30 January 2022: Articles 
Distal Renal Tubular Acidosis Associated with Autoimmune Diseases: Reports of 3 Cases and Review of Mechanisms
Unusual clinical course, Challenging differential diagnosis, Diagnostic / therapeutic accidents, Unusual setting of medical care, Educational Purpose (only if useful for a systematic review or synthesis), Rare coexistence of disease or pathology
Marcelo Augusto Duarte Silveira12ABDEF*, Antônio Carlos Seguro23DEF, Samirah Abreu Gomes34DEF, Maria Helena Vaisbich5DEF, Lúcia Andrade23ADEFDOI: 10.12659/AJCR.933957
Am J Case Rep 2022; 23:e933957
Abstract
BACKGROUND: Distal renal tubular acidosis (dRTA) is a defect in the urinary acidification process that limits the elimination of protons [H+] by alpha intercalated cells in the collecting tubules, with consequent metabolic acidosis with a normal plasma anion gap. The relationship between this tubulopathy and immune-mediated diseases like Sjögren syndrome, rheumatoid arthritis, autoimmune hepatitis, primary biliary cirrhosis, systemic lupus erythematosus, and thyroiditis is well known. Further, the pathophysiological mechanisms are diverse, but, unfortunately, many are not yet fully understood. We report 3 cases of dRTA in patients with different autoimmune diseases and review the pathophysiological mechanisms already described.
CASE REPORT: The first case involved a 29-year-old woman with autoimmune hepatitis. She had metabolic acidosis with persistent hypokalemia, and a kidney stone was also identified. The second case involved a 67-year-old woman diagnosed with rheumatoid arthritis. She had metabolic acidosis with hypokalemia. The third case involved a 30-year-old woman with Sjögren syndrome and persistent metabolic acidosis. In addition to the presence of metabolic acidosis with a normal plasma anion gap, all 3 patients exhibited urine with a supraphysiologic pH (above 5.3).
CONCLUSIONS: Autoimmune diseases may be associated with deficits in urinary acidification with consequent metabolic acidosis and, therefore, systemic repercussions. This association must be remembered and researched because correct diagnosis and treatment will serve to reduce complications.
Keywords: Acidosis, Renal Tubular, Autoimmune Diseases, Physiology, Adult, Female, Hepatitis, Autoimmune, Humans, Hypokalemia, Kidney Calculi, Sjogren's Syndrome
Background
Renal tubular acidosis (RTA) is an impairment of the urinary acidification mechanism, which can involve any of the following steps: reabsorption, regeneration (through carbonic anhydrases), and bicarbonate generation, as well as the excretion of protons (mainly in the form of ammonium) [1,2]. Various pathologies and disorders can cause RTA [1]. Patients with RTA typically have a normal glomerular filtration rate and hyper-chloremic metabolic acidosis [1].
Distal RTA (dRTA), also known as type 1 RTA, was the first form of the condition to be described. It is characterized by an inability to eliminate protons via the alpha-type intercalated cells in the collecting tubule and a consequent inability to achieve final urinary acidification [1]. In patients with dRTA, the urinary pH is usually above 5.3, even in the presence of metabolic acidosis. Other clinical features are hypokalemia or normal serum potassium level, growth deficit, rickets, hypercalciuria (usually in childhood), hypocitraturia, nephrolithiasis, and nephrocalcinosis [1,3,4]. The difficulty in excreting protons translates to reduced excretion of the ammonium ion, which promotes a positive urinary anion gap [1]. In addition, dRTA can be due to genetic defects or secondary to systemic diseases such as some autoimmune diseases [5]. Homozygous and heterozygous mutations with autosomal dominant or recessive transmission have been described in the chloride/bicarbonate exchanger or band 3 (AE1) and proton adenosine triphosphatase (H+-ATPase) subunits present in the basolateral and luminal membranes of alpha-type intercalated cells, respectively [5–8].
The autoimmune diseases reportedly associated with dRTA include Sjögren syndrome, rheumatoid arthritis, autoimmune hepatitis, primary biliary cirrhosis, systemic lupus erythematosus, and thyroiditis, although the mechanisms involved have yet to be fully clarified [2,8]. Here, we report 3 cases of dRTA associated with immune-mediated diseases and review their underlying pathogenic mechanisms.
Case Reports
CASE 1:
A 29-year-old woman who had been diagnosed, at 10 years of age, with chronic liver disease (Child-Pugh score, B8) due to autoimmune hepatitis, presented with metabolic acidosis and persistent hypokalemia. Serological testing was negative for viral hepatitis and positive for anti-smooth muscle antibodies (titer 1: 160). Blood tests showed the following: urea, 11 mg/dL; creatinine, 1.03 mg/dL; sodium, 139 mEq/L; potassium, 3.4 mEq/L; chlorine, 115 mEq/L; phosphorus, 2.9 mg/dL; total bilirubin 1.44 mg/dL (direct bilirubin 0.82 mg/dL and indirect bilirubin 0.62 mg/dL); albumin, 2.0 g/dL; thyroid stimulating hormone (TSH), 1.12 µIU/mL; arterial blood gas pH, 7.30; PCO2 35.5; bicarbonate, 17 mEq/L; and anion gap (corrected for albumin), 12. Urine tests showed the following: pH, 7.0; erythrocytes, absent; protein, absent; sodium, 118 mEq/L; potassium, 20 mEq/L; chlorine, 109 mEq/L; citrate, 117 mg/day; calcium, 89 mg/day; and anion gap, 29 (positive). Abdominal computed tomography revealed small renal calculi (Figure 1). On the basis of the biochemical profile, a diagnosis of dRTA was made. The patient was started on treatment with 38 mEq/ day of alkali (in bicarbonate and citrate forms) and 24 mEq/ day of oral potassium.
CASE 2:
A 67-year-old woman who had been diagnosed with rheumatoid arthritis (RA) at 33 years of age was hospitalized for severe peripheral arterial disease (PAD), which necessitated amputation of her left leg. The amputation procedure was elective, there was no presence of dehydration, and glomerular filtration was normal. During hospitalization, metabolic acidosis with persistent hypokalemia was noted. Blood tests showed the following: urea, 40 mg/dL; creatinine, 0.51 mg/dL; potassium, 3.1 mEq/L; magnesium, 1.72 mg/dL; phosphorus, 3.4 mg/dL; TSH, 2.4 uIU/mL; pH, 7.38; bicarbonate, 15.8 mEq/L; chlorine, 113 mEq/L; albumin, 2.2; and anion gap (corrected for albumin), 11. Urine tests showed the following: pH, 6.5; erythrocytes, absent; protein, absent; sodium, 31 mEq/L; potassium, 48 mEq/L; chlorine, 20 mEq/L; citrate, 111 mg/day; and anion gap, 59 (positive). Renal ultrasound showed no alterations.
On the basis of these findings, the patient was diagnosed with dRTA and started on treatment with potassium citrate (20 mEq/day) and oral potassium chloride (48 mEq/day), to which she responded well.
CASE 3:
A 30-year-old woman with recurrent arthritis presented with a 6-month history of inflammation of the parotid gland, and was admitted to the hospital. A serum anti-Sjögren syndrome antibody (SSA) test was positive and salivary gland biopsy confirmed the suspected diagnosis of Sjögren syndrome. Blood tests showed persistent metabolic acidosis: urea, 27 mg/dL; creatinine, 0.70 mg/dL; sodium, 136 mEq/L; potassium, 3.3 mEq/L; phosphorus 3.2 mg/dL chlorine, 107 mEq/L; pH, 7.36; bicarbonate, 17 mEq/L; anion gap, 12; TSH, 1.8 uIU/mL. Anti-nuclear antibody (ANA), Anti-DNA, and Anti-smooth muscle antibody (ASMA) were negative. Urine tests showed the following: pH, 7.0; erythrocytes, absent; protein, absent; sodium, 52 mEq/L; potassium, 17 mEq/L; chlorine, 43 mEq/L; citrate, 129 mg/day; and anion gap, 26 (positive). Renal ultrasound showed only a simple cyst in the right kidney and no stones. The patient was started on treatment with potassium citrate (20 mEq/day) and oral potassium chloride (24 mEq/day), with metabolic control.
Discussion
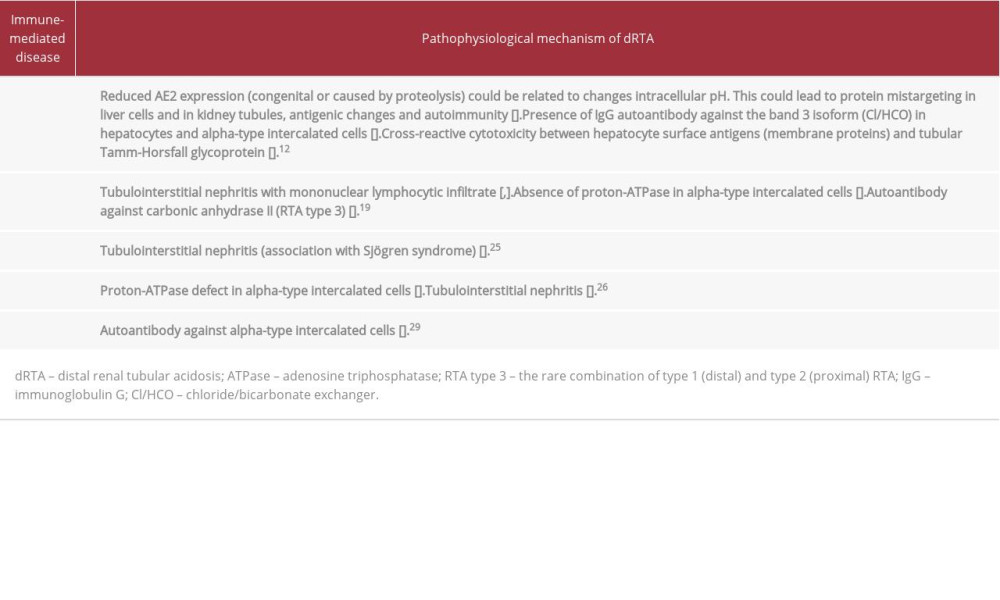
Autoimmune diseases can present with dRTA. Despite being a well-known association, the pathophysiological mechanisms are diverse and not yet fully understood (Table 1). All 3 of the patients described here had hyperchloremic metabolic acidosis (normal plasma anion gap), as well as mild hypokalemia, alkaline urine, and hypocitraturia, which are characteristics compatible with dRTA.
The patients did not have any degree of proteinuria (which could correspond to low-molecular-weight proteins) or phosphorus disturbance, nor any biochemical signs compatible with proximal tubulopathy. Furthermore, supraphysiological urinary pH would not be compatible with established proximal RTA.
The first patient was diagnosed with autoimmune hepatitis at 10 years of age and presented with nephrolithiasis. Hypocitraturia, which is a typical finding in patients with dRTA, is a risk factor for this complication [3,4,8]. Nephrolithiasis and nephrocalcinosis are common complications, seen in 5–30% of cases, underscoring the need for early diagnosis [9,10].
The association between autoimmune liver diseases (autoimmune hepatitis and primary biliary cirrhosis) and dRTA is well known and affects approximately 30% of patients with such diseases, although the mechanisms are diverse and have not been fully elucidated [11]. The chloride/bicarbonate exchanger (an isoform of band 3) is present in various organs and tissues such as the liver and renal tubular cells [12]. This exchanger participates in the maintenance of intracellular pH (through chlorine influx and bicarbonate efflux) [12]. Low intracellular pH itself causes a reduction in chloride/bicarbonate exchanger activity. It can also promote proteolysis and antigenic and autoimmune changes (Figure 2A) [12–14].
In chronic autoimmune liver disease, the suggested mechanism is the presence of immunoglobulin G autoantibodies against the chloride/bicarbonate anion exchanger 2 (AE2) present in hepatocytes, and its chloride/bicarbonate exchanger isoform (AE1), which is present in the basolateral membrane in alpha-type intercalated cells in the collecting tubule [12–14]. In addition, the mechanism of dRTA development might involve cross-reactive cytotoxicity between hepatocyte membrane surface lipoproteins (after hepatocellular damage) and Tamm-Horsfall glycoprotein-producing tubular cells (Figure 2B) [15].
Bile cast nephropathy is another type of tubular lesion that is possible in patients with cirrhosis. The bilirubin value of patient 1 was not compatible with this type of injury, which is usually associated with severe hyperbilirubinemia [16].
Patient 1 had liver disease and was using a low dose of prednisone (10 mg/day) and furosemide (40 mg/day). Corticosteroid use can interfere with plasma potassium levels. Mineralocorticoid stimulation promotes increased urinary excretion of potassium and H+ (protons), which can promote hypokalemia and urinary acidification with a predisposition to metabolic alkalosis [17]. The patient had mild hypokalemia, which may in part be explained by the use of low doses of corticosteroids and furosemide, but she had metabolic acidosis with a normal plasma anion gap and impairment in urinary acidification with a positive urinary anion gap, which further corroborates the diagnosis of dRTA.
Liver cirrhosis may be associated with respiratory alkalosis. The patient with autoimmune hepatitis presented arterial blood gases showing pH 7.30, PCO2 35.5, and HCO3 17. In the case of metabolic acidosis, the expected PCO2, applying the Winters’ equation 1.5× HCO3 +8±2, was 33.5±2 (31.5 to 35.5), and there was no respiratory disorder [18].
The main form of renal injury in Sjögren syndrome is tubulointerstitial nephritis [19,20], and the estimated prevalence of RTA in such patients is approximately 30% [21–24]. Although dRTA is the most common form, with an estimated prevalence of 25%, the pathophysiological mechanism is not fully understood [22]. One proposed mechanism is the influence of autoantibodies against collecting tubule cells, although the absence of proton-ATPase without autoantibody detection has been reported [21]. In addition, it has been suggested that type 3 RTA can occur because of the presence of antibodies against carbonic anhydrase II [22].
There have been few reports of dRTA in individuals with rheumatoid arthritis. A few studies, mostly case reports, have described dRTA in patients with rheumatoid arthritis and Sjögren syndrome, suggesting that the mechanism involves tubulointerstitial nephritis [25].
In patients with systemic lupus erythematosus (SLE), the presence of dRTA has also been previously reported [26,27]. This occurs mainly in SLE patients with other immunological diseases, such as thyroiditis and Sjögren syndrome [26]. A proton-ATPase defect in the luminal membrane of alpha-type intercalated cells has been demonstrated in renal tissue collected from SLE patients with dRTA [26]. Tubulointerstitial nephritis and the presence of autoantibodies against carbonic anhydrase II have been implicated in dRTA and type 3 RTA [28]. In addition, type 4 RTA (specifically hyporeninemic hypoaldosteronism) has also been observed in SLE patients [26]. Patients with autoimmune thyroiditis can also develop dRTA. One experimental study identified autoantibodies against intercalated cells as a possible explanation for the occurrence of the disorder [29].
There have been case reports describing the combination of fibrosing alveolitis and RTA [30]. Cases of alveolitis in patients with Sjögren syndrome and hypergammaglobulinemia have both been observed, which suggests an immunological mechanism in its pathogenesis [30].
Mutations in the ATP6V1B1 gene encoding the B1 subunit of vacuolar H-ATPase result in autosomal recessive dRTA associated with sensorineural hearing loss [31]. None of the 3 patients reported here reported hearing alterations. Genetic testing was also not performed in these cases.
Alkali results, especially with potassium citrate salts, are interesting in dRTA. Citrate can provide a longer alkali half-life as it is still metabolized to bicarbonate in the liver. Furthermore, potassium citrate is less associated with hypercalciuria, as is the case with sodium-based alkali [32].
Our patients used potassium citrate, which stabilized plasma pH and maintained bicarbonate (BIC) at 22–26 mEq/L. They used oral potassium chloride (24 to 48 mEq/day) to help maintain normal potassium.
Conclusions
We conclude that there are a number of autoimmune diseases that are associated with dRTA, although the pathophysiological mechanisms are diverse and are not fully understood. In clinical practice, is important to keep this possible association in mind and to monitor the electrolyte and metabolic balance in patients with autoimmune diseases. It is recommended that imaging examinations be performed periodically for the early detection of nephrolithiasis or nephrocalcinosis. Earlier treatment is associated with better prognosis.
Figures
References:
1.. Soriano JR, Renal tubular acidosis: the clinical entity: J Am Soc Nephrol, 2002; 13; 2160-70
2.. Yaxley J, Pirrone C, Review of the diagnostic evaluation of renal tubular acidosis: Ochsner J, 2016; 16(4); 525-30
3.. Trepiccione F, Prosperi F, de la Motte LR, New findings on the pathogenesis of distal renal tubular acidosis: Kidney Dis (Basel), 2017; 3(3); 98-105
4.. Norman ME, Feldman NI, Cohn RM, Urinary citrate excretion in the diagnosis of distal renal tubular acidosis: J Pediatr, 1978; 92; 394-400
5.. Alexander RT, Cordat E, Chambrey R, Acidosis and urinary calcium excretion: Insights from genetic disorders: J Am Soc Nephrol, 2016; 27(12); 3511-20
6.. Jarolim P, Shayakul C, Prabakaran D: J Biol Chem, 1998; 273(11); 6380-88
7.. Miura K, Sekine T, Takahashi K, Mutational analyses of the ATP6V1B1 and ATP6V0A4 genes in patients with primary distal renal tubular acidosis: Nephrol Dial Transplant, 2013; 28(8); 2123-30
8.. Watanabe T, Improving outcomes for patients with distal renal tubular acidosis: Recent advances and challenges ahead: Pediatric Health Med Ther, 2018; 9; 181-90
9.. Both T, Zietse R, Hoorn EJ, Everything you need to know about distal renal tubular acidosis in autoimmune disease: Rheumatol Int, 2014; 34(8); 1037-45
10.. Brenner RJ, Spring DB, Sebastian A, Incidence of radiographically evident bone disease, nephrocalcinosis, and nephrolithiasis in various types of renal tubular acidosis: N Engl J Med, 1982; 307; 217-21
11.. Toblli JE, Findor J, Sorda J, Latent distal tubular acidosis (dRTA) in primary biliary cirrhosis (PBC) and chronic autoimmune hepatitis (CAH): Acta Gastroenterol Latinoam, 1993; 23; 235-38
12.. Stakisaitis D, Didziapetriene J, Maciulaitis R, Tschaika M, Possible association between cell membrane band 3 impairment function and renal tubular acidosis (liver diseases, malignancies and adverse drug reactions): Medicina (Kaunas), 2004; 40(1); 9-15
13.. Benedetti A, Strazzabosco M, Ng OC, Boyer J: Proc Natl Acad Sci USA, 1994; 91; 792-96
14.. Al-Awquati Q, Plasticity in epithelial polarity of renal intercalated cells: Targeting of the H+-ATPase and band 3: Am J Physiol, 1996; 270C; 1571-80
15.. Cochrane AM, Tsantoulos DC, Moussouros A, Lymphocyte cytotoxicity for kidney cells in renal tubular acidosis of autoimmune liver disease: Br Med J, 1976; 2(6030); 276-78
16.. Chan S, Spraggon ES, Francis L, Wolley MJ, Bile cast nephropathy in a patient with obstructive jaundice: Kidney Int Rep, 2018; 4(2); 338-40
17.. Harrington JT, Hulter HN, Cohen JJ, Madias NE, Mineralocorticoid-stimulated renal acidification: The critical role of dietary sodium: Kidney Int, 1986; 30(1); 43-48
18.. Albert MS, Dell RB, Winters RW, Quantitative displacement of acid-base equilibrium in metabolic acidosis: Ann Intern Med, 1967; 66(2); 312-22
19.. Evans RD, Laing CM, Ciurtin C, Walsh SB, Tubulointerstitial nephritis in primary Sjögren syndrome: Clinical manifestations and response to treatment: BMC Musculoskelet Disord, 2016; 17; 2
20.. Kidder D, Rutherford E, Kipgen D, Kidney biopsy findings in primary Sjögren syndrome: Nephrol Dial Transplant, 2015; 30(8); 1363-69
21.. Cohen EP, Bastani B, Cohen MR, Absence of H(+)-ATPase in cortical collecting tubules of a patient with Sjögren’s syndrome and distal renal tubular acidosis: J Am Soc Nephrol, 1992; 3(2); 264-71
22.. Takemoto F, Hoshino J, Sawa N, Autoantibodies against carbonic anhydrase II are increased in renal tubular acidosis associated with Sjögren syndrome: Am J Med, 2005; 118(2); 181-84
23.. Ram R, Swarnalatha G, Dakshinamurty KV, Renal tubular acidosis in Sjögren’s syndrome: A case series: Am J Nephrol, 2014; 40(2); 123-30
24.. Jung SW, Park EJ, Kim JS, Renal tubular acidosis in patients with primary Sjögren’s syndrome: Electrolyte Blood Press, 2017; 15(1); 17-22
25.. Okada M, Suzuki K, Hidaka T, Rapid improvement of osteomalacia by treatment in a case with Sjögren’s syndrome, rheumatoid arthritis, and renal tubular acidosis type 1: Intern Med, 2001; 40(8); 829-32
26.. Kozeny GA, Barr W, Bansal VK, Occurrence of renal tubular dysfunction in lupus nephritis: Arch Intern Med, 1987; 147(5); 891-95
27.. Fang JT, Chan YC, Systemic lupus erythematosus presenting initially as hydrogen ATPase pump defects of distal renal tubular acidosis: Ren Fail, 2000; 22(4); 517-21
28.. Inagaki Y, Jinno-Yoshida Y, Hamasaki Y, Ueki H, A novel autoantibody reactive with carbonic anhydrase in sera from patients with systemic lupus erythematosus and Sjögren’s syndrome: J Dermatol Sci, 1991; 2(3); 147-54
29.. Gaarder PI, Heier HE, A human autoantibody to renal collecting duct cells associated with thyroid and gastric autoimmunity and possibly renal tubular acidosis: Clin Exp Immunol, 1983; 51(1); 29-37
30.. Mason AM, McIllmurray MB, Golding PL, Hughes DT, Fibrosing alveolitis associated with renal tubular acidosis: Br Med J, 1970; 4(5735); 596-99
31.. Subasioglu Uzak A, Cakar N, Comak E, ATP6V1B1 mutations in distal renal tubular acidosis and sensorineural hearing loss: Clinical and genetic spectrum of five families: Ren Fail, 2013; 35(9); 1281-84
32.. Trepiccione F, Walsh SB, Ariceta G, Distal renal tubular acidosis: ERKNet/ ESPN clinical practice points: Nephrol Dial Transplant, 2021; 36(9); 1585-96
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133