05 August 2021: Articles 
Encephalitis Due to Co-Infection with Cytomegalovirus and Herpes Simplex Virus Type 2 in a Patient with Acquired Immunodeficiency Syndrome
Unusual clinical course, Challenging differential diagnosis
Maleeha Zahid1ABCDEF*, Kishore KumarDOI: 10.12659/AJCR.931821
Am J Case Rep 2021; 22:e931821
Abstract
BACKGROUND: Opportunistic infections are commonly seen in immunocompromised patients. Here, we present an interesting case of a patient with poorly controlled human immunodeficiency virus (HIV) infection who presented with multiple opportunistic infections.
CASE REPORT: A 44-year-old woman with medical history of HIV infection (CD4 <20 cells/µl, viral load 172 996 copies/ml), presented with symptoms of headache for 2 days and changes in mentation. She was recently treated for pulmonary mycobacterium avium complex infection. Her physical examination revealed normal breath sounds and her abdominal examination was unremarkable. She did not have any focal neurological deficits, nuchal rigidity, or papilledema on examination. Computed tomography (CT) head was negative for any acute lesions. She was empirically started on vancomycin and piperacillin-tazobactam. Due to persistent symptoms, a lumbar puncture was performed, which revealed elevated total proteins in CSF, and a viral polymerase chain reaction test was positive for herpes simplex virus type 2 (HSV-2) and cytomegalovirus (CMV). Magnetic resonance imaging of the brain showed mild enhancement of the ventricular lining. She was treated with acyclovir, which was later changed to ganciclovir, with resulting clinical improvement. The patient had clinical improvement and was discharged home.
CONCLUSIONS: Multiple opportunistic co-infections should be considered in patients with poorly controlled HIV infection.
Keywords: Encephalitis, Herpes Simplex, HIV Infections, Acquired Immunodeficiency Syndrome, coinfection, Encephalitis, Herpesvirus 2, Human
Background
The central nervous system (CNS) is a common focus of opportunistic infection in patients with human immunodeficiency virus (HIV) infection and especially those with acquired immunodeficiency syndrome (AIDS) [1]. Cytomegalovirus (CMV), progressive multifocal leukoencephalopathy (PML), toxoplasma encephalitis, and cryptococcal meningitis are some of the common CNS infections seen in HIV-infected patients [2]. In the era of highly active antiretroviral therapy (HAART), the neurological manifestations in HIV-infected patients have changed, [3] but largely remain unaltered in patients who have high viral loads, especially those with noncompliance with HAART.
Initial management for HIV-infected patients suspected to have a CNS infection includes carefully taking a history of prior opportunistic infections, imaging studies, cerebrospinal fluid (CSF) analysis including testing for viral polymerase chain reaction (herpes simplex virus), and empiric treatment [4]. Brain biopsy is rarely required for diagnosis. Patients who have a history of prior opportunistic infection can subsequently present with disseminated disease involving the CNS. However, co-infection by 2 different viruses is very rare. Here, we present a case of a patient with AIDS and poorly controlled HIV viral load with non-mycobacterium tuberculosis pulmonary infection, who subsequently presented with acute encephalitis. Subsequent evaluation revealed herpes simplex virus type 2 (HSV-2) and CMV co-infection as the etiology for encephalitis.
Case Report
A 44-year-old woman with a medical history of HIV/AIDS (CD4 <20 cells/UL, viral load 172 996 copies/ml), non-adherence to anti-retroviral therapy (HAART), and prior history of intravenous drug use was readmitted 2 days after prolonged hospitalization with headache for 2 days and changes in mentation. Two weeks prior to the index admission, she had presented with upper respiratory tract symptoms and abnormal chest radiography. During the hospital stay, she underwent testing with serial acid-fast bacilli sputum (AFB) analysis, which came back positive. She was initially placed on airborne isolation and treated for mycobacterium tuberculosis. Later, mycobacterium avium complex was identified in the AFB culture and treatment was changed to ethambutol, azithromycin, and rifabutin. Atovaquone was given for empiric
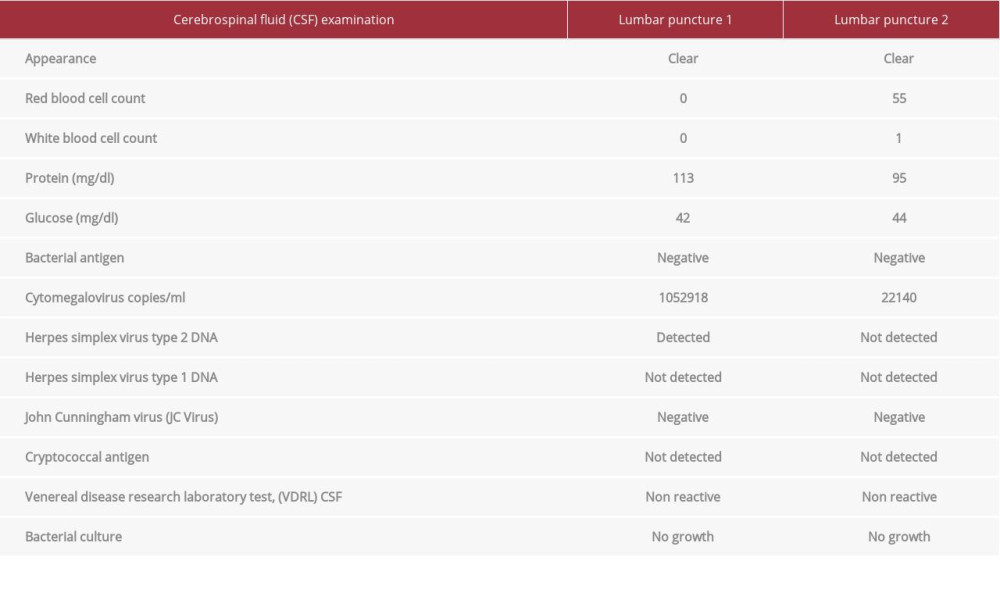
She did not have any focal neurological deficits, nuchal rigidity, or papilledema on examination. Her repeat chest X-ray showed improving pneumonia. Computed tomography (CT) head was negative for any acute lesions. She was empirically started on vancomycin and piperacillin-tazobactam. Due to persistent headache and suspicion for opportunistic and bacterial infection in the setting of acquired immune deficiency syndrome, a spinal tap was performed. CSF analysis revealed elevated total protein in cerebrospinal fluid (CSF) and a viral polymerase chain reaction test (PCR) was positive for herpes simplex virus type 2 (HSV-2) and cytomegalovirus (CMV).
John Cunningham virus (JC Virus) PCR and the cryptococcal antigen were negative (Table 1). HIV viral testing was not performed in the CSF. Acyclovir was added and piperacillin-tazobactam was changed to cefepime. Magnetic resonance imaging of the brain done during the admission showed mild enhancement of the ventricular lining. No mass lesions were identified in the brain parenchyma, brainstem, or meninges. The ventricles were within normal limits in size, position, and configuration for the patient’s age (Figure 1). There were also a few foci of likely subacute infarct in the basal ganglia and cerebellum. Acyclovir was changed to ganciclovir due to its enhanced efficacy against both HSV and CMV infection. Vancomycin and cefepime were discontinued. The highly active retroviral therapy (HAART) was deferred due to presence of multiple opportunistic infection and high risk for immune reconstitution syndrome (IRS). The patient was gradually improving but continued to have waxing and waning mental status; hence, repeat lumbar puncture was performed to evaluate the effectiveness of therapy. The repeat examination of CSF was negative for HSV, and revealed interval decrease in CMV viral copies (Table 1). Given the interval decrease in CMV titers, ganciclovir resistance was not suspected. The patient was maintained on ganciclovir therapy for secondary prophylaxis against CMV due to presence of advanced immunosuppression. Her mental status subsequently returned to baseline and she was discharged to a short-term rehabilitation center (Figure 2).
Discussion
The presented case shows the importance of evaluating all common etiologies of CNS infection in patients with AIDS, even in the presence of other opportunistic infection. The clinical presentation may assist in differentiating opportunistic infections in patients with HIV. Nontuberculous mycobacterium (NTM) infections are common in HIV-infected patients and usually involve the lungs [5,6]. Patients presenting with movement disorder can have underlying toxoplasmosis and those with cortical blindness need to be evaluated for PML. Imaging studies are usually the initial step of evaluation. Magnetic resonance imaging (MRI) is considered more sensitive than computed tomography (CT) [7,8]. Imaging studies revealing ring-enhancing lesions usually suggest toxoplasma encephalitis. [9] Toxoplasma serology can also help differentiate this infection from malignant etiologies like lymphoma [10].
In addition to infectious complications, HIV patients with low CD4 count and high plasma viral load are at risk of developing HIV encephalitis. This disease entity is thought to be caused by direct damage of the central nervous system by the HIV virus in combination with the body’s immune response to the virus [11]. The disease can involve any part of the brain; however, basal ganglia and nigrostriatal areas are more commonly involved early on in the disease [12]. Clinical presentation of these patients varies from asymptomatic disease to minor neurocognitive dysfunction. In advance cases, patient can present with severe dementia. CSF analysis is used to rule out opportunistic infections, as the presence of HIV RNA in CSF is a non-specific finding. Similarly, the presence of HIV virus in cultures obtained from CSF, nerve, and brain tissue is also non-specific and does not correlate with neurological signs and symptoms. MRI brain usually shows cortical atrophy and, in advanced stages, multiple foci of non-enhancing but hyper-intense lesion in a subcortical distribution [13]. HAART is the backbone of HIV encephalitis treatment [14].
Host immune and nonimmune cells play an important role in the body’s antiviral response. There are specialized receptors on the host cells, called pattern recognition receptors (PPRs), that identify pathogen-associated molecular patterns (PAMPs) as well as stress signals by the host cells [15]. These stress signals are called damage-associated patterns (DAMPs) [16]. PPRs uses these PAMPs and DAMPs signals to perform intra-cellular transcription and translation of antiviral genes. These pathways establish protective immunity as well as effective virus control.
Cytomegalovirus (CMV), a family of herpes virus, accounts for less than 2% of CNS infections in patient with HIV infection, though autopsy findings have shown up to 10% to 40% of patient with CMV neuropathic infection [17]. This difference in prevalence in autopsy series affirms the wide spectrum of CMV infection, which may range from asymptomatic infection to life-threatening presentation. Retinitis, esophageal ulcer, colitis, and pneumonitis are the other manifestations of CMV infection [18]. Another autopsy series showed that patients with CNS infection can have concomitant infection of other sites [19]. The same autopsy series presented by Hollang et al revealed that 58% of patient have associated MAC infection [19].
CMV infection is generally manifested in severely immunocom-promised HIV-positive patients in whom CD4 counts fail to rise above 100 cells per µL [20]. Cytotoxic T lymphocytes play an important role in control of CMV infection, thereby making patients with poorly controlled HIV at higher risk of severe infection [21]. In addition, CMV inhibits the antigen presenting function and lymphocyte proliferation, which can lead to impairment of T cell response to the infection [22–24]. Neurological manifestations can vary from polyradiculopathy to encephalitis. Imaging finding of the encephalitis may reveal multifocal nodular or ventriculoencephalitis, which can present with focal neurological symptoms in the form of progressive delirium, nystagmus, ataxia, and cranial nerve deficit [25]. Brain biopsy is specific for CMV brain infection and is only required in patients whose diagnosis remains uncertain despite serological and CSF analysis [26]. Human cytomegalovirus (HCMV) pp65 detection in CSF fluid is highly specific for the diagnosis [27]. Spinal fluid CMV PCR is specific for the detection of the virus; however, low levels can be present in asymptomatic carriers. Hence, the level of the CMV DNA should be utilized to guide the management [28].
HSV encephalitis occurs sporadically across the United States. After infection, HSV stays latent in trigeminal ganglia until its reactivation. Toll-like receptors are expressed on the surface of astrocytes and participate in immune signaling pathways that restrict HSV spread in the CNS [29]. Defects in Toll-like receptor expression has been thought to play a role in neuroinvasion of HSV. After entry into the CNS, the virus causes neuronal injury and clinical disease. HSV encephalitis in immunocompromised patients may have atypical clinical and radiological features and outcomes also vary [30]. HSV -1 encephalitis is more common than HSV-2 neurological infection [31]. Hence, HSV-2 encephalitis in HIV-infected individuals is considered rare [32].
The optimal approach for CMV encephalitis is not well-defined. Ganciclovir has been used as first-line therapy and resistance is sometimes reported. An induction regimen with foscarnet and ganciclovir has also been effective for CMV encephalitis. Serial CSF analysis for CMV DNA can guide the treatment for suspected ganciclovir resistance [33]. Acyclovir is preferred for HSV encephalitis treatment [34]. Ganciclovir is an alternative agent when acyclovir cannot be used [35]. However, foscarnet is preferred for acyclovir-resistant HSV [36]. Information on encephalitis with HSV and CMV co-infection is scare and limited to autopsy series [37]. Serial CSF titer for CMV can be used to transition to an oral formulation of valganciclovir, which is usually continued until CD4 count is increased to above 100 cells per µL. However, immune reconstitution syndrome due to HAART can obscure clinical improvement of CMV encephalitis, and it is suggested to hold HAART in the initial 2 weeks of treatment of opportunistic infection [38].
Conclusions
Encephalitis due to a co-infection with CMV and HSV-2 in an HIV-infected individual is rare, and a co-infection with the pulmonary MAC has not been reported in the literature. The presented case emphasizes the importance of investigating the possibility of opportunistic co-infection in patients with poorly controlled HIV infection.
References:
1.. Collazos J, Opportunistic infections of the CNS in patients with AIDS: Diagnosis and management: CNS Drugs, 2003; 17(12); 869-87
2.. Solomon FB, Angore BN, Koyra HC, Spectrum of opportunistic infections and associated factors among people living with HIV/AIDS in the era of highly active anti-retroviral treatment in Dawro Zone hospital: a retrospective study: BMC Res Notes, 2018; 11(1); 604
3.. Bowen LN, Smith B, Reich D, HIV-associated opportunistic CNS infections: Pathophysiology, diagnosis and treatment: Nat Rev Neurol, 2016; 12(11); 662-74
4.. Bowen LN, Smith B, Reich D, HIV-associated opportunistic CNS infections: Pathophysiology, diagnosis and treatment: Nat Rev Neurol, 2016; 12(11); 662-74
5.. Lapinel NC, Jolley SE, Ali J, Welsh DA, Prevalence of non-tuberculous mycobacteria in HIV-infected patients admitted to hospital with pneumonia: Int J Tuberc Lung Dis, 2019; 23(4); 491-97
6.. Anjos LRBD, Parreira PL, Torres PPTS, Non-tuberculous mycobacterial lung disease: A brief review focusing on radiological findings: Rev Soc Bras Med Trop, 2020; 53; e20200241
7.. Farhadian S, Patel P, Spudich S, Neurological complications of HIV infection: Curr Infect Dis Rep, 2017; 19(12); 50
8.. Saylor D, Neurologic complications of human immunodeficiency virus infection: Continuum (Minneap Minn), 2018; 24(5, Neuroinfectious Disease); 1397-421
9.. Elsheikha HM, Marra CM, Zhu XQ, Epidemiology, pathophysiology, diagnosis, and management of cerebral toxoplasmosis: Clin Microbiol Rev, 2020; 34(1); e00115-19
10.. Pierce MA, Johnson MD, Maciunas RJ, Evaluating contrast-enhancing brain lesions in patients with AIDS by using positron emission tomography: Ann Intern Med, 1995; 123(8); 594-98
11.. McGuire JL, Gill AJ, Douglas SD, Kolson DL, Central and peripheral markers of neu-rodegeneration and monocyte activation in HIV-associated neurocognitive disorders: J Neurovirol, 2015; 21(4); 439-48
12.. Langford TD, Letendre SL, Larrea GJ, Masliah E, Changing patterns in the neuropathogenesis of HIV during the HAART era: Brain Pathol, 2003; 13(2); 195-210
13.. Valcour V, Paul R, Chiao S, Screening for cognitive impairment in human immunodeficiency virus: Clin Infect Dis, 2011; 53(8); 836-42
14.. Chan P, Hellmuth J, Spudich S, Valcour V, Cognitive impairment and persistent CNS injury in treated HIV: Curr HIV/AIDS Rep, 2016; 13(4); 209-17
15.. Mogensen TH, Pathogen recognition and inflammatory signaling in innate immune defenses: Clin Microbiol Rev, 2009; 22; 240-73
16.. Johnson KE, Chikoti L, Chandran B, Herpes simplex virus 1 infection induces activation and subsequent inhibition of the IFI16 and NLRP3 inflammasomes: J Virol, 2013; 87; 5005-18
17.. Petito CK, Cho E-S, Lemann W, Neuropathology of acquired immuno-deficiency syndrome (AIDS): An autopsy review: J Neuropathol Exp Neurol, 1986; 45(6); 635-46
18.. Perello R, Vergara A, Monclus E, Cytomegalovirus infection in HIV-infected patients in the era of combination antiretroviral therapy: BMC Infect Dis, 2019; 19(1); 1030
19.. Holland NR, Power C, Mathews VP, Cytomegalovirus encephalitis in acquired immunodeficiency syndrome (AIDS): Neurology, 1994; 44(3 Pt 1); 507-14
20.. Springer KL, Weinberg A, Cytomegalovirus infection in the era of HAART: Fewer reactivations and more immunity: J Antimicrob Chemother, 2004; 54(3); 582-86
21.. Wills MR, Carmichael AJ, Mynard K, The human cytotoxic T-lymphocyte (CTL) response to cytomegalovirus is dominated by structural protein pp 65: Frequency, specificity, and T-cell receptor usage of pp 65-specific CTL: J Virol, 1996; 70(11); 7569-79
22.. Varani S, Frascaroli G, Landini MP, Soderberg-Naucler C, Human cytomegalovirus targets different subsets of antigen-presenting cells with pathological consequences for host immunity: Implications for immunosuppression, chronic inflammation and autoimmunity: Rev Med Virol, 2009; 19(3); 131-45
23.. Rinaldo CR, Carney WP, Richter BS, Mechanisms of immunosuppression in cytomegaloviral mononucleosis: J Infect Dis, 1980; 14(4); 488-95
24.. Rinaldo CR, Rinaldo RL, Alteration of immunoregulatory mechanisms during cytomegalovirus mononucleosis: effect of in vitro culture on lymphocyte blastogenesis to viral antigens: Clin Immunol Immunopathol, 1983; 28(1); 46-55
25.. Skiest DJ, Focal neurological disease in patients with acquired immunodeficiency syndrome: Clin Infect Dis, 2002; 34(1); 103-15
26.. Moulignier A, Mikol J, Gonzalez-Canali G, AIDS-associated cytomegalovirus infection mimicking central nervous system tumors: A diagnostic challenge: Clin Infect Dis, 1996; 22(4); 626-31
27.. Revello MG, Percivalle E, Sarasini A, Diagnosis of human cytomegalovirus infection of the nervous system by pp65 detection in polymorpho-nuclear leukocytes of cerebrospinal fluid from AIDS patients: J Infect Dis, 1994; 170(5); 1275-79
28.. Arribas JR, Clifford DB, Fichtenbaum CJ, Level of cytomegalovirus (CMV) DNA in cerebrospinal fluid of subjects with AIDS and CMV infection of the central nervous system: J Infect Dis, 1995; 172(2); 527-31
29.. Jack CS, Arbour N, Manusow J, TLR signaling tailors innate immune responses in human microglia and astrocytes: J Immunol, 2005; 175; 4320-30
30.. Tan IL, McArthur JC, Venkatesan A, Nath A, Atypical manifestations and poor outcome of herpes simplex encephalitis in the immunocompromised: Neurology, 2012; 79(21); 2125-32
31.. Cook M, Koehn T, Simmons D, Frattalone A: A rare case of herpes simplex virus type 2 encephalitis in an immunocompetent adult (P5.340), 2018; 90(15 Suppl); P5.340
32.. Pagliano P, Ascione T, Carleo MA, HIV positive patient with HSV-2 encephalitis: Case report: Infez Med, 2016; 24(3); 245-49
33.. Cohen BA, Prognosis and response to therapy of cytomegalovirus encephalitis and meningomyelitis in AIDS: Neurology, 1996; 46(2); 444-50
34.. Whitley RJ, Alford CA, Hirsch MS, Vidarabine versus acyclovir therapy in herpes simplex encephalitis: N Engl J Med, 1986; 314(3); 144-49
35.. Bradshaw MJ, Venkatesan A, Herpes simplex virus-1 encephalitis in adults: Pathophysiology, diagnosis, and management: Neurotherapeutics, 2016; 13(3); 493-508
36.. Erlich KS, Mills J, Chatis P, Acyclovir-resistant herpes simplex virus infections in patients with the acquired immunodeficiency syndrome: N Engl J Med, 1989; 320(5); 293-96
37.. Laskin OL, Stahl-Bayliss CM, Morgello S, Concomitant herpes simplex virus type 1 and cytomegalovirus ventriculoencephalitis in acquired immunodeficiency syndrome: Arch Neurol, 1987; 44(8); 843-47
38.. Kaplan JE, Benson C, Holmes KK, Guidelines for prevention and treatment of opportunistic infections in HIV-infected adults and adolescents: Recommendations from CDC, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America: MMWR Recomm Rep, 2009; 58(Rr-4); 1-207 ; quiz CE1-4
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133