24 January 2022: Articles 
A 63-Year-Old Man with a Diagnosis of Re-Infection with SARS-CoV-2 Nine Weeks After an Initial Hospital Admission with COVID-19 Pneumonia
Unusual clinical course, Challenging differential diagnosis, Management of emergency care, Educational Purpose (only if useful for a systematic review or synthesis)
Ilona Dudek1ABCDEF, Marzena Jesiotr1BC, Agnieszka Rzeszotarska1DEF, Krzysztof KłosDOI: 10.12659/AJCR.932999
Am J Case Rep 2022; 23:e932999
Abstract
BACKGROUND: This report describes a 63-year-old Polish man presenting with COVID-19 (Coronavirus Disease 2019) pneumonia in early 2020, before vaccines to prevent severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection were available. Nine weeks following recovery from the initial infection, he tested positive again for SARS-CoV-2.
CASE REPORT: Man, age 63, was admitted to the Military Institute of Medicine on March 12, 2020, with body temperature 40°C, a cough, and breathlessness. On March 12, 2020, SARS-CoV-2 RNA was found in a nasopharynx smear. A chest X-ray (RTG) showed discrete areas of interstitial densities. On June 13, 2020, after 32 days of hospitalization and 2 negative real-time polymerase chain rection (RT-PCR) test results, patient was released home in good general condition. On July 23, 2020 he reported to the emergency room with fever of 39°C and general weakness. A nasopharynx smear confirmed SARS-CoV-2 infection. On admission, the patient was in moderately good condition with auscultatory changes typical for pneumonia on both sides of the chest. On the seventh day of hospitalization, the patient was transported to the Intensive Care Unit (ICU) due to drastic deterioration in respiratory function. Respiratory support with non-invasive high-flow oxygen therapy (Opti-Flow) was used. On August 20, 2020, after negative RT-PCR test results, he was discharged in good general condition.
CONCLUSIONS: This case of COVID-19 pneumonia presented early in the COVID-19 pandemic of 2020, and the laboratory diagnosis of the initial and subsequent SARS-CoV-2 infection relied on the laboratory methods available at that time. However, several cases of repeat SARS-CoV-2 infection have been described before the development of vaccines in late 2020.
Keywords: COVID-19, COVID-19 Diagnostic Testing, Real-Time Polymerase Chain Reaction, COVID-19, Hospitalization, Hospitals, Humans, Male, Pandemics, RNA, Viral, reinfection, SARS-CoV-2, United States
Background
COVID-19 is an infectious disease caused by beta coronavirus and related to SARS-CoV-2. Currently, the disease constitutes a threat to global health security. On February 18, 2021 according to the World Health Organisation (WHO), 109 426 406 cases of SARS-CoV-2 infection and 2 419 363 deaths due to COVID-19 were confirmed [1]. SARS-CoV-2 is transmitted via airborne droplets, through close contact, or by discharge from the respiratory tract of an infected person. Symptoms occur 3 to 14 days after infection; the most common are fever over 38°C, cough, shortness of breath, and weakness. Other rarer symptoms include muscle and head pain, hemoptysis, and diarrhea [1,2]. SARS-CoV-2 is an enveloped virus, whose genome has positive polarity, single-stranded RNA composed of 29 903 nucleotides. The characteristic “crown” is constructed of glycoprotein S protruding on the surface. Glycoprotein S determines the recognition of receptors on the surface of the host cell and the penetration of the virus into it. The virus exhibits tropism to the epithelial cells lining the respiratory tract and the digestive system. The receptor for SARS-CoV-2 is the ACE protein (angiotensin-converting enzyme II), found in both humans and bats, which, among other roles, regulates contractions of small arteries and blood pressure [2]. Sigrist et al suggested that SARS-C0V-2 can connect to other receptors (integrin), which is a unique feature of glycoprotein S. This structure of SARS-CoV-2 can extend the tropism of the virus to various target cells and increase the pathogenic virulence [3]. Infection with SARS-CoV-2, similar to other pathogens, should cause the acquisition of immunity, especially the formation of immune memory, as well as the formation of neutralizing antibodies, which, upon repeated contact, would lead to an accelerated and effective response [4]. It is not clear whether the level of acquired immunity depends on other factors such as age or general health, which may influence the course of re-infection. There have been several reports of recontamination with the virus [5–8]. The time-course of PCR positivity and seroconversion may vary in children and other groups, including the large population of asymptomatic individuals who go undiagnosed without active surveillance. Many questions remain, particularly how long potential immunity lasts in individuals, both asymptomatic and symptomatic, who are infected with SARS-CoV-2 [9,10]. Therefore, this report describes the case of 63-year-old Polish man who was diagnosed with COVID-19 pneumonia in early 2020, before the SARS-CoV-2 vaccinations were available. Nine weeks after he had recovered from the initial infection, he again had positive test results for SARS-CoV-2 [9].
Case Report
RESULTS OF LABORATORY TESTS, CLINICAL INDICATORS, AND TREATMENT:
The treatment included empirical antibiotic therapy, recombinant human granulocyte growth factor, and COVID-19 therapy in line with the recommendations of the WHO Clinical Management of COVID-19 guidelines at that time (May 18, 2020) [9,15]. During hospitalization, his fever subsided, laboratory test indices improved, and inflammatory lesions regressed on the chest X-ray image. The first negative result was obtained on June 6, 2020 [7], the 31st day of hospitalization. After receiving the second negative result for the presence of the virus, the patient was discharged home on June 13, 2020 [7] in good general condition without further health problems. The patient’s immune response to virus infection was also checked, showing the weak reaction of the immune system, with the production of IgM antibodies (10 501) and the lack of IgG antibodies (1279) for SARS-CoV-2 (Table 2). Determination of the antibody titer was performed with the use of a COVID-19 ELISA IgM + IgA kit and COVID-19 ELISA IgG (Vircell Microbiologists, Spain) [16] on the Dynex Magellan Biosciences apparatus. The results were read using DS Matrix 1.40.3 software (Table 3). After 7 weeks at home, on June 13, 2020, the patient reported to the MIM Hospital Emergency Department with a cough and a 7-day fever that did not respond to antipyretic medications. The chest X-ray revealed diffused interstitial compaction in the upper field of the right lung and peribronchial compaction in the middle and lower fields on the left side. Laboratory tests showed high markers of inflammation (CRP: 14.29 ng/dl [0–0.8 mg/dl]; procalcitonin: 0.22 ng/ml [0.05–0.1 ng/ml]), slight abnormalities in the peripheral blood smear, and increased activity of transaminases (ALT: 103 U/l [0–41 U/l]). SARS-CoV-2 virus RNA was not detected in 3 nasopharyngeal swabs taken. Despite empirical broad-spectrum antibiotic therapy, the patient still had a fever. Blood cultures were performed 4 times, and no microorganisms or presence of genetic material of Epstein-Barr viruses and cytomegaloviruses were detected. Computed tomography of the chest showed extensive interstitial changes in the upper and middle lobes of both lungs, with the presence of a small amount of fluid in both pleural cavities. Diagnostic bronchoscopy with aspirate collection was performed, in which the presence of Candida albicans colonies was detected. After modification of antimicrobial treatment and steroid therapy, quick clinical improvement was achieved. On day 15, the patient was discharged home in good general condition. On July 23, 2020 [7], the patient returned to the Emergency Department due to a high fever of 39ºC and severe general weakness. He denied coughing and chest pain. He was admitted to the MIM Department of Infectious Diseases and Allergology after a several-hour stay in the isolation room and detection of SARS-CoV-2 virus genetic material in a nasopharyngeal swab. Real-Time PCR was performed using the GeneFinder COVID-19 Plus RealAmp Kit (GeneFinder Anyang, Gyeonggi, Korea, LOT: 2009-R45-05) as described above (Figure 3, Table 4).
On admission, the patient was in moderately good condition, normotension was noted, and auscultation changes typical for pneumonia on both sides of the chest were found. Sat. O2 measured with a pulse oximeter while breathing room air was 92%. In laboratory tests, elevated markers of inflammation were observed (CRP 14.3 ml/dl [0–0.8 mg/dl]; procalcitonin: 0.16 ng/ ml [0.05–0.1 ng/ml]) as well as persistent slight lymphopenia in the peripheral blood smear (lymphocytes: 0.29×103/l [0.9–4.5×103/ml]). A bedside chest X-ray showed parenchymal densification with massive consolidation of shadows in the cavity and in the peripheral parts of both lungs. Due to the increasing inflammatory parameters and the deterioration of the patient’s clinical condition, as indicated by exacerbation of respiratory failure requiring oxygen supplementation through a mask with a reservoir at maximum flows of 15 L/mn, negative results of atypical infections (Legionella pneumophila, Mycoplasma pneumoniae, and Chlamydia pneumoniae), and failure of modified respiratory failure therapy, the patient was transferred to the Intensive Care Unit. Respiration was supported with non-invasive high-flow oxygen therapy (Opti-Flow). In the following days, gradual improvement in the patient’s clinical condition occurred, respiratory efficiency increased, inflammatory markers normalized, and partial regression of inflammatory changes and complete remission of fluid in the pleural cavities in the chest X-ray scan were observed. On August 8, after 2 consecutive negative results of nasopharyngeal swabs for SARS-CoV-2 virus were obtained, the patient was discharged home in good general condition (Figure 1: Course of the patient’s infection).
Discussion
We present a case of COVID-19 pneumonia reported at the beginning of the COVID-19 pandemic in 2020. The laboratory diagnosis of initial and subsequent SARS-CoV-2 infection were based on laboratory methods available at that time. However, several cases of SARS-CoV-2 re-infection were reported prior to vaccine development in late 2020 [8,17,18]. The available data in the literature suggest that re-obtaining a positive RT-PCR result in convalescents may be a fairly common phenomenon, while there is little research on re-infection with SARS-COV-2.
In a study by Yuan et al, in 25 out of 172 patients discharged from the hospital, SARS-CoV-2 was detected again after an average of 7 days [19]. In a group of 55 survivors, 5 (9%) had another positive RT-PCR result between 4 and 17 days [14]. However, Zhang et al described the case of a 57-year-old patient in whom the SARS-CoV-2 virus was reappeared on day 32 [20]. In a study by Tang et al in a group of 70 patients, 15 (21.4%) patients discharged from hospital were positive again between 21 and 36 days. In the above-cited studies, some patients with a positive test result showed mild symptoms such as sore throat, dry cough, and fever, but the others showed no symptoms. The health status of the patients, despite the positive result, did not indicate an active infection. In the case of our patient, the re-detection of the SARS-CoV-2 virus in the 7th week after obtaining 2 consecutive negative results correlated with a significant deterioration of health, requiring intensive medical care and oxygen therapy. The conducted tests excluded the possibility of bacterial and opportunistic infections. The patient’s health status and the results of RTG and RT-PCR clearly indicated re-infection caused by SARS-CoV-2. The long time since the first negative result was obtained and re-testing positive may reflect a long process of elimination of ribonucleic acid from the tissues. Data available in the literature suggest that shedding of SARS-CoV-2 takes an average of 20–22 days after the onset of symptoms, with shedding lasting up to 44 days in some cases [21]. For many other viral diseases, such as Zika, RNA can be detected long (several weeks) after infectious virus clearance [22]. The age and severity of the initial infection may also correlate with prolonged viral shedding and a positive re-test. In a 71-year-old woman with severe COVID-19, positive RT-PCR results were observed for 60 days from the onset of symptoms and 36 days after complete symptom relief [13,23]. A similar case of an 82-year-old severely infected patient was reported by Duggan et al [21], in which 2 negative smears were obtained on days 39 and 40 of the disease in combination with symptomatic recovery, and on the 55th day after the onset of the first symptoms, another positive result was obtained [21]. In our 63-year-old patient, on day 55 from the diagnosis of the infection, another 2 negative results were obtained. On day 20, a questionable result was obtained (only the N gene was detected). For the next 32 days, the results obtained were alternating, either questionable (no RdRp core gene) or negative (32 and 45 days). During this time, the patient showed improvement in health, which did not indicate an active infection. In the other hand, Tillett and coworkers described an investigation of 2 instances of SARS-CoV-2 infection in the same individual [23]. A 25-year-old had 2 positive tests for SARS-CoV-2, the first on April 18, 2020, and the second on June 5, 2020, separated by 2 negative tests done during follow-up in May 2020. In that study, the genomic analysis of SARS-CoV-2 showed genetically significant differences between each variant associated with each instance of infection [23]. As in our case, the second infection was symptomatically more severe than the first.
We did not perform genomic analysis, but considering that the patient was in the ICU due to drastic deterioration in respiratory function, we suspected re-infection. Sharma et al reported re-infection in a middle-aged man with asymptomatic SARS-CoV-2 infection who later developed mild symptomatic COVID-19 after a period of 3 months [24]. During the second infection, a real-time PCR test and tests for the presence of COVID-19 immunoglobulin IgM and IgG antibodies were positive [24]. The researchers speculated that asymptomatic infections may not provide long-term protective immunity to all patients, which could make them susceptible to re-infection [24]. In our study, a 63-year-old man had symptoms during the first and second infection, but our patient did not develop immunity during the first infection due to a comorbid disease, and thus he developed a severe re-infection. Hoang et al described recurrence of positive SARS-CoV-2 PCR results in patients discharged from hospital after 2 consecutive negative PCR results [11]. They speculated that the first negative results were false-negative, and the re-positive results indicated a significantly longer nucleic acid conversion time. However, unlike our patient, these patients did not have recurrence of symptoms coinciding with the repeat positive results [9,11,25]. Alternating negative and positive results may be associated with an insufficient amount of virus when its titer reaches a level close to the lower detection limit of the test. The success of virus isolation and testing depends on the initial amount of virus on the swab. Samples containing <106 copies/ml (or copies per sample) did not produce a good-quality isolate [13]. Researchers note that re-testing positive often occurs in late reaction cycles (>35 CT) in real time, which in turn indicates a low initial viral load in the sample [16,21]. In our case, doubtful/positive results between day 20 and day 55 were characterized by a late reaction cycle (>35 CT). Scientists also suggest that, as in our patient, the detection of only the N gene, which is characterized by the highest purity, may indicate an insufficient amount of the virus, which is related to a decrease in the titer of virus excreted by the patient or an incorrectly collected swab [26,27]. In turn, the re-detection of the SARS-CoV-2 virus (in the 7th week from the last 2 negative results) occurred in about 23 CT, which in turn proves the high content of the virus and its re-multiplication. Statistically compiled clinical data indicate that anti-SARS-CoV-2 IgM antibodies sometimes become detectable 3 days after the onset of symptoms. IgG anti-SARS-CoV-2 antibodies start to increase along with the decrease in IgM concentration after about 10 days, although they can be identified as early as day 7 [28]. IgG antibodies, indicating acquired immunity, last for about 2-3 months [27]. Studies on macaques [29] and epidemiological studies involving SARS and MERS virus infection show that the presence of anti-SARS-CoV-2 IgG antibodies protects patients against re-infection with SARS-CoV-2 [28,30]. In our patient, on the 35th day after detection of infection, the concentration of IgM (10 501) indicated an infection. IgM antibodies began to wear off on day 59 (7946) and remained low (5005) despite a relapse at week 15 after the infection was discovered. Throughout the course of the disease, the patient did not develop IgG antibodies, the mean titer of which was 1.393. The patient probably did not develop immunity during the first infection due to a comorbid disease (multiple myeloma); therefore, he developed a severe re-infection requiring hospitalization. Jing et al showed that the low lymphocyte count in 25 patients re-infected with SARS-CoV-2 virus was significantly positively correlated (r=0.52,
RT-PCR testing to distinguish an active virus from its RNA fragments is expensive. When monitoring active infection, quick cassette tests are recommended [31]. The frequency of antigen tests is generally lower than that of RT-PCR tests, although their specificity is comparable [31]. It has been shown that antigen tests are more effective in detecting active infection when viral load in the nasopharynx is higher [31]. Antigen tests give a false-negative result when the patient is asymptomatic or in the final or early stage of infection, or when the viral load is below the detection limit of the test. Scientists suggest that only the infectivity/culture test by inoculating cell lines (eg, Vero/hSLAM or Vero/E6 cells) with the stalk virus from a patient’s nasopharyngeal swab [17,25] gives 100% confidence. Korean scientists showed that a repeated positive RTPCR result in 260 subjects was associated with the detection of RNA fragments [32]. Therefore, in patients with clinical improvement without symptoms and resolution of radiographic changes, as in the cases described by Lan et al [14] and others [16,19,25], a repeat positive RT-PCR test result does not reflect re-infection. Kang et al emphasized that the COVID-19 patients who tested positive again for SARS-CoV-2 should be managed differently than patients with a first-time positive test result [33]. Patients with COVID-19 (who received glucocorticoid therapy, had comorbidities, were older than 65) may have longer hospital stay because of the prolonged clearance of virus [33]. These discharged patients with COVID-19 should be under quarantine management and health monitoring for 14 days, instead of “self-monitoring for 14 days” [9,25,33].
Conclusions
This case of COVID-19 pneumonia early in the COVID-19 pandemic (2020) and the laboratory diagnosis of the initial and subsequent SARS-CoV-2 infection relied on the laboratory methods available at that time. However, several cases of repeat SARS-CoV-2 infection have been described before the development of vaccines in late 2020. In case of repeated positive results, the patient’s health assessment should include antibody response, including IgM and IgG, lymphocyte levels, and detailed clinical assessment, followed by further follow-up after discharge from the hospital. Patients with coexisting diseases for whom the elimination period was extended (>22 days) should be carefully monitored. Although the literature indicates that RNA re-detection does not necessarily mean that active virus is present, and re-infection is unlikely [23,28,34], our case of a 63-year-old patient clearly indicates the possibility of re-infection with SARS-CoV-2 virus.
Figures
Tables
Table 1.. Results of laboratory tests, clinical indicators and treatment. Included empirical antibiotic therapy, recombinant human granulocyte growth factor and Covid-19 therapy in line with the WHO recommendations for clinical management of COVID-19 at that time (May 18, 2020).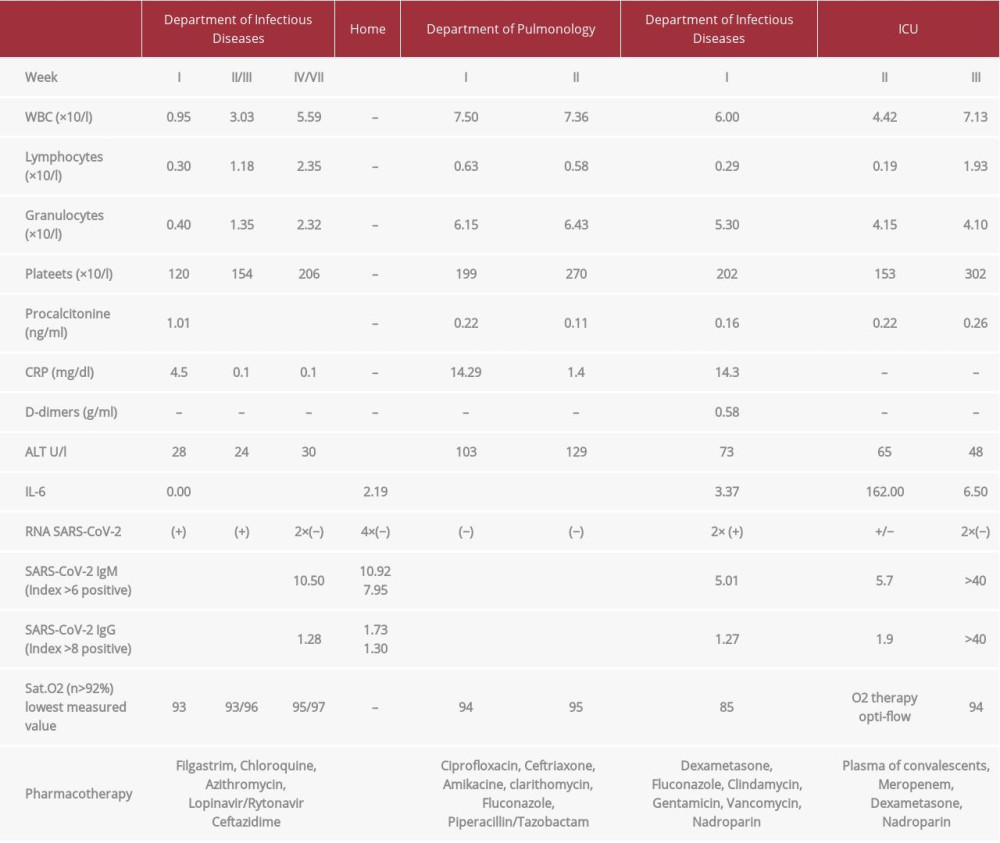 Table 2.. Interpretation of anti-SARS-CoV-2 antibodies results.
Table 2.. Interpretation of anti-SARS-CoV-2 antibodies results.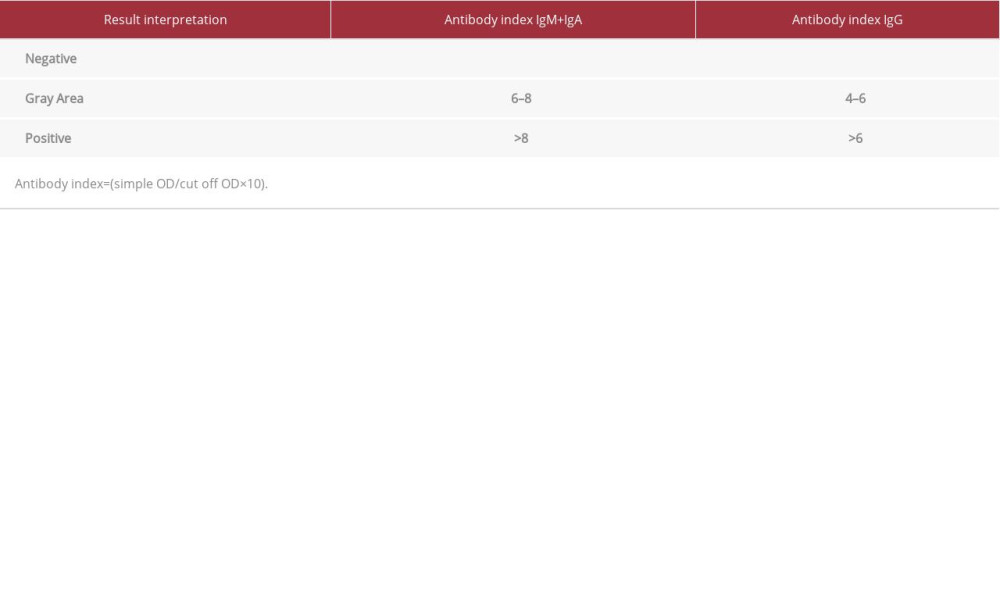 Table 3.. Results of the determined SARS-CoV-2 antibodies in the IgM+IgA class and in the IgG class in our patient during his stay at the Military Institute of Medicine Department of Infectious Diseases and Allergology.
Table 3.. Results of the determined SARS-CoV-2 antibodies in the IgM+IgA class and in the IgG class in our patient during his stay at the Military Institute of Medicine Department of Infectious Diseases and Allergology.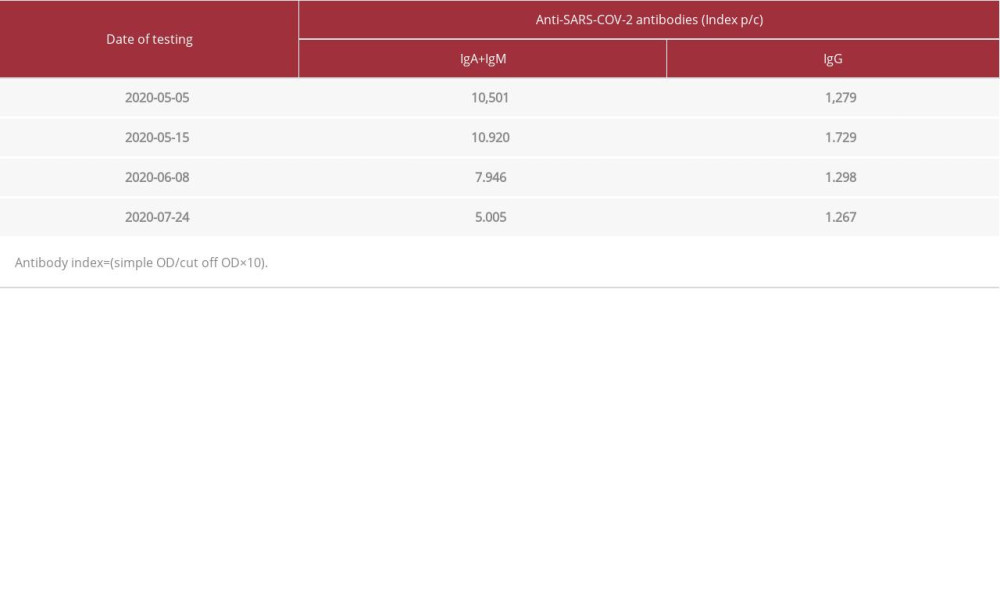 Table 4.. Results of the presence of the genetic material of the SARS-CoV-2 virus in nasopharyngeal swabs taken from our patient during his stay at the Department of Infectious Diseases and Allergology of the Military Institute of Medicine.
Table 4.. Results of the presence of the genetic material of the SARS-CoV-2 virus in nasopharyngeal swabs taken from our patient during his stay at the Department of Infectious Diseases and Allergology of the Military Institute of Medicine.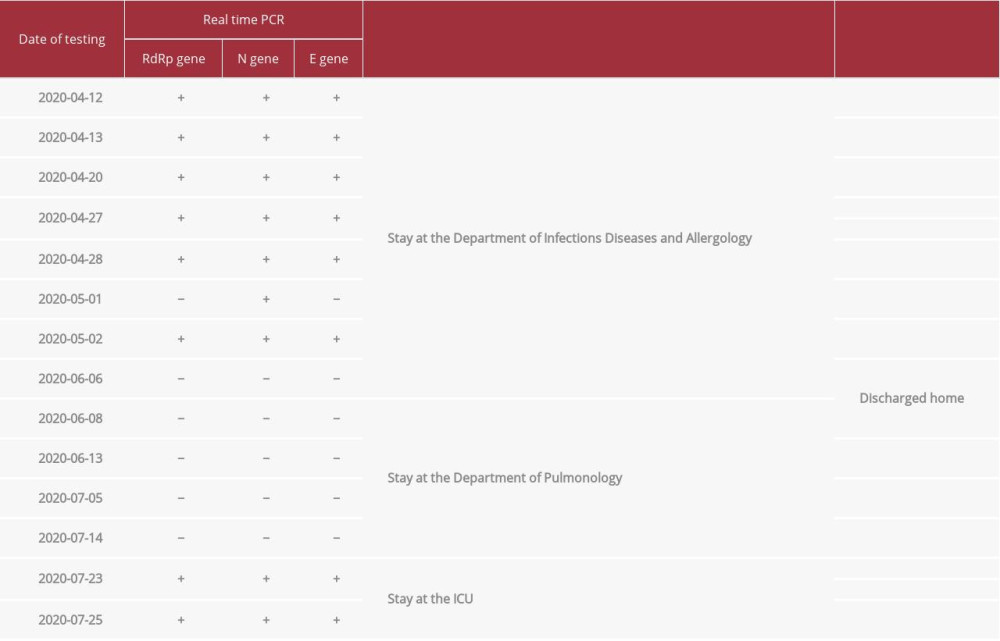
References:
1.. Andersen KG, Rambaut A, Lipkin WI, The proximal origin of SARSCoV-2: Nat Med, 2020; 26(4); 450-52
2.. Zhou P, Yang XL, Wang XG, A pneumonia outbreak associated with a new coronavirus of probable bat origin: Nature, 2020; 579(7798); 270-73
3.. Sigrist JA, Bridge A, Le Mercier P, A potential role for integrins in host cell entry by SARS-CoV2: Antiviral Res, 2020; 177; 104759
4.. Ju B, Zhang Q, Ge J, Human neutralizing antibodies elicited by SARSCoV2 infection: Nature, 2020; 584; 115-19
5.. KK-W To, IF-N Hung, JD Ip, COVID-19 re-infection by phylogenetically destined SARS-coronavirus-2 strain confirmed by whole genome sequencing: Clin Infect Dis, 2020 [Online ahead of print]
6.. Van Elslaude J, Vermeenrsch P, Vandeeroort K, Symptomic SARSCoV2 reinfection by a phylogenetically destined strain: Clin Infect Dis, 2020; 73(2); 354-56
7.. Prado-Vivar B, Becerra-Wang M, Guadalupe JJ, COVID-19 re-infection by a phylogenetically destined SARS-CoV2 variant first confirmed event inSouth America: SSRN Aug 25, 2020; ciaa1275
8.. , WHO Guideline Development group for Clinical management of COVID-19, 18 May 2020 Available from: https://cdn.who.int/media/docs/default-source/documents/emergencies/covid-19-guideline-development-group-clinical-management-bios.pdf?sfvrsn=3aab40bb_2
9.. Tillett RL, Sevinsky JR, Hartley PD, Genomic evidence for reinfection with SARS-CoV-2: A case study: Lancet Infect Dis, 2021; 21(1); 52-58
10.. Sethuraman N, Jeremiah SS, Ryo A, Interpreting diagnostic tests for SARSCoV-2: JAMA, 2020; 323(22); 2249-51
11.. Hoang VT, Dao TL, Gautret P, Recurrence of positive SARS-CoV-2 in patients recovered from COVID-19: J Med Virol, 2020; 92(11); 2366-67
12.. , GeneFinderTM COVID-19 Plus RealAmp Kit Available at: https://www.fda.gov/media/137116/download
13.. Li J, Zhang L, Liu B, Song D, Case report: Viral shedding for 60 days in a woman with COVID-19: Am J Trop Med Hyg, 2020; 102(6); 1210-13
14.. Lan L, Xu D, Ye G, Positive RT-PCR test results in patients recovered from COVID-19: JAMA, 2020; 323(15); 1502-3
15.. Yuan J, Kou S, Liang Y, Polymerase chain reaction assays reverted to positive in 25 discharged patients With COVID-19: Clin Infect Dis, 2020; 71(16); 2230-32
16.. Xiao AT, Tong YX, Zhang S, False negative of RT-PCR and prolonged nucleic acid conversion in COVID-19: Rather than recurrence: J Med Virol, 2020; 92(10); 1755-56
17.. Caly L, Druce J, Roberts J, Isolation and rapid sharing of the 2019 novel coronavirus (SARS-CoV-2) from the first patient diagnosed with COVID-19 in Australia: Med J Aust, 2020; 212(10); 459-62
18.. , Topic: Can people with previous COVID-19 infection become re-infected by the SARS-CoV-2virus? [updated May 122020] © 2020: Alberta Health Services May 12, 2020
19.. Yea G, Panb Z, Pana Y, Clinical characteristics of severe acute respiratory syndrome coronavirus 2 reactivation: J Infect, 2020; 80(5); e14-17
20.. Zhang JF, Yan K, Ye HH, SARS-CoV-2 turned positive in a discharged patient with COVID-19 arouses concern regarding the present standard for discharge: Int J Infect Dis, 2020; 97; 212-14
21.. Duggan NM, Ludy SM, Shannon BC, Is novel coronavirus 2019 reinfection possible? Interpreting dynamic SARS-CoV-2 test results: Am J Emerg Med, 2021; 39; 256.e1-e3
22.. Villamil-Gomez WE, Guijarro E, Castellanos J, Congenital Zika syndrome with prolonged detection of Zika virus RNA: J Clin Virol, 2017; 95; 52-54
23.. Zhou F, Yu T, Du R, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study: Lancet, 2020; 395(10229); 1054-62
24.. Sharma R, Sardar S, Mohammad Arshad A, Patient with asymptomatic SARS-CoV-2 infection who presented 86 days later with COVID-19 pneumonia possibly due to reinfection with SARS-CoV-2: Am J Case Rep, 2020; 21; e927154
25.. Wölfel R, Corman VM, Guggemos W, Virological assessment of hospitalized patients with COVID-2019: Nature, 2020; 581(7809); 465-69
26.. Nalla AK, Casto AM, Huang MW, Comparative performance of SARSCoV-2 detection assays using seven different primer/probe sets and one assay kit: J Clin Microbiol, 2020; 58(6); e00557-20
27.. Yoo SY, Lee Y, Lee GH, Kim DH, Reactivation of SARS-CoV-2 after recovery: Pediatr Int Jul, 2020; 62(7); 879-81
28.. Chaturvedi R, Naidu R, Sheth S, Chakravarthy K, Efficacy of serology testing in predicting reinfection in patients with SARS-CoV-2: Disaster Med Public Health Prep, 2020 [Online ahead of print]
29.. Bao L, Deng W, Gao H, Reinfection could not occur in SARS-CoV-2 infected rhesus macaques: bioRxiv, 2020; 2020; 990226
30.. Aldridge RW, Lewer D, Beale S, Seasonality and immunity to laboratory-confirmed seasonal coronaviruses (HCoV-NL63, HCoV-OC43, and HCoV-229E): Results from the Flu Watch cohort study: Wellcome Open Res, 2020; 5; 52
31.. Candel FJ, Barreiro P, San Román J, Recommendations for use of antigenic tests in the diagnosis of acute SARS-CoV-2 infection in the second pandemic wave: Attitude in different clinical settings: Rev Esp Quimioter, 2020; 33(6); 466-84
32.. Bo-gyung K, Tests in recovered patients found false positives, not reinfections, experts say: The Korea Herald, 2020 Available at: http://www.koreaherald.com/view.php?ud=20200429000724&fbclid=IwAR2txK96Lvzsw9cdAmDYJcyNf-wZsE2_lnLhOXUB5LIsV6_qmt6tfX6SfOY
33.. Kang H, Wang Y, Tong Z, Liu X, Retest positive for SARS-CoV-2 RNA of “recovered” patients with COVID-19: Persistence, sampling issues, or re-infection?: J Med Virol, 2020; 92(11); 2263-65
34.. Alizargar J, Risk of reactivation or reinfection of novel coronavirus (COVID-19): J Formos Med Assoc, 2020; 119; 1123
Figures
Tables
 Table 1.. Results of laboratory tests, clinical indicators and treatment. Included empirical antibiotic therapy, recombinant human granulocyte growth factor and Covid-19 therapy in line with the WHO recommendations for clinical management of COVID-19 at that time (May 18, 2020).
Table 1.. Results of laboratory tests, clinical indicators and treatment. Included empirical antibiotic therapy, recombinant human granulocyte growth factor and Covid-19 therapy in line with the WHO recommendations for clinical management of COVID-19 at that time (May 18, 2020). Table 2.. Interpretation of anti-SARS-CoV-2 antibodies results.
Table 2.. Interpretation of anti-SARS-CoV-2 antibodies results. Table 3.. Results of the determined SARS-CoV-2 antibodies in the IgM+IgA class and in the IgG class in our patient during his stay at the Military Institute of Medicine Department of Infectious Diseases and Allergology.
Table 3.. Results of the determined SARS-CoV-2 antibodies in the IgM+IgA class and in the IgG class in our patient during his stay at the Military Institute of Medicine Department of Infectious Diseases and Allergology. Table 4.. Results of the presence of the genetic material of the SARS-CoV-2 virus in nasopharyngeal swabs taken from our patient during his stay at the Department of Infectious Diseases and Allergology of the Military Institute of Medicine.
Table 4.. Results of the presence of the genetic material of the SARS-CoV-2 virus in nasopharyngeal swabs taken from our patient during his stay at the Department of Infectious Diseases and Allergology of the Military Institute of Medicine. Table 1.. Results of laboratory tests, clinical indicators and treatment. Included empirical antibiotic therapy, recombinant human granulocyte growth factor and Covid-19 therapy in line with the WHO recommendations for clinical management of COVID-19 at that time (May 18, 2020).
Table 1.. Results of laboratory tests, clinical indicators and treatment. Included empirical antibiotic therapy, recombinant human granulocyte growth factor and Covid-19 therapy in line with the WHO recommendations for clinical management of COVID-19 at that time (May 18, 2020). Table 2.. Interpretation of anti-SARS-CoV-2 antibodies results.
Table 2.. Interpretation of anti-SARS-CoV-2 antibodies results. Table 3.. Results of the determined SARS-CoV-2 antibodies in the IgM+IgA class and in the IgG class in our patient during his stay at the Military Institute of Medicine Department of Infectious Diseases and Allergology.
Table 3.. Results of the determined SARS-CoV-2 antibodies in the IgM+IgA class and in the IgG class in our patient during his stay at the Military Institute of Medicine Department of Infectious Diseases and Allergology. Table 4.. Results of the presence of the genetic material of the SARS-CoV-2 virus in nasopharyngeal swabs taken from our patient during his stay at the Department of Infectious Diseases and Allergology of the Military Institute of Medicine.
Table 4.. Results of the presence of the genetic material of the SARS-CoV-2 virus in nasopharyngeal swabs taken from our patient during his stay at the Department of Infectious Diseases and Allergology of the Military Institute of Medicine. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








