08 June 2021: Articles 
Neutropenia, Thrombocytopenia, and Eosinophilia: An Unusual Triad in a Patient on Long-Term Vancomycin Therapy
Unusual clinical course, Adverse events of drug therapy
Hendrik Lintel1ABDEF*, Mohammad Saffaf1ABDEFDOI: 10.12659/AJCR.931647
Am J Case Rep 2021; 22:e931647
Abstract
BACKGROUND: Vancomycin is an antibiotic commonly used for management of severe gram-positive infections. It is infrequently associated with hematologic adverse effects, ranging from isolated thrombocytopenia or neutropenia to pancytopenia. Although the mechanism is poorly understood, it is considered an immune-mediated phenomenon.
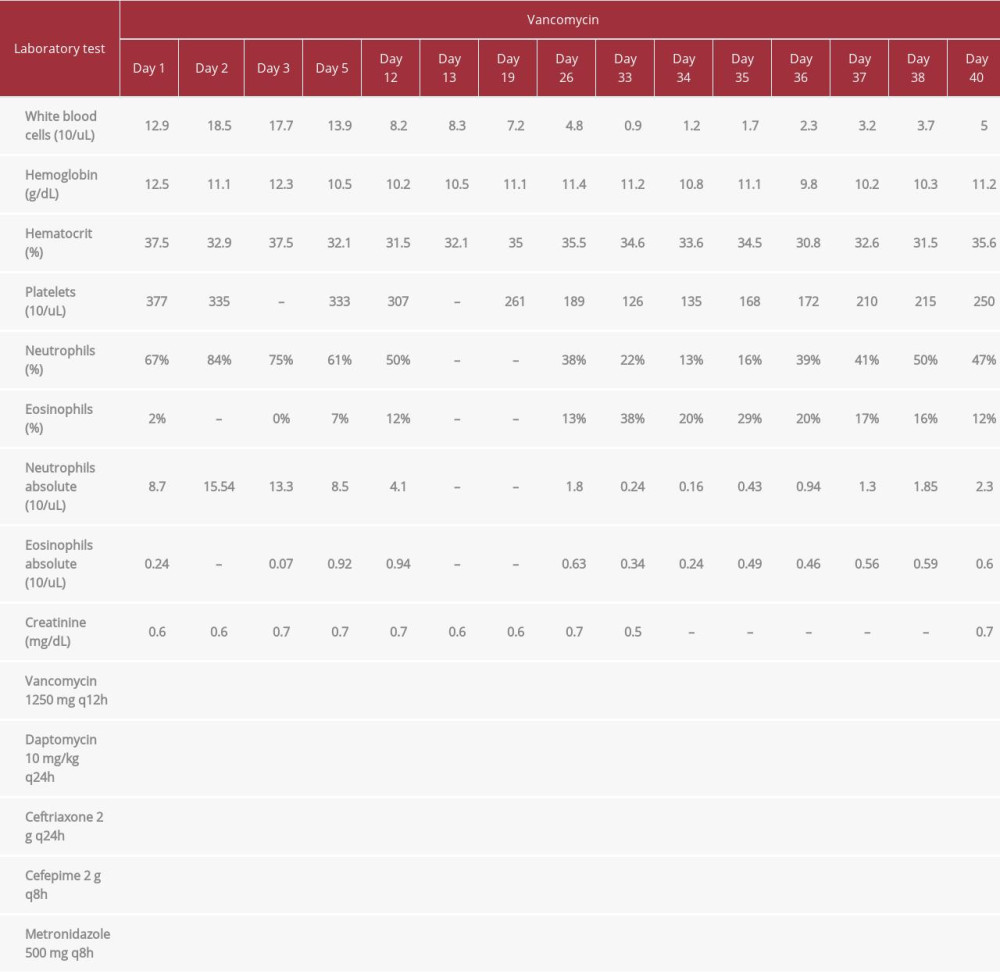
CASE REPORT: A 46-year-old woman with a history of intravenous drug use presented having 2 months of lower back pain associated with new acute lower-extremity weakness, numbness, paresthesia, and urinary/fecal incontinence. Magnetic resonance imaging revealed L5-S1 osteomyelitis with an epidural phlegmon, and broad-spectrum antibiotic coverage, including vancomycin, was initiated. On day 33 of treatment, the patient was noted to have developed neutropenia, thrombocytopenia, and eosinophilia. Vancomycin was the suspected cause and was replaced with daptomycin; laboratory tests for alternative causes of the bicytopenia were negative. Resolution of the bicytopenia occurred 5 days after vancomycin was stopped, and the eosinophilia continued to improve. The Naranjo adverse drug reaction probability scale score was 6, deeming vancomycin as the “probable” cause.
CONCLUSIONS: Routine blood analysis during long-term vancomycin therapy is crucial to identifying hematologic suppression early. Prompt discontinuation of vancomycin is key to the management of the condition, with some case reports advocating for filgrastim adjuvant therapy to accelerate recovery. Cases of recurrence of the cytopenia with reexposure to vancomycin have been documented, and therefore inquiry into prior adverse reactions to vancomycin is recommended. Given the widespread use of vancomycin and the potential risks of bleeding and infection associated with thrombocytopenia and neutropenia, respectively, we caution physicians to be aware of this rare adverse effect in patients on long-term vancomycin therapy.
Keywords: Eosinophilia, Neutropenia, Thrombocytopenia, Vancomycin, Anti-Bacterial Agents
Background
Vancomycin is a tricyclic glycopeptide, which is most often used in the treatment of severe gram-positive bacterial infections, in particular those caused by methicillin-resistant
Case Report
A 46-year-old woman with a past medical history of intravenous drug use and chronic anemia initially presented with a 2-month history of significant back pain. The day before presentation, the pain became unbearable, with acute onset of weakness in her left leg, bilateral lower-extremity numbness and tingling, and urinary and fecal incontinence. On examination, the patient had tenderness to palpation over the L3-S1 vertebrae as well as decreased rectal tone and weakness in both dorsiflexion and plantar flexion of the bilateral lower extremities. She was also noted to have a left anterior shin ulcer, which the patient reported to be chronic in nature. Magnetic resonance imaging (MRI) of the lumbar spine revealed L5-S1 osteomyelitis with associated paravertebral fluid collection, concerning for epidural abscess. The patient was given vancomycin and ceftriaxone for empiric coverage on day 1 of hospitalization. She subsequently underwent an L4-S1 laminectomy. Blood cultures and cultures from the epidural phlegmon were obtained and resulted in no growth after 5 days. Left shin wound cultures showed growth of
Other differential diagnoses were considered for this patient’s hematologic findings. Hemophagocytic lymphohistiocytosis was considered unlikely owing to the patient’s normal ferritin level, absence of fever, and the fact that eosinophilia is not a common finding in this condition. Disseminated intravascular coagulation was also unlikely because this patient had a calculated score of 2 based on the International Society of Thrombosis and Hemostasis criteria [3]. Given the patient’s concomitant leg wound and eosinophilia, vasculitis was also considered but was ruled out based on the wound biopsy results. An autoimmune etiology was deemed less likely given the lack of personal and family autoimmune history as well as the better explanation of vancomycin-induced cytopenia, given the time course. The Naranjo adverse drug reaction probability for vancomycin was calculated as a 6, making vancomycin a “probable” cause since this adverse reaction has previous conclusive reports, appeared after the drug was given, improved when the drug was discontinued, and other causes were ruled out. Notably, daily metabolic panels revealed no concordant renal dysfunction, indicating vancomycin-induced nephrotoxicity was unlikely to be the cause of the eosinophilia, although it has been shown to be associated with elevated eosinophil counts [4].
Discussion
Given the time course of the bicytopenia in our patient, it is likely that the same pathophysiologic process was responsible for the neutropenia and thrombocytopenia. While the etiology of vancomycin-induced cytopenias has not yet been fully elucidated, most evidence supports immune-mediated peripheral destruction as the cause over alternative possibilities such as bone marrow suppression or sequestration. Antibodies to neutrophils and platelets have been detected in cases of both neutropenia and thrombocytopenia.
In cases of vancomycin-induced neutropenia, onset typically occurs after a minimum of 12 days of vancomycin administration [5]. Numerous studies have found an increase in serum ANCA during episodes of neutropenia, although a causal relationship has never been established; one case report found ANCA testing was negative following the withdrawal of vancomycin after previously being positive during an episode of vancomycin-induced neutropenia [6–8]. ANCA is thought to induce peripheral destruction via a complement-mediated mechanism, according to an in vitro cytotoxicity test done by Akamizu et al on the serum of a patient with propylthiouracil-induced neutropenia with elevated ANCA [9]. As an alternative explanation, bone marrow suppression is a more unlikely cause given the inconsistency across case reports on bone marrow microscopic findings, the time lag after initial vancomycin dose to the neutropenia, the absence of dose dependence, and the rapid restoration of neutrophil and platelet counts once vancomycin is discontinued, as was seen in our patient [8].
Thrombocytopenia induced by vancomycin is postulated to also occur via an immune-mediated mechanism and most often reaches a nadir 8 days after initiation of therapy, although later times of onset have been documented [10]. Von Drygalski et al identified the presence of vancomycin-dependent, platelet-reactive antibodies in 34 patients (20%) suspected of having vancomycin-induced thrombocytopenia; no antibodies were found in 25 patients taking vancomycin without thrombocytopenia [10]. In the presence of vancomycin, the antibody is thought to bind to glycoprotein IIb/IIIa and cause phagocytic clearance by peripheral macrophages [11].
Eosinophilia is also an adverse drug reaction commonly linked to vancomycin, although the mechanism of drug-induced eosinophilia has yet to be elucidated [12]. Blumenthal et al found that 30.25% (n=314) of patients treated with vancomycin developed peripheral eosinophilia when compared with control patients [13]. Isolated eosinophilia is commonly believed to be a benign finding and not a cause for alteration of management; however, some reports suggest further scrutiny may be required because an increased risk of rash, renal injury, and DRESS syndrome can be linked to drug-induced eosinophilia [12,13]. For example, some studies have found eosinophilia to be associated in particular with cases of vancomycin-induced nephrotoxicity; therefore, evaluation of renal function is prudent in patients on long-term therapy who develop eosinophilia [4].
The neutropenia and thrombocytopenia in our patient were significant and required changes in her clinical care. Our patient was placed on neutropenic precautions to avoid infection owing to her immunocompromised status. She was also regularly monitored for evidence of active bleeding owing to the thrombocytopenia. While drug-induced thrombocytopenia is not typically associated with ecchymoses and hemorrhage, one-third of the vancomycin-induced thrombocytopenia cases reviewed by Von Drygalski et al had evidence of significant bleeding at very low platelet counts [10].
In our patient, when the hematologic abnormalities were identified, vancomycin was promptly discontinued, and recovery of her cell counts was noted by the fifth day after stoppage. For continued MRSA coverage, we changed her medication to daptomycin, but other options, per the Infectious Disease Society of America guidelines, could have included linezolid or clindamycin [14]. Another option to consider in similar patients is teicoplanin, an alternative glycopeptide; however, a retrospective study by Wu et al showed evidence that patients with a history of vancomycin-induced neutropenia had an increased incidence of developing neutropenia during long-term teicoplanin exposure [15]. While some reported cases have used filgrastim to aid in restoring neutrophil counts, our patient and other reported cases demonstrate that neutrophil counts can recover spontaneously without adjuvant therapy [5,6,8].
It is important to note that reexposure to vancomycin can potentially induce a rapid reoccurrence of a previous cytopenia because of the presence of long-lasting antibodies. Cases of rapid development of thrombocytopenia with severe bleeding within 24 hours of vancomycin reexposure in patients with history of vancomycin-induced thrombocytopenia have been reported [16]. Similar observations of reoccurrence of neutropenia have been made in patients with a history of vancomycin-induced neutropenia [17].
Lastly, it is worth noting that hematologic adverse reactions to vancomycin are not isolated to adult cases. Occurrences of thrombocytopenia and pancytopenia in children as young as newborns have been reported [18,19].
Conclusions
Vancomycin is an antibiotic commonly used for gram-positive infections and is associated with various hematologic adverse effects. In our case of suspected vancomycin-induced neutropenia, thrombocytopenia, and eosinophilia, prompt discontinuation of vancomycin resulted in a rapid restoration of cell counts without adjuvant therapy. Regular monitoring of cell counts, regardless of patient age, and inquiring about prior adverse hematologic reactions to vancomycin are key in the management of patients in need of long-term vancomycin therapy.
References:
1.. Bruniera FR, Ferreira FM, Saviolli LR, The use of vancomycin with its therapeutic and adverse effects: A review: Eur Rev Med Pharmacol Sci, 2015; 19(4); 694-700
2.. Rocha JL, Kondo W, Baptista MI, Uncommon vancomycin-induced side effects: Braz J Infect Dis, 2002; 6(4); 196-200
3.. Taylor FB, Toh CH, Hoots WK, , Scientific Subcommittee on Disseminated Intravascular Coagulation (DIC) of the International Society on Thrombosis and Haemostasis (ISTH). Towards definition, clinical and laboratory criteria, and a scoring system for disseminated intravascular coagulation: Thromb Haemost, 2001; 86(5); 1327-30
4.. Madigan LM, Fox LP, Vancomycin-associated drug-induced hypersensitivity syndrome: J Am Acad Dermatol, 2019; 81(1); 123-28
5.. Segarra-Newnham M, Tagoff SS, Probable vancomycin-induced neutropenia: Ann Pharmacother, 2004; 38(11); 1855-59
6.. di Fonzo H, Villegas Gutsch M, Castroagudin A, Agranulocytosis induced by vancomycin. Case report and literature review: Am J Case Rep, 2018; 19; 1053-56
7.. Grayson PC, Sloan JM, Niles JL, Antineutrophil cytoplasmic antibodies, autoimmune neutropenia, and vasculitis: Semin Arthritis Rheum, 2011; 41(3); 424-33
8.. Schwartz MD, Vancomycin-induced neutropenia in a patient positive for an antineutrophil antibody: Pharmacotherapy, 2002; 22(6); 783-88
9.. Akamizu T, Ozaki S, Hiratani H, Drug-induced neutropenia associated with anti-neutrophil cytoplasmic antibodies (ANCA): Possible involvement of complement in granulocyte cytotoxicity: Clin Exp Immunol, 2002; 127(1); 92-98
10.. Von Drygalski A, Curtis BR, Bougie DW, Vancomycin-induced immune thrombocytopenia: N Engl J Med, 2007; 356(9); 904-10
11.. Warkentin TE, Drug-induced immune-mediated thrombocytopenia – from purpura to thrombosis: N Engl J Med, 2007; 356(9); 891-93
12.. Ramírez E, Medrano-Casique N, Tong HY, Eosinophilic drug reactions detected by a prospective pharmacovigilance programme in a tertiary hospital: Br J Clin Pharmacol, 2017; 83(2); 400-15
13.. Blumenthal KG, Patil SU, Long AA, The importance of vancomycin in drug rash with eosinophilia and systemic symptoms (DRESS) syndrome: Allergy Asthma Proc, 2012; 33(2); 165-71
14.. Liu C, Bayer A, Cosgrove SE: Clin Infect Dis, 2011; 52(3); e18-55
15.. Hsiao SH, Chou CH, Lin WL, High risk of cross-reactivity between vancomycin and sequential teicoplanin therapy: J Clin Pharm Ther, 2012; 37(3); 296-300
16.. Getz TM, Packer CD, Rapid-onset vancomycin-induced thrombocytopenia with reexposure: Ann Pharmacother, 2019; 53(12); 1259-61
17.. Duff JM, Moreb JS, Muwalla F, Severe neutropenia following a prolonged course of vancomycin that progressed to agranulocytosis with drug reexposure: Ann Pharmacother, 2012; 46(1); e1
18.. Kalra K, Mittal HG, Maria A, Vancomycin-induced thrombocytopenia in a newborn: Drug Metab Pers Ther, 2016; 31(4); 235-37
19.. Leng B, Yan G, Li T, Hou N, Vancomycin-induced reversible pancytopenia and rash in a 16-month-old boy with osteomyelitis: A case report: Int J Clin Pharmacol Ther, 2020; 58(4); 242-46
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133