26 November 2021: Articles 
A New Potential Strategy for Acute Non-Alcoholic Steatohepatitis (NASH)
Unusual or unexpected effect of treatment
Anupamjeet Kaur Sekhon1BDEF*, Aniruddha Gollapalli1E, Dharamjeet Kaur2E, Bryan Janssen3D, Mark L. StevensDOI: 10.12659/AJCR.932961
Am J Case Rep 2021; 22:e932961
Abstract
BACKGROUND: Non-alcoholic fatty liver disease (NAFLD) is the most common cause of chronic liver disease in the United States, and 25% of patients with NAFLD progress to non-alcoholic steatohepatitis (NASH). NAFLD is predicted to be the most common indication for liver transplantation by 2030. Despite associated high morbidity and mortality, there is currently no approved therapy for NASH. PCSK9 inhibitors are approved for reducing LDL in patients who are statin-intolerant or need further LDL reduction. Increased LDL levels are independently associated with an elevated risk of NAFLD.
CASE REPORT: We present a case of a 39-year-old woman with acute NASH with familial hypercholesterolemia that was refractory to lifestyle modifications and HMG-CoA reductase inhibitors. An episode of rhabdomyolysis warranted a search for alternatives to statin therapy. Results of a liver biopsy showed microvesicular and macrovesicular steatosis with ballooning degeneration, indicating acute NASH. She was started on PCSK9 inhibitors as salvage therapy. Three monthly doses resulted in a more than an 80% reduction in ALT and AST and a 48% reduction in LDL levels. A liver biopsy done 8 months after the first biopsy showed normalization of liver histology.
CONCLUSIONS: The use of PCSK9 inhibitors showed a dramatic response in this patient who failed conventional therapies, and the encouraging results seen in this case merit further research into the use of PCSK9 inhibitors as first-line therapy for the acute phase of NASH.
Keywords: Hyperlipidemia, Familial Combined, Liver Function Tests, Non-alcoholic fatty liver disease, PCSK9 Protein, Human, Hypercholesterolemia, Fatty Liver, Female, Humans, Hydroxymethylglutaryl-CoA Reductase Inhibitors, Liver, Proprotein Convertase 9
Background
Non-alcoholic fatty liver disease (NAFLD) is estimated to be the most common cause of chronic liver disease in the United States. It affects from 80 million to 100 million individuals, among whom nearly 25% progress to non-alcoholic steatohepatitis (NASH) [1]. About 20% of NASH ases progress to end-stage liver disease or cirrhosis [2]. NAFLD is predicted to be the most common indication for liver transplantation by 2030 [3]. Despite it having high morbidity and mortality, there is no approved therapy for NASH. We present a case of NASH with statin- refractory hypercholesterolemia in which liver enzymes, cholesterol indices, and liver histology normalized after proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor treatment.
Case Report
A 39-year-old woman with familial hypercholesterolemia had suboptimal control of cholesterol indices despite HMG-CoA reductase inhibitor therapy (Table 1). She had a body mass index of 25 kg/m2 (normal range, 18–25 kg/m2). She had been on rosuvastatin 10 mg for 10 years along with lifestyle modifications, including vigorous physical activity 4 to 5 times per week and a low-fat diet, with minimal results. Subsequently, in April 2019, the patient developed rhabdomyolysis (creatinine kinase 131 632 U/L [reference range, 22–269 U/L] and myoglobin 325 ng/mL [range, 14–66 ng/mL]) secondary to statin use, requiring cessation of her rosuvastatin regimen. During this time, she had an increase in her liver function tests (LFTs) (Table 2), which was deemed secondary to rhabdomyolysis. Six months after this episode, persistent elevation of her LFTs necessitated ultrasound imaging, which showed a 1.3-cm septated lesion of the right hepatic lobe. The patient was consequently diagnosed with a liver hemangioma, and her oral contraceptive pills were stopped.
Additional tests were performed to determine the underlying etiology of her persistently elevated LFTs. The hepatitis panel, ceruloplasmin levels, autoimmune panel, alpha-1 antitrypsin antibody, ferritin levels, hemoglobin A1c, and thyroid panel all came back within normal limits (Table 3). Genetic testing for the solute carrier organic anion transporter family member 1B1 genotype showed normal organic anion transporter poly-peptide 1B1 function, suggesting a typical risk for myopathy and no excessive circulating levels of statins. She had a fibrosis score of 0.46, classified as stage F1–F2, with portal and bridging fibrosis with few septa, and moderate steatosis grade S2, with a steatosis score of 0.63. Her occasional alcohol consumption and an aspartate transaminase/alanine transaminase (AST/ALT) ratio of less than 2 suggested against alcoholic liver disease.
Later, she developed nausea and elevated gamma-glutamyl transpeptidase (GGT) (212 U/L; reference range <40 U/L), secondary to chronic cholelithiasis. She underwent a laparoscopic cholecystectomy and a liver biopsy in December 2019. Liver histology showed mixed microvesicular and macrovesicular steatosis with megamitochondria and foci of ballooning degeneration, pointing to a diagnosis of NASH (Figure 1).
The patient’s LFTs continued to rise despite the cholecystectomy, with ALT levels reaching 1862 U/L (range, 5–40 U/L). She was then referred to a hepatologist and learned that her condition could either reverse and resolve or progress from NASH to end-stage liver disease, eventually requiring a liver transplant.
Owing to the lack of desired results with the standard of care and her statin intolerance, the patient was started on evolocumab, a monoclonal antibody that inhibits PCSK9, as salvage therapy. After receiving evolocumab, she had a significant improvement in her lipid and LFT levels. After the first dose, ALT decreased by 89% (1251 to 133 U/L), AST by 86% (366 to 53 U/L [range, 9–40 U/L]), alkaline phosphatase (ALP) by 16% (140 to 118 U/L [range, 40–112 U/L]), total cholesterol (TC) by 36% (306 to 197 mg/dL [range, <200 mg/dL]), and low-density lipoprotein (LDL) by 48% (226 to 117 mg/dL [range, <100 mg/dL]). Liver enzymes and cholesterol indices normalized after the third dose of evolocumab (ALT of 33 U/L, AST of 33 U/L, ALP of 93 U/L, TC of 173 mg/dL, and LDL of 98 mg/dL). Since then, the values have stayed within normal limits, and she has continued to tolerate evolocumab well (Figures 2, 3). A follow-up liver biopsy in August 2020, performed 8 months after the initial biopsy, was negative for active and chronic hepatitis and steatosis (Figure 4). There was no pericellular fibrosis or hepatocyte ballooning on histology, but only occasional histiocytes containing debris, suggestive of prior hepatic injury and indicating a successful PCSK9 inhibitor treatment.
Discussion
NAFLD encompasses the entire spectrum of fatty liver disease in individuals lacking significant alcohol consumption, ranging from fatty liver to steatohepatitis to cirrhosis. The pathological progression of NAFLD follows a “3-hit” process, namely steatosis, lipotoxicity, and inflammation. NASH is defined as the presence of ≥5% hepatic steatosis with inflammation and hepatocyte injury (ballooning), with or without fibrosis. NASH can progress to cirrhosis, decompensated liver failure, and liver cancer [4]. NASH has an all-cause mortality rate of 25.6 per 1000 person-years and a liver-specific mortality rate of 11.8 per 1000 person-years [5]. Younossi et al described a 9% annual increase of hepatocellular carcinoma cases related to NAFLD over a period of 6 years, from 2004 to 2009 [6]. According to the World Gastroenterology Organization (WGO), American Association for the Study of Liver Diseases (AASLD), and European Association of Study of Liver (EASL), the current mainstay of treatment for NAFLD/NASH includes lifestyle modifications, such as diet, exercise, and weight loss. Weight loss ≥ 5% of the current body weight has been linked to improvement in hepatic steatosis, while ≥ 7% weight loss is linked to histo-logical improvement in NASH [7]. Johnson et al demonstrated that aerobic exercises might lower the hepatic and visceral lipids in individuals with obesity even without weight loss [8].
Pharmacological therapy is indicated only in patients with progressive fibrosis, in patients with biopsy-proven NASH, and in patients who fail lifestyle interventions after a period of 6 months [4,9]. Currently, there is no universally approved treatment for NASH; however, EASL and AASLD recommend HMG-CoA reductase inhibitors for NASH patients with hyper-cholesterolemia to reduce LDL cholesterol, despite no proven improvement in liver histology. Pioglitazone has been shown to improve histology, metabolism, and ALT levels in NASH [6,10]. The Pioglitazone, Vitamin E, or Placebo for Non-Alcoholic Steatohepatitis (PIVENS) trial demonstrated that vitamin E improved the histological features of NASH in 43% of patients, compared with in 19% of patients who received placebo for 94 weeks [11]. Various drugs, including elafibranor, obeticholic acid, glucagon-like peptide-1 (GLP-1) agonists, ursodeoxycholic acid, and antifibrotic agents (anti-lysyl oxidase-like monoclonal antibodies), are being tested in late-phase clinical trials for the treatment of NASH [4,12,13]. Semaglutide, a GLP-1 agonist in a phase 2 trial, showed NASH resolution, compared with placebo [14]. Bariatric surgery-induced weight loss has been proven to improve NASH in 85% of patients after 1 year [15]. Surgical intervention is recommended by the WGO, AASLD, and EASL only in patients with morbid obesity who fail to improve with lifestyle changes and pharmacological measures. Liver transplantation is advocated in patients with end-stage liver disease and liver fibrosis despite cardiovascular mortality, NASH recurrence, and transplant failure. Thus far, there is no recognized pharmacological treatment for the acute phase of NASH.
There is growing interest in the use of the PCSK9 inhibitor in alcoholic liver disease and NAFLD. PCSK9 is an enzyme that plays a crucial role in cholesterol metabolism [16]. LDL receptors, which are proteins on the surface of hepatocytes, bind to LDLs and remove them from the bloodstream. The LDL receptor, at that point, will either be recycled or targeted for destruction by PCSK9 [16]. PCSK9 inhibitors, which are primarily monoclonal antibodies, function by binding to PCSK9, thereby allowing recycling of LDL receptors to the cell surface [16]. This increases availability of LDL receptors on the cell surface to further facilitate reduction in circulating LDL. The PCSK9 inhibitors evolocumab and alirocumab significantly lowered LDL levels in 2 recent randomized controlled trials: The Open-Label Study of Long-Term Evaluation Against LDL Cholesterol (OSLER) study and the ODYSSEY Outcomes study [17].
The OSLER extension study program showed a 61% reduction in LDL when patients were placed on adjuvant therapy with evolocumab as opposed to standard of care alone [18]. In 2015, the U.S. Food and Drug Administration (FDA) approved the use of PCSK9 inhibitors for the prevention of major adverse cardiac events in adults with established atherosclerotic cardiovascular disease on maximally tolerated statin therapy who require further reduction in LDL [19]. These medications also play a significant role in LDL reduction in patients who are statin intolerant, as statin-related myopathy is an issue that is reported by 5% to 20% of the patient population. The Goal Achievement after Utilizing an Anti-PCSK9 Antibody in Statin Intolerant Subjects-3 (GAUSS-3) randomized clinical trial found that the use of evolocumab in statin-intolerant patients, compared with ezetimibe, resulted in a significant decrease in lipid levels after a 24-week period [20].
Evolocumab and alirocumab can both be administered as a biweekly or monthly subcutaneous injection for primary hyper-lipidemia as well as cardiovascular event prevention, which makes patient compliance easier, compared to oral medications that need to be taken every day. A recent study done by Lee et al in rat models showed that anti-PCSK9 treatment with alirocumab decreases alcohol-induced hepatocellular steatosis, inflammation, and oxidative injury; however, PCSK9 inhibitors are not currently indicated or approved for the treatment of NASH [21]. Ruscica et al suggested that PCSK9 levels are associated with severity of steatosis and lipogenesis and might be associated with the pathogenesis of NAFLD [22]. By contrast, a meta-analysis by Wargny et al suggested that PCSK9 levels have no correlation with the severity of hepatic fat accumulation and histological markers of NASH [23]. A recent study in Wenzhou, China, by Sun et al has hinted at a significant correlation between LDL-C levels and the incidence of NAFLD [24]. To the best of our knowledge, the successful use of PCSK9 inhibitors for NAFLD has not been reported in the literature.
This present case illustrates a novel indication for PCSK9 inhibitor to reverse NASH-related liver changes. The patient had familial hypercholesterolemia, with more than 10 years of lifestyle modifications and HMG-CoA reductase inhibitor use, and her cholesterol and liver enzymes did not normalize, nor was the progression of NAFLD to NASH halted. She was then started on PCSK9 inhibitors as salvage therapy, and after the first dose of evolocumab, her liver enzymes and cholesterol indices started trending down. Eight months after the initial biopsy and evolocumab treatment, a repeat biopsy was negative for NASH-related histologic changes.
There are limitations to this case report, as it was observed in 1 patient, and we need more patients to corroborate these results. High cost and prior authorization from insurance providers are some of the other barriers in the accessibility of PCSK9 inhibitors. Therefore, they are currently used for secondary prevention in high-risk groups only.
PCSK9 inhibitors have been evaluated for safety for up to 5 years in clinical trials. Immunologic effects, such as local injection site reactions, are the most commonly reported adverse effects, which include erythema, pain, and bruising. Post-hoc analysis and post-marketing surveillance will shed light on serious adverse effects that were noticed in animal studies, but additional data are needed in humans [21].
Conclusions
PCSK9 Inhibitors are FDA approved for major adverse cardiac event prevention and they play a significant role in cholesterol metabolism. In the present case, the use of PCSK9 inhibitors in the management of acutely progressive NASH showed reversal of liver histology in a patient that failed conventional therapies, with normalization of both liver enzymes and cholesterol indices. Further research, including clinical trials, is warranted on PCSK9 inhibitor use as a first-line therapy in NAFLD, as it is expected to become the leading cause of chronic liver disease in the foreseeable future and there is no approved therapy for the acute phase of NASH, which has high morbidity and mortality.
Figures
References:
1.. Perumpail BJ, Khan MA, Yoo ER, Clinical epidemiology and disease burden of non-alcoholic fatty liver disease: World J Gastroenterol, 2017; 23(47); 8263-76
2.. Matteoni CA, Younossi ZM, Gramlich T, Non-alcoholic fatty liver disease: A spectrum of clinical and pathological severity: Gastroenterology, 1999; 116(6); 1413-19
3.. Wong RJ, Aguilar M, Cheung R, Non-alcoholic steatohepatitis is the second leading etiology of liver disease among adults awaiting liver transplantation in the United States: Gastroenterology, 2015; 148(3); 547-55
4.. Chalasani N, Younossi Z, Lavine JE, The diagnosis and management of non-alcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases: Hepatology, 2018; 67(1); 328-57
5.. Sheka AC, Adeyi O, Thompson J, Nonalcoholic steatohepatitis: A review: JAMA, 2020; 323(12); 1175-83
6.. Younossi ZM, Otgonsuren M, Henry L, Association of non-alcoholic fatty liver disease (NAFLD) with hepatocellular carcinoma (HCC) in the United States from 2004 to 2009: Hepatology, 2015; 62(6); 1723-30
7.. Musso G, Cassader M, Rosina F, Impact of current treatments on liver disease, glucose metabolism and cardiovascular risk in non-alcoholic fatty liver disease (NAFLD): A systematic review and meta-analysis of randomised trials: Diabetologia, 2012; 55(4); 885-904
8.. Johnson NA, Sachinwalla T, Walton DW, Aerobic exercise training reduces hepatic and visceral lipids in obese individuals without weight loss: Hepatology, 2009; 50(4); 1105-12
9.. LaBrecque DR, Abbas Z, World Gastroenterology Organisation global guidelines: Non-alcoholic fatty liver disease and non-alcoholic steatohepatitis: J Clin Gastroenterol, 2014; 48(6); 467-73
10.. Belfort R, Harrison SA, Brown K, A placebo-controlled trial of pioglitazone in subjects with non-alcoholic steatohepatitis: N Engl J Med, 2006; 355(22); 2297-307
11.. Sanyal AJ, Chalasani N, Kowdley KV, Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis: N Engl J Med, 2010; 362(18); 1675-85
12.. , EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease: J Hepatol, 2016; 64(6); 1388-402
13.. Sumida Y, Yoneda M, Current and future pharmacological therapies for NAFLD/NASH: J Gastroenterol, 2018; 53(3); 362-76
14.. Newsome PN, Buchholtz K, Cusi K, A placebo-controlled trial of sub-cutaneous semaglutide in nonalcoholic steatohepatitis: N Eng J Med, 2021; 384; 1113-24
15.. Lassailly G, Caiazzo R, Buob D, Bariatric surgery reduces features of nonalcoholic steatohepatitis in morbidly obese patients: Gastroenterology, 2015; 149(2); 379-88
16.. Horton JD, Cohen JC, Hobbs HH, PCSK9: A convertase that coordinates LDL catabolism: J Lipid Res, 2008; 50(Suppl.); S172-77
17.. Kastelein JJ, Ginsberg HN, Langslet G, ODYSSEY FH I and FH II: 78-week results with alirocumab treatment in 735 patients with heterozygous familial hypercholesterolaemia: Eur Heart J, 2015; 36; 2996-3003
18.. Sabatine M, Giugliano R, Wiviott S, Efficacy and safety of evolocumab in reducing lipids and cardiovascular events: J Vasc Surg, 2015; 62(4); 1089
19.. Bandyopadhyay D, Ashish K, Hajra A, Cardiovascular outcomes of PCSK9 inhibitors: With special emphasis on its effect beyond LDL-cholesterol lowering: J Lipids, 2018; 2018; 1-13
20.. Nissen SE, Stroes E, Dent-Acosta RE, Efficacy and tolerability of evolocumab vs ezetimibe in patients with muscle-related statin intolerance: The GAUSS-3 randomized clinical trial: JAMA, 2016; 315(15); 1580-90
21.. Lee JS, Mukhopadhyay P, Matyas C, PCSK9 inhibition as a novel therapeutic target for alcoholic liver disease: Sci Rep, 2019; 9(1); 17167
22.. Ruscica M, Ferri N, Macchi C, Liver fat accumulation is associated with circulating PCSK9: Ann Med, 2016; 48(5); 384-91
23.. Wargny M, Ducluzeau PH, Petit JM, Circulating PCSK9 levels are not associated with the severity of hepatic steatosis and NASH in a high-risk population: Atherosclerosis, 2018; 278; 82-90
24.. Sun D-Q, Liu W-Y, Wu S-J, Increased levels of low-density lipoprotein cholesterol within the normal range as a risk factor for non-alcoholic fatty liver disease: Oncotarget, 2015; 7(5); 5728-37
Figures
Tables
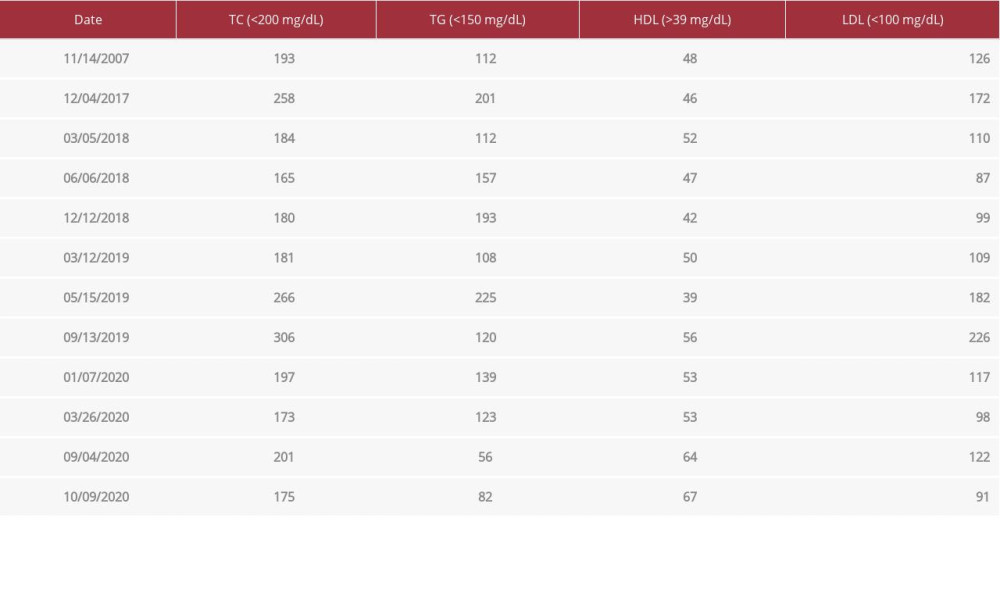 Table 1.. Lipid panel from initial diagnosis.
Table 1.. Lipid panel from initial diagnosis.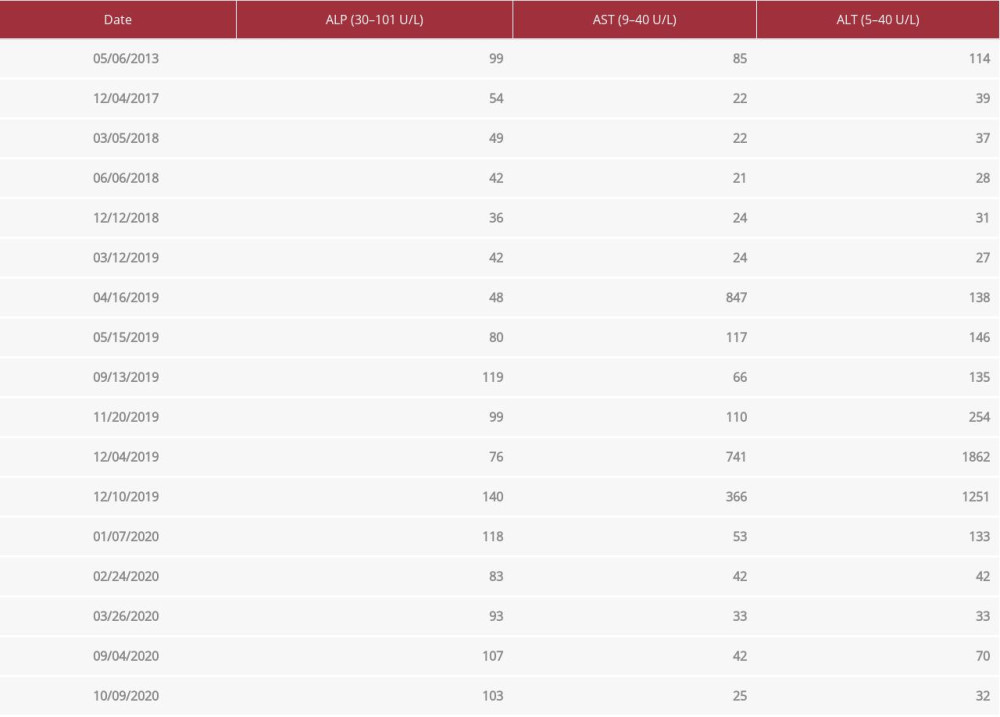 Table 2.. Liver function tests from initial diagnosis.
Table 2.. Liver function tests from initial diagnosis.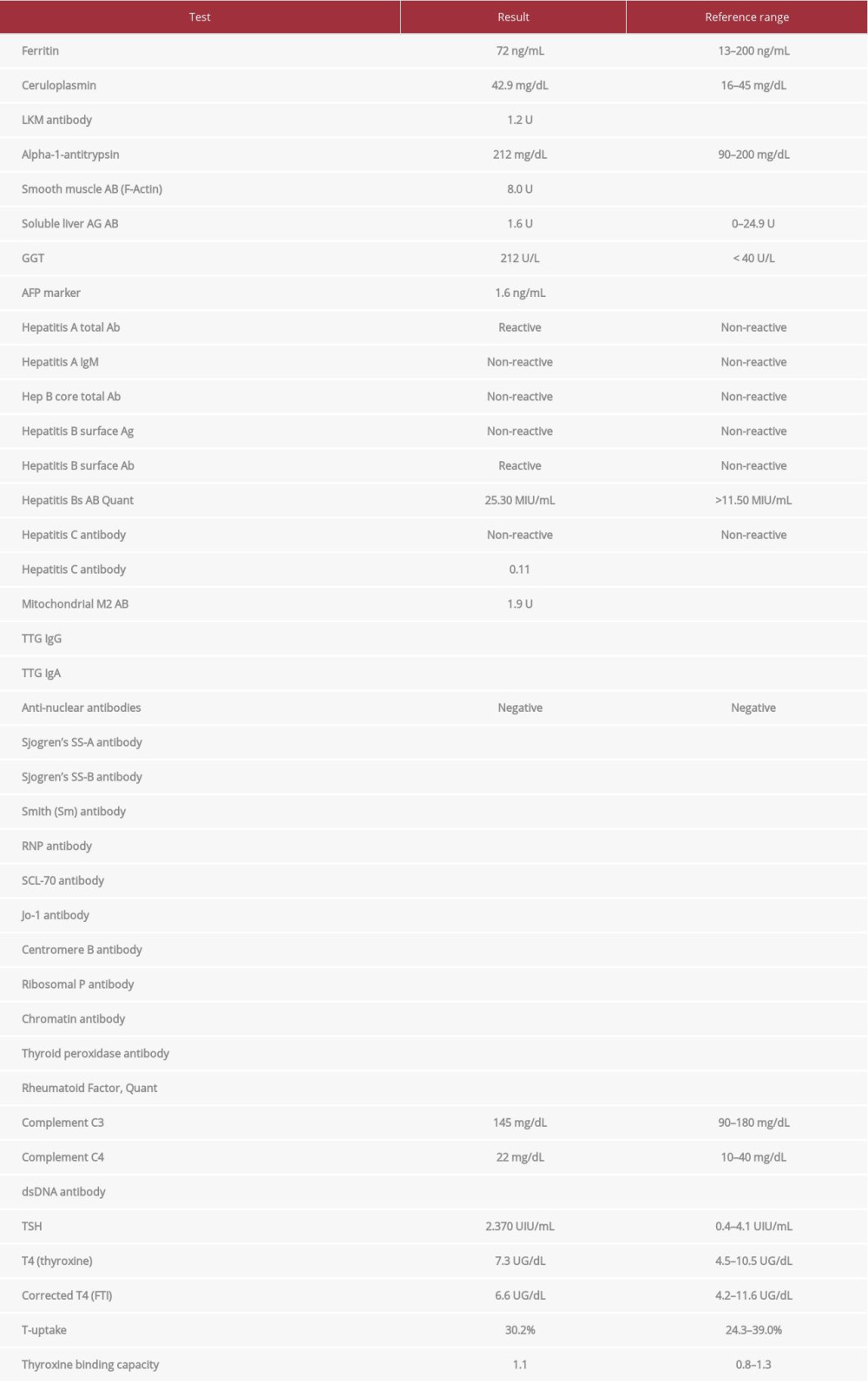 Table 3.. Laboratory studies before first liver biopsy.
Table 3.. Laboratory studies before first liver biopsy. Table 1.. Lipid panel from initial diagnosis.
Table 1.. Lipid panel from initial diagnosis. Table 2.. Liver function tests from initial diagnosis.
Table 2.. Liver function tests from initial diagnosis. Table 3.. Laboratory studies before first liver biopsy.
Table 3.. Laboratory studies before first liver biopsy. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








