at least 4–7 days in the ICU develop either critical illness myopathy or critical illness polyneuropathy on clinical examination, and this rate is even higher when electrodiagnostic testing is used [17]. Unsurprisingly, a number of case studies have been published in which patients with severe COVID-19, usually those who required prolonged intubation and ICU stays, have developed weakness and difficulty weaning from the ventilator and have subsequently been diagnosed with CIM [12,13]. One case series looked at neurologic complications of 841 patients hospitalized with COVID-19, and found that myopathy occurred in 3.1% of these patients, but found that most cases of myopathy were related to critical illness and longer ICU admissions [11]. Another case report looked at 5 studies that have been published thus far on myopathy in patients with COVID-19 and found that most of the patients discussed were diagnosed with myopathy during their ICU stay and, compared to patients without symptoms of weakness, were ventilated for longer and had overall longer ICU stays, which suggests that an element of critical illness myopathy was likely contributing to these cases [14]. Our patient did have a prolonged hospitalization and associated immobilization during her initial admission for COVID-19, which raised the concern that a critical illness myopathy led to her presentation. She was also exposed to steroids, which has been shown to increase the risk for CIM [17]. However, the delayed nature of her presentation with a normal CK during her initial hospitalization is not consistent with a diagnosis of CIM (Figure 1) or with the presentation of cases of myopathy in critically ill patients with COVID-19 that are currently published, suggesting that this was likely not the primary driver of her clinical syndrome.
Separate from the myopathy seen in critically ill patients with COVID-19, there is also the hypothesis that COVID-19 itself has the potential to cause an acute myositis that may lead to rhabdomyolysis. It has long been known that certain infections, particularly viral infections, can cause rhabdomyolysis through an acute muscle injury [1,2]. The most common precipitating infection is influenza, found to be the cause in 42% of cases associated with a viral infection in a review by Singh and Scheld [2]. Numerous case reports in the literature suggest that COVID-19 may have the potential to cause an acute viral myositis similar to influenza [3–8]. One review article, which focused on the neurologic effects of COVID-19, found that approximately 4% of patients had findings consistent with skeletal muscle injury [9,10], but larger-scale studies are limited.
A few case reports have been published that suggest COVID-19 may also be associated with a delayed muscle injury and rhabdomyolysis [15,16], but there is very little information on the long-term effects of COVID-19 on the neurologic and musculo-skeletal system. This case adds to the presently available data by describing a case of rhabdomyolysis following recovery from severe COVID-19. It suggests that COVID-19 may have the potential to cause a post-viral myopathy that can lead to rhabdomyolysis in severe cases. Limitations of this case report include the fact that a statin-induced myopathy or a critical illness myopathy could have also contributed to the patient’s presentation, confounding the proposed association, although the patient’s time course of presentation make these diagnoses less likely.
Statin-associated muscle injury is usually sub-acute to chronic, with a modest rise in creatine kinase, whereas our patient had a very severe, acute presentation. In large clinical trials, severe rhabdomyolysis leading to renal failure was not seen with statin therapy alone, only in the setting of other clinical risk factors (usually concurrent use of other medications known to cause muscle injury or CYP3A4 inhibitors), so although it is possible our patient’s prior statin use contributed to her presentation, it was felt to be unlikely to be the sole driver of her muscle injury [18].
Conclusions
This case highlights the wide differential diagnosis of rhabdomyolysis and the work-up of rhabdomyolysis in a patient with recent COVID-19 and prolonged hospitalization. It raises the possibility that rhabdomyolysis may be associated with severe COVID-19 and could potentially represent a late complication of SARS-CoV-2 infection not yet described in the literature. However, data are limited, and more research, including larger-scale observational studies, is needed to further elucidate and confirm this suggested association.
Clinicians should be aware that COVID-19 has the potential to cause muscle injury, which in extreme cases can lead to rhabdomyolysis, both in the acute setting and after recovery. Although the data are not strong enough to recommend routine evaluation for rhabdomyolysis in this population, we would recommend that clinicians strongly consider this diagnosis when a patient presents with generalized weakness and/or an acute kidney injury with a history of recent COVID-19 disease.
Figures
References:
1.. Fadila M, Wool K, Rhabdomyolysis secondary to influenza A infection: A case report and review of the literature: N Am J Med Sci, 2015; 7(3); 122-24
2.. Singh U, Scheld W, Infectious etiologies of rhabdomyolysis: Three case reports and review: Clin Infect Dis, 1996; 22(4); 642-49
3.. Suwanwongse K, Shabarek N, Rhabdomyolysis as a presentation of 2019 novel coronavirus disease: Cureus, 2020; 12(4); e7561
4.. Rivas-García S, Bernal J, Bachiller-Corral J, Rhabdomyolysis as the main manifestation of coronavirus disease 2019: Rheumatology, 2020; 59(8); 2174-76
5.. Husain R, Corcuera-Solana I, Dayan E, Rhabdomyolysis as a manifestation of a severe case of COVID-19: A case report: Radiol Case Rep, 2020; 15(9); 1633-37
6.. Tram N, Chiodini B, Montesinos I, Rhabdomyolysis and acute kidney injury as leading COVID-19 presentation in an adolescent: Pediatr Infect Dis J, 2020; 39(10); e314-15
7.. Beydon M, Chevalier K, Al Tabaa O, Myositis as a manifestation of SARS-CoV-2: Ann Rheum Dis, 2020 [Online ahead of print]
8.. Singh B, Kaur P, Reid R, Case reports: Rhabdomyolysis associated with COVID-19: Am Fam Physician, 2020; 102(11); 645-48
9.. Orsucci D, Ienco EC, Nocita G, Neurological features of COVID-19 and their treatment: A review: Drugs Context, 2020; 9; 2020-5-1
10.. Mao L, Jin H, Wang M, Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan: China. JAMA Neurol, 2020; 77(6); 683-90
11.. Alcahut-Rodriguez C, Ahmad J, Del Valle-Perez J, Neurologic manifestations in hospitalized patients with COVID-19: The ALBACOVID registry: Neurology, 2020; 95(8); e1060-70
12.. Anderson H, Harbo T, Markvardsen K, Critical illness myopathy as a consequence of COVID-19 infection: Clin Neurophysiol, 2020; 131(8); 1931-32
13.. Bagnato S, Boccagni C, Marino G, Critical illness myopathy after COVID-19: Int J Infect Dis, 2020; 99; 276-78
14.. Ferrazzoli D, Kofler M, Loscher W, Case report: Myopathy in critically ill COVID-19 patients: A consequence of hyperinflammation?: Front Neurol, 2021; 12; 625144
15.. Jin M, Tong Q, Rhabdomyolysis as potential late complication associated with COVID-19: Emerg Infect Dis, 2020; 26(7); 1618-20
16.. Singh B, Kaur P, Mechineni A, Rhabdomyolysis in COVID-19: Report of four cases: Cureus, 2020; 12(9); e10686
17.. Shepherd S, Batra A, Lerner D, Review of critical illness myopathy and neuropathy: Neurohospitalist, 2017; 7(1); 41-48
18.. Baker S, Rosenson R, Statin muscle-related adverse events: UpToDate, 2021, Waltham, MA
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133