07 December 2021: Articles 
Leiomyoadenomatoid Tumors of the Uterus: A Case Report and Literature Review
Challenging differential diagnosis, Rare disease
Bayan Hafiz1ABEF*, Mariam H. Silimi2AE, Walaa Felmban3BEFDOI: 10.12659/AJCR.934012
Am J Case Rep 2021; 22:e934012
Abstract
BACKGROUND: An adenomatoid tumor is a benign neoplasm that originates in the mesothelial lining, commonly present in the male and female genital tracts. The lieomyoadenomatoid tumor (LMAT) is rare and considered an adenomatoid variant, characterized microscopically by prominent smooth muscle proliferation within an adenomatoid tumor. Areas of pseudo-glandular infiltration and tubular and slit-like spaces can mimic metastatic carcinoma or malignant tumors. Most cases of LMAT were incidentally discovered microscopically after an impression of leiomyoma. Eighteen cases have been reported in studies published in English.
CASE REPORT: We present a case of a 35-year-old woman who experienced recurrent vaginal bleeding and underwent a myomectomy. Microscopically, the mass showed smooth muscle proliferation in fascicles with areas of tubular, slit-like spaces, and gland-like areas, which showed reactivity for Wilms tumor-1 and calretinin by an immunohistochemistry study. The final pathology examination showed a 9-cm LMAT, which is the largest size ever reported to the best of our knowledge. There was no recurrence or other symptoms at the 2-year follow-up.
CONCLUSIONS: An LMAT should be considered in the differential diagnosis of leiomyomas that present areas of pseudo-infiltrative glands and slit-like spaces, which can lead to misdiagnosis as a malignant tumor.
Keywords: Adenomatoid Tumor, Leiomyoma, Neoplasms, Mesothelial, Female, Humans, Immunohistochemistry, Male, Uterine Neoplasms, Uterus
Background
An adenomatoid tumor (AT) is a tumor of mesothelial origin that commonly develops in the male and female genital tracts [1]. This type of tumor was initially discovered by Sakaguchi in 1916 and was called adenomyomata before Golden et al renamed the condition as “adenomatoid tumor” [2,3]. The microscopic features include the presence of hypertrophied smooth muscle bundled in areas exhibiting anastomosing tubular, gland-like, vascular-like, or cystic branch morphologies. However, when the smooth muscle is exceedingly prominent and gives the appearance of leiomyoma within the AT, the lesion is then referred to as a leiomyoadenomatoid tumor (LMAT) [4]. Clinically, LMAT is considered an incidental finding during hysterectomies or leiomyoma diagnosis.
It is important to recognize these tumors because the prominence of smooth muscle in this particular gland-like structure can mimic features of malignancy.
We report the case of a 35-year-old woman with an LMAT incidentally found in her uterus, which was mistaken for a different kind of tumor and was therefore removed as a uterine fibroid. The diagnosis was challenging due to the presence of cribriform gland-like morphology infiltrating the smooth muscle.
Case Report
A 35-year-old woman arrived at the Obstetrics and Gynecology clinic with concerns for lower abdominal pain. The pain started 3 months previously, was characterized by intermittent occur-rences, and was usually managed with regular analgesia. No constitutional symptoms were recorded. The patient’s menstrual history was regular; however, 7 months prior to her presentation, her menses became heavy and was associated with severe lower abdominal pain. During the physical examination, the patient appeared conscious, oriented, and alert. Her vital signs were within the standard ranges. The abdominal examination revealed a soft and lax abdomen with no masses or tenderness. Other systemic examinations were unremarkable. The initial investigation, which consisted of a complete blood count, blood electrolytes, and hepatic function tests, were all within the standard ranges. A radiological study was conducted, including an ultrasound, revealing multiple uterine fibroids. Based on the clinical and radiological findings, a myomectomy was performed, and the specimen was sent for histopathology examination.
On gross examination, multiple well-circumscribed white-tan pieces were noted, and the largest piece measured 9.0×3.0×2.5 cm. The others measured in aggregate 3.0×2.0×0.5 cm. The outer surfaces of all pieces were smooth and characterized by trabeculae, white-tan firm surfaces, and a whorled appearance. As shown in Figure 1, no necrosis or hemorrhage was seen.
The microscopic testing uncovered bundles of smooth muscle arranged in fascicles separated by well-vascularized connective tissue. The cells were elongated with eosinophilic cytoplasm and distinct cell membranes. A gland-like, tubular, and slit-like structure infiltrated the smooth muscle. Those structures were lined by a flattened cuboidal lining with small and uniform nuclei and contained a scanty and eosinophilic cytoplasm, as illustrated in Figure2A–2D. The mitosis was rarely less than 3 per 10 high-power fields. No necrosis was observed. Immunohistochemistry revealed diffuse strong staining to the calretinin and D2-40, as displayed in Figure 2E–2F; low-molecular-weight cytokeratin (CK7); wide-spectrum CKs; and a negative reaction to CK20, endothelial antigens (CD31), and Ki 67, which revealed a low proliferative index (<3%). The smooth muscle component expressed positive activity in reaction to smooth muscle actin. A follow-up appointment 3 months afterward showed the patient was doing well, with no signs of recurrence or malignant transformation.
Discussion
An AT is a benign tumor that arises from mesothelial cells [4]. ATs commonly form in organs covered by mesothelial linings, such as the genital tract, and therefore mainly occur in the testis and uterus [4]. Extra-genital sites, particularly in the organs closed to serosal membranes (eg, pleura, peritoneum, and pericardium), the adrenal gland, and other visceral organs can be involved [5]. An AT is usually located subserosally in the posterior wall near the uterine cornu and is < 4 cm in size. Previous studies have mentioned the association between leiomyomata and adenomyosis [5,6].
The microscopic description of the AT shows an intersecting hypertrophied smooth muscle with unremarkable nuclei within a slit-like cystic area as well as glands infiltrating between the muscles lined by round to cuboidal cells. Those cells express immunoreactivity for Wilms tumor-1, calretinin, CK AE1/ AE3, and CK7, as in our case.
Differential diagnosis is not uncommon in cases of AT owing to various histological patterns, as mentioned previously. These include leiomyoma, angioleimyoma, leiomyoma with malignancies, malignant mesothelioma, yolk sac tumors, and metastatic adenocarcinoma. AT can mimic leiomyoma or malignant tumors infiltrating smooth muscle when smooth muscle is prominent, thus concealing the AT component. This type of tumor, the LMAT, was described by Epstein in 1992 and is considered a variant of the AT with a prominent smooth muscle component [4]. In the present case, the first impression was a metastatic tumor infiltrating the smooth muscle, which made it challenging to reach the diagnosis of LMAT.
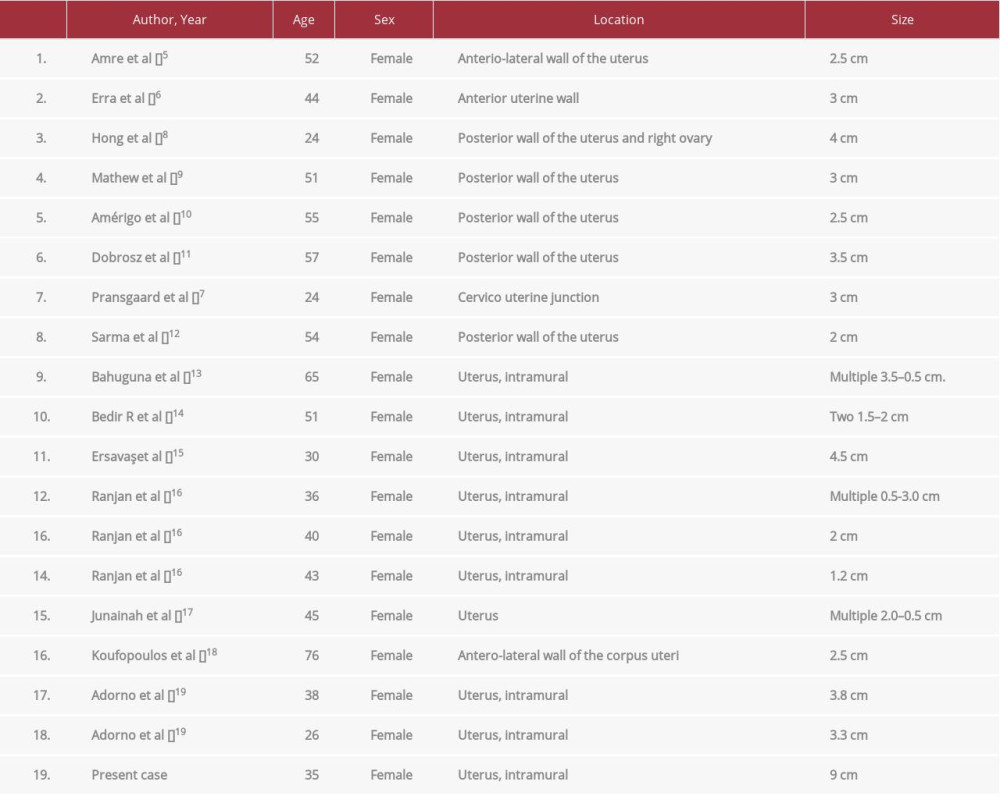
After a review of the available studies published in English on PubMed, Ovid, and Google Scholar, we found only 18 cases that were diagnosed as LMATs, as shown in Table 1 [5–19]. One was in the cervico-uterine junction [7], 1 was in the ovary [8], and the remaining 16 cases, in addition to ours, were discovered in the uterus. The patients’ ages ranged from 24 to 76 years, and the size of the tumors ranged from 1.5 to 4 cm. However, the size of the tumor in our case was 9 cm, which was the largest tumor so far, to the best of our knowledge. We found only 1 other case in Saudi Arabia, reported by Junainah et al [16], and our case is the second reported in the country.
Conclusions
We conducted a literature review on LMATs and reported a case study of a female patient with this condition. An LMAT was found incidentally in the patient’s uterus. We recommend that if the leiomyoma has prominent smooth muscles, LMAT should be considered in the differential diagnosis to avoid being mistaken for an invasive malignancy.
Figures
References:
1.. David LW, Wunsch PH, Hartmann A, Adenomatoid tumors of the female and male genital tract. A comparative clinicopathologic and immunohistochemical analysis of 47 cases emphasizing their site-specific morphologic diversity: Virchows Arch, 2011; 458; 593-602
2.. Phillip AI, Gary LK, Sebo TJ, Adenomatoid tumor of the adrenal gland A clinicopathologic study of five cases and review of the literature: Am J Surg Pathol, 2003; 27; 969-77
3.. Golden A, Ash JE, Adenomatoid tumors of the genital tract: Am J Pathol, 1945; 21; 63-79
4.. Epstein JI: Urologic disorders: Differential diagnosis in pathology, 1992; 173-74, New York, NY, Igasku-Shoin
5.. Amre R, Constantino J, Lu S, Charney D, Pathologic quiz case: A 52-year-old woman with a uterine mass. Leiomyo-adenomatoid tumor of the uterus: Arch Pathol Lab Med, 2005; 129; e77-78
6.. Erra S, Pastormerlo M, Gregori G, A case of leiomyoadenomatoid tumour of uterine serosa: Speculations about differential diagnosis: BMJ Case Rep, 2009; 1586; bcr02.2009.1586
7.. Pransgaard T, Lykke R, Hansen ES, Leiomyoadenomatoid tumor of the uterus: Report of a rare entity: J Gynecol Surg, 2013; 29; 219-21
8.. Hong R, Choi DY, Choi SJ, Lim SC, Multicentric infarcted leiomyoadenomatoid tumor: A case report: Int J Clin Exp Pathol, 2009; 2; 99-103
9.. Mathew M, Goel G, Leiomyoadenomatoid tumor of the uterus: Turk Patoloji Derg, 2010; 26; 168-69
10.. Amérigo J, Amérigo-Góngora M, Giménez-Pizarro A, Leiomyoadenomatoid tumor of the uterus: A distinct morphological entity?: Arch Gynecol Obstet, 2010; 282(4); 451-54
11.. Dobrosz Z, Palen P, Wlaszczuk P, Stojko R, An atypical leiomyoadenomatoid tumor of the uterus: A case report and literature review: Ginekol Pol, 2013; 84; 730-32
12.. Sarma NH, Srinivasulu M, Suchitra MJ, Leiomyoadenomatoid tumor of the uterus: Report of a rare entity and review of the literature: Indian J Pathol Microbiol, 2014; 57; 450-52
13.. Bahuguna G, Misra D, Malhotra V, Sinha A, Multicentric leiomyoadenomatoid tumor of the uterus: A rare and distinct morphological entity: J Obstet Gynaecol India, 2014; 64(Suppl. 1); 152-53
14.. Bedir R, Üstüner I, Mürtezaoglu AR, Leiomyoadenomatoid tumor of the uterus: Case report and literature review: Natl Lab Med, 2014; 3; 15-18
15.. Ersavaş S, Eliyatkin N, Sayhan S, Zihni I, Leiomyoadenomatoid tumor of the uterus in pregnancy: A case report: Journal of Tepecik Education and Research Hospital, 2015; 25; 120-24
16.. Ranjan R, Singh L, Nath D, Uterine adenomatoid tumors: a study of five cases including three cases of the rare leiomyoadenomatoid variant: J Obstet Gynaecol India, 2015; 65(4); 255-58
17.. Junainah EM, Elrashidy A, Elnashar H, Multifocal leiomyo-adenomatoid tumour of the uterus a distinct pathological entity: Allied J Clinicl Path, 2017; 1(1); 13-15
18.. Koufopoulos N, Karopoulou E, Arapaki A, EP727 Presentation of the 14th case of leiomyoadenomatoid tumor: English literature International J Gynecol Cancer, 2019; 29; A411
19.. Adorno FÁ, Coelho KM, Bublitz GS, Leiomyoadenomatoid tumor of uterus: Two case reports with literature review: Surg Exp Pathol, 2021; 4(1); s42047-020-00086-9
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133