13 February 2022: Articles 
Pulmonary Function and Chest Computed Tomography (CT) Scan Findings After Antifibrotic Treatment for COVID-19-Related Pulmonary Fibrosis
Unusual setting of medical care
Jan Michael Jesse Lomanta1ABCDEF*, Milraam L. Quinto12BCDEF, Sheen C. Urquiza3CD, Joel M. Santiaguel14ADEFDOI: 10.12659/AJCR.934830
Am J Case Rep 2022; 23:e934830
Abstract
BACKGROUND: Physicians worldwide have been reporting many cases of COVID-19-induced pulmonary fibrosis. We report the case of a 51-year-old Filipino asthmatic woman who developed post-COVID-19 pulmonary fibrosis subsequently treated with Nintedanib.
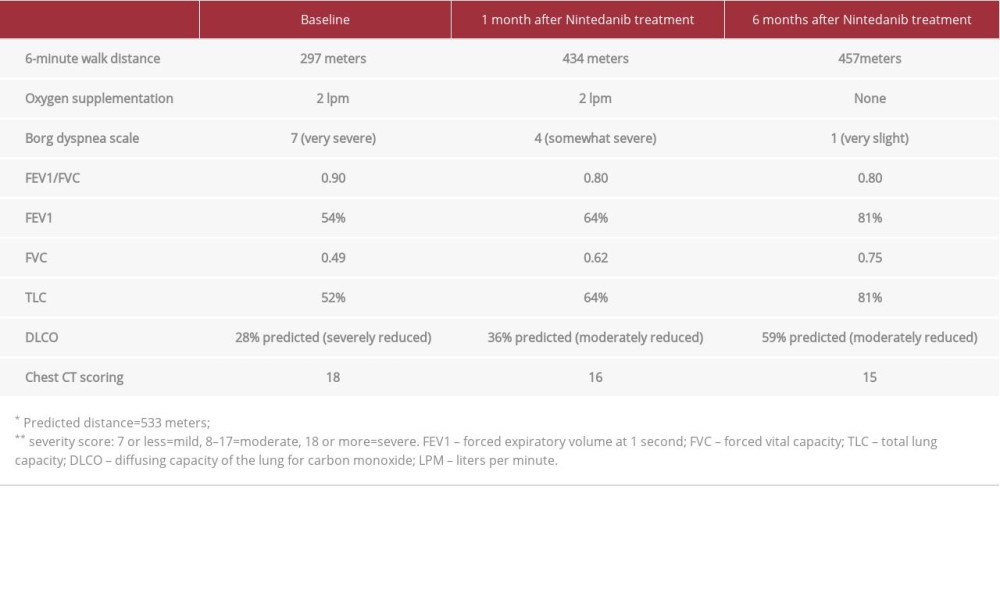
CASE REPORT: The patient presented with a 4-day history of flu-like symptoms in September 2020 and was eventually diagnosed with severe COVID-19 pneumonia. Despite receiving Dexamethasone, Tocilizumab, Remdesivir, and multiple antibiotics, there was increasing oxygen requirement, necessitating ICU admission and high-flow nasal cannula (HFNC). An additional course of hydrocortisone was given due to asthma exacerbation, gradually liberating her from the HFNC. A chest CT scan showed extensive parenchymal changes, for which she received methylprednisolone and physical rehabilitation with persistence of respiratory symptoms. After 40 days of hospitalization, she was sent home on oxygen support and Nintedanib. The patient initially had severe dyspnea (Borg Scale 7) with 6-minute walk distance (6MWD) of 295 meters. Pulmonary function showed moderately severe restrictive lung defect at 52% predicted total lung capacity (TLC) and severely reduced DLCO (28% predicted). Chest CT scoring indicated severe lung involvement. One month after Nintedanib treatment, her Borg Scale improved to 4. Her 6MWD, TLC, and DLCO increased to 434 meters, 64% predicted, and 36% predicted, respectively. A chest CT scan showed regressing fibrosis. After 6 months of treatment, her pulmonary function normalized. DLCO remained moderately reduced (59% predicted) but her 6MWD (457 meters) and CT scan results continued to improve.
CONCLUSIONS: Nintedanib, along with other interventions, may have potentially improved pulmonary function and CT scan findings in a COVID-19 survivor with pulmonary fibrosis 6 months after treatment.
Keywords: COVID-19, nintedanib, COVID-19, Female, Humans, SARS-CoV-2, Tomography, X-Ray Computed
Background
Declared as a pandemic by the World Health Organization (WHO) in March 2020, Coronavirus Disease 2019 (COVID-19) has become a global health crisis with over 220 million confirmed cases and 4.6 million deaths as of September 2021 [1]. The first case of COVID-19 in the Philippines was reported in January 2020, which led to a sustained community transmission in March 2020, now reaching a total of 2.2 million cases as of this paper’s writing [1,2]. Although the emergence of effective therapeutic regimens subsequently improved clinical outcomes and increased the number of survivors, a subset of patients remained symptomatic even in the post-infectious period [3]. In fact, a study by Logue et al (2021) demonstrated that as much as 30% of recovered patients had persistent symptoms, such as fatigue, shortness of breath, cough, and headache 9 months after COVID-19 diagnosis [4]. One interesting cause of the development of these lingering respiratory symptoms is post-acute respiratory distress syndrome (ARDS) pulmonary fibrosis [3].
Pulmonary fibrosis occurs in response to inflammation. ARDS, which results from a variety of infectious and non-infectious causes, may very well be the ultimate inflammatory condition of the lungs. Pulmonary function following ARDS is frequently reduced. Among survivors of ARDS in the pre-COVID era, up to 80% of patients had reduced DLCO and 20% had restriction on spirometry [5]. Moreover, the impact of ARDS is not limited to pulmonary function. CT scan abnormalities have been demonstrated among post-ARDS patients showing changes consistent with fibrosis, especially among those with worse severity score, prolonged mechanical ventilation, and higher oxygen requirement and lung pressures [6,7]. These changes may persist up to 5 years [8]. Among survivors of severe COVID-19, a prospective study by Han et al (2021) revealed that more than one-third of these patients have persistent fibrotic-like changes in CT scan, even at 6-month follow-up [9].
Among survivors of the severe acute respiratory syndrome (SARS) epidemic, significant impairment in DLCO as well as persistent radiographic changes consistent with fibrosis were also noted on follow-up imaging, more severe among those who were critically ill [10]. This raised the concern over the possible trajectory of patients recovering from COVID-19. Similar findings were demonstrated among survivors of ARDS secondary to Middle-East respiratory syndrome (MERS), another coronavirus and severe influenza H1N1 pneumonitis [11–13]. Treatment of these complications was usually limited to physical rehabilitation, but there is a case report of 3 patients with post-H1N1 pulmonary fibrosis who responded well to treatment with Pirfenidone, an antifibrotic, Azithromycin, and prednisolone [14].
Antifibrotic drugs such as Nintedanib and Pirfenidone have been used for the treatment of fibrotic lung diseases. Data from 2 replicate randomized controlled trials (INPULSIS 1 and 2) showed that Nintedanib slows the decline of forced vital capacity (FVC) and reduces the incidence of exacerbation among patients with idiopathic pulmonary fibrosis (IPF) [15]. As the pathway for COVID-19-related pulmonary fibrosis seems to be similar to that of interstitial lung diseases, Nintedanib, a tyrosine kinase inhibitor that inhibits fibrogenesis, might be beneficial [16]. However, there are limited data on its use for COVID-19-associated pulmonary fibrosis, and clinical trials are still underway.
Here, we present the case of Filipino woman who developed severe pulmonary fibrosis after COVID-19 treated with Nintedanib. This is the first report on the use of Nintedanib in a Filipino patient with post-COVID-19 pulmonary fibrosis.
Case Report
A 51-year-old Filipino woman with controlled bronchial asthma developed a 4-day history of nonproductive cough, fever, and exertional dyspnea in September 2020, and her RT-PCR test result for SARS-CoV-2 was positive. She was subsequently admitted at a local hospital, started on supplemental oxygen by nasal cannula at 5 liters/minute, and was managed as a case of severe COVID-19 pneumonia. Despite completing 10 days of Remdesivir, 10 days of Dexamethasone, 1 dose of Tocilizumab, and multiple antibiotics, she developed increasing oxygen requirement as high as 10 liters/minute, oxygen partial pressure (PaO2)/fraction of inspired oxygen (Fio2) ratio consistently less than 150, and persistent dyspnea.
During her second week of illness, she was transferred to our institution due to the progression of symptoms. She was treated as a case of acute respiratory failure from severe COVID-19 pneumonia and asthma exacerbation. She was admitted in the Intensive Care Unit (ICU) and was placed on high-flow nasal cannula (HFNC) with 60% Fio2 at 35 liters/minute airflow. The patient received hydrocortisone for 7 days and inhaled budesonide-formoterol. Following these interventions, she was weaned from HFNC to low-flow nasal cannula after 1 week and was eventually transferred out of the ICU. However, she had persistent bothersome cough and exertional dyspnea, which was associated with progression of infiltrates on serial chest X-ray. A chest computed tomography (CT) scan (Figure 1) demonstrated extensive intralobular septal thickening and consolidation. After ensuring that all septic work-ups were negative, she was given methylprednisolone pulse therapy for 5 days in light of the worsening radiologic findings and difficulty weaning from oxygen support, which at that time was attributed to a possible organizing pneumonia. However, there was no significant improvement. Pulmonary rehabilitation and incentive spirometry were initiated upon transfer to the wards.
Deep breathing exercises with scapular retraction and relaxation techniques were taught to the patient.
After 2 weeks of continued rehabilitation, the patient’s symptoms gradually improved, but she still had residual exertional dyspnea. She did not develop other hospital-related complications. Finally, 40 days after symptom onset, she was sent home on oxygen support at 2 liters/minute and Nintedanib 150 mg/tab twice per day. Prior to discharge, 6-minute walk test (6MWT), Borg Dyspnea Scale, and spirometry with body plethysmography and diffusing capacity for carbon monoxide (DLCO) were conducted.
Initially, the patient had severe dyspnea (Borg 7) with 6MWD of 295 meters while on 2 liters/minute oxygen support. Spirometry with body plethysmography showed decreased total lung capacity (TLC) at 52% predicted, indicative of moderately severe lung restriction. DLCO was severely reduced at 28% predicted. CT scoring, which was computed according to the protocol of Pan et al (2020) and Li et al (2020) showed severe lung involvement (CT score: 18) [17,18]. Table 1 summarizes the pulmonary function results and CT scoring.
After 4 weeks of Nintedanib treatment, Borg Scale decreased to 4. Her 6MWD increased to 434 meters but she still needed oxygen support at 2 liters/min. TLC and DLCO also improved to 64% predicted and 36% predicted, respectively. Repeat chest CT scan demonstrated regressing fibrosis (Figure 2), consistent with improved chest CT scoring.
Six months after treatment, her pulmonary function had returned to normal. DLCO remained moderately reduced (59% predicted) but her 6MWD (457 meters) and CT score (15) continued to improve. She was completely weaned off from oxygen support during this time. Throughout the duration of treatment, the patient continued her physical rehabilitation.
While taking Nintedanib, the patient reported having tolerable dysphagia, frontal headache, and upper-extremity numbness, which eventually resolved a few weeks after Nintedanib treatment. These adverse effects did not lead to discontinuation of treatment. No other adverse effects were reported.
Discussion
As the efforts of researchers paid off and began to improve the clinical outcomes of COVID-19 patients, the world is now seeing the consequences of post-COVID-19 infections. The new challenge is managing COVID-19 sequelae.
The patient described above showed similar characteristics to a cohort of COVID-19 survivors who developed persistent symptoms despite recovery. This subset of patients, who were found to demonstrate persistent symptomatology beyond 4 weeks from symptom onset, were eventually given the diagnosis of long COVID or post-acute sequelae of COVID-19 (PASC) [3,19]. These patients may present with fatigue, post-exertional malaise, brain fog, dyspnea, insomnia, chest tightness, cough, and anxiety, as well as radiologic abnormalities of patchy ground-glass opacities with or without pulmonary fibrosis [3,20,21]. PASC patients may also exhibit abnormal pulmonary function, including impaired DLCO, decreased TLC and 6MWD, even after recovery from acute illness [22,23]. Although knowledge of the duration of functional and radiologic abnormalities during long COVID is limited to short-term follow-up studies, a literature review by Proal and VanElzakker (2021) indicated that these abnormalities may extend at least 9 months from symptom onset [3].
The mechanisms of lung injury in COVID-19 are not completely understood. However, a study by Liu and colleagues (2020) implicated direct viral cytopathic effects and immune-mediated damage as possible mechanisms [24]. This unregulated immune response is thought to contribute to the occurrence of acute respiratory distress syndrome (ARDS) and promote pulmonary fibrosis, a condition characterized by haphazard reconstruction of the injured alveolar epithelium, excessive accumulation of collagen, and persistence of fibroblasts [25]. The evolution of ARDS is marked by 3 overlapping histologic stages: exudative, proliferative, and fibrotic phases [26]. The proliferative phase, which is signified by epithelial cell regeneration and fibroblast proliferation, heralds the start of lung repair [26]. In some patients, however, the injury is so extensive that lung remodeling progresses to irreversible deposition of thick and collagenous connective tissue, which characterize the fibrotic phase. In a review by Rai et al (2021), risk factors for the development of post-COVID-19 pulmonary fibrosis include advanced age (over 50 years old), increased disease severity, prolonged ICU stay, longer mechanical ventilation, and current smoking status [27].
The natural course of pulmonary fibrosis following COVID-19 is yet to be established. A retrospective study by Zou et al (2021) on the evolution of post-COVID-19 pulmonary fibrosis showed that linear opacities, interlobular septal thickening, and reticulations on CT scan improved 30 days, 60 days, and 90 days after discharge compared to baseline [28]. They also reported that interlobular septal thickening decreased to 18.1% at 6 months compared to 36.4% from baseline [28]. Although the authors reported gradual improvement of fibrosis over time, even without definite intervention, this was confounded by a small population size and high dropout rates [28]. Some SARS survivors have additional improvement in radiographic and pulmonary function even 2 years after hospital discharge [29]. Whether the interventions given to our patient accelerated the improvement of very severe lung disease cannot be answered by this case report.
Organizing pneumonia, which presents with CT findings of peripheral consolidation, reticulations, and ground-glass infiltrates, has also been reported to complicate the course of COVID-19 patients in 12.5% of cases [30–32]. The diagnosis of organizing pneumonia requires tissue biopsy and is characterized by patchy alveolar and bronchial filling by loose connective tissue on histologic evaluation [33]. Viral infection is a common etiology of organizing pneumonia and has been documented among patients with H1N1 and MERS [34,35]. Glucocorticoids are the mainstay of treatment for organizing pneumonia and result in rapid clinical and radiologic improvement [36,37]. Cottin and Cordiet (2012) reported that clinical improvement is usually dramatic even during the first days of steroid therapy, which was not seen in our patient [38]. Our patient needed supplemental oxygen and was dyspneic despite the high-dose methylprednisolone in addition to the previous steroid doses.
At present, the definitive treatment for post-COVID-19 pulmonary fibrosis has not been established [27]. This lack of definitive management is highlighted by the number of ongoing clinical trials evaluating various drugs, including steroids, for the treatment of COVID-19-related fibrosis [39]. Five of these clinical trials are currently evaluating the antifibrotic drugs Pirfenidone and Nintedanib. Pirfenidone and Nintedanib are given for chronic, progressive fibrotic lung disease, mostly in IPF, but are also used in non-IPF, unclassifiable fibrotic lung diseases. Data from large randomized trials on the efficacy and safety of Nintedanib for IPF (the INPULSIS studies) showed that Nintedanib reduces the decline in FVC among IPF patients, consistent with slowing disease progression [15]. Although in the INPULSIS-1 and 2 trials Nintedanib was given for 52 weeks, a significant difference in the mean FVC between the treatment and control group was noted as early as 4–6 weeks from the initiation of intervention [15]. Nintedanib is a tyrosine kinase inhibitor shown to interfere with pathways leading to fibrosis, such as fibroblast proliferation, migration and differentiation, and secretion extra-cellular matrix in in vitro studies, and has shown anti-inflammatory and antifibrotic activity in animal models [40].
Locally, the only available antifibrotic medication is Nintedanib. It was given in this case at 150 mg twice daily. On follow-up there was significant improvement in the patient’s exercise tolerance and regression of pulmonary fibrosis on repeat chest CT scans 1 month and 6 months after treatment. This positive response to Nintedanib was similar to the case of a 78-year-old Japanese woman with post-COVID-19 fibrosis and poor exercise tolerance who was given Nintedanib at a similar dose [41]. After 3 months of Nintedanib treatment, there was improvement in the patient’s functional capacity as demonstrated by decreasing oxygen requirement during walking. A recent study published by Umemura and colleagues (2021) supported the potential benefit of Nintedanib in reducing lung injury among mechanically ventilated COVID-19 patients given early in the disease course. This interventional study, which enrolled 30 intubated COVID-19 patients under the Nintedanib arm (given at the time of intubation) and 30 patients in the control group, showed that the duration of mechanical ventilation was shorter in the treatment group versus the control (
This patient also received a course of steroids, initially as a standard-of-care dose of Dexamethasone 6 mg once daily, with extension to about 3 weeks. Corticosteroids mitigate inflammatory response in the lungs and are used for the treatment of exacerbations of IPF. A case report of 3 patients with post-COVID-19 pulmonary fibrosis also showed improvement of symptoms and chest X-ray findings after varying durations of steroid treatment [43]. The use of corticosteroids in this case may have contributed to the patient’s improvement, as she was liberated from HFNC during the early part of the disease at the time steroids were being given. However, we are not certain how much of the improvement can be attributed to steroids after the patient’s discharge. Clinical trials have yet to confirm its effects on COVID-19-related fibrosis. The results of a randomized controlled trial on short-term low-dose steroids for the management of post-COVID-19 fibrosis are still pending [44].
Physical rehabilitation, which the patient also received, has long been known to benefit pulmonary patients. Initiation of early mobilization immediately after admission for acute respiratory failure has been shown to accelerate recovery and reduce functional decline [45]. This contributed to the improvement of the symptomatology but cannot explain the improvement of chest CT scan results.
Diarrhea is the most common adverse effect of Nintedanib cited in the literature, but our patient reported headache, dysphagia, and upper-extremity numbness, which eventually resolved after treatment discontinuation.
Conclusions
Pulmonary fibrosis is an emerging sequela of COVID-19. This report demonstrated a case of pulmonary fibrosis after severe COVID-19 pneumonia, with improvement from baseline in terms of symptomatology, pulmonary function, and chest CT scan findings after a short course of Nintedanib, steroids, and physical rehabilitation. The individual contribution of each intervention, however, needs further evaluation using well-designed clinical trials.
Figures
References:
1.. [Internet]. WHO Coronavirus (COVID-19) Dashboard; c2021 [cited 2021 July 4] Available from: https://covid19.who.int
2.. Edrada EM, Lopez EB, Villarama JB, First COVID-19 infections in the Philippines: A case report: Trop Med Health, 2020; 48(1); 21
3.. Proal A, VanElzakker M, Long COVID or post-acute sequelae of COVID-19 (PASC): An overview of biological factors that may contribute to persistent symptoms: Front Microbiol, 2021; 12; 698169
4.. Logue J, Franko N, McCulloch D, Sequelae in adults at 6 months after COVID-19 infection: JAMA Netw Open, 2021; 4(2); e210830
5.. Orme J, Romney J, Hopkins R, Pulmonary function and health-related quality of life in survivors of acute respiratory distress syndrome: Am J Respir Crit Care Med, 2003; 167(5); 690-94
6.. Desai SR, Wells AU, Rubens MB, Hansell DM, Acute respiratory distress syndrome: CT abnormalities at long-term follow-up: Radiology, 1999; 210(1); 29-35
7.. Nöbauer-Huhmann IM, Eibenberger K, Schaefer-Prokop C, Changes in lung parenchyma after acute respiratory distress syndrome (ARDS): Assessment with high-resolution computed tomography: Eur Radiol, 2001; 11(12); 2436-43
8.. Wilcox ME, Patsios D, Murphy G, Radiologic outcomes at 5 years after severe ARDS: Chest, 2013; 143(4); 920-26
9.. Han X, Fan Y, Alwalid O, Six-month follow-up chest CT findings after severe COVID-19 pneumonia: Radiology, 2021; 299(1); E177-86
10.. Hui DS, Wong KT, Ko FW, The 1-year impact of severe acute respiratory syndrome on pulmonary function, exercise capacity, and quality of life in a cohort of survivors: Chest, 2005; 128(4); 2247-61
11.. Park WB, Jun KI, Kim G, Correlation between pneumonia severity and pulmonary complications in Middle East respiratory syndrome: J Korean Med Sci, 2018; 33(24); e169
12.. Hsieh MJ, Lee WC, Cho HY, Recovery of pulmonary functions, exercise capacity, and quality of life after pulmonary rehabilitation in survivors of ARDS due to severe influenza A (H1N1) pneumonitis: Influenza Other Respir Viruses, 2018; 12(5); 643-48
13.. Das KM, Lee EY, Singh R, Follow-up chest radiographic findings in patients with MERS-CoV after recovery: Indian J Radiol Imaging, 2017; 27(3); 342-49
14.. Saha A, Vaidya PJ, Chavhan VB, Combined pirfenidone, azithromycin and prednisolone in post-H1N1 ARDS pulmonary fibrosis: Sarcoidosis Vasc Diffuse Lung Dis, 2018; 35(1); 85-90
15.. Richeldi L, du Bois R, Raghu G, Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis: N Engl J Med, 2014; 370; 2071-82
16.. George PM, Wells AU, Jenkins RG, Pulmonary fibrosis and COVID-19: The potential role for antifibrotic therapy: Lancet Respir Med, 2020; 8(8); 807-15
17.. Pan F, Ye T, Sun P, Time course of lung changes at chest CT during recovery from coronavirus disease 2019 (COVID-19): Radiology, 2020; 295(3); 715-21
18.. Li K, Wu J, Wu F, The clinical and chest CT features associated with severe and critical COVID-19 pneumonia: Invest Radiol, 2020; 55(6); 327-31
19.. Nalbandian A, Sehgal K, Gupta A, Post-acute COVID-19 syndrome: Nat Med, 2021; 27(4); 601-15
20.. Carfì A, Bernabei R, Landi F, Persistent symptoms in patients after acute COVID-19: JAMA, 2020; 324(6); 603-5
21.. Huang C, Huang L, Wang Y, 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study: Lancet, 2021; 397; 220-32
22.. Huang Y, Tan C, Wu J, Impact of coronavirus disease 2019 on pulmonary function in early convalescence phase: Respiratory Research, 2020; 21(1); 163
23.. Zhao YM, Shang YM, Song WB, Follow-up study of the pulmonary function and related physiological characteristics of COVID-19 survivors three months after recovery: EClinicalMedicine, 2020; 25; 100463
24.. Liu J, Zheng X, Tong Q, Overlapping and discrete aspects of the pathology and pathogenesis of the emerging human pathogenic coronaviruses SARS-CoV, MERS-CoV, and 2019-nCoV: J Med Virol, 2020; 92(5); 491-94
25.. Sime PJ, O’Reilly KM, Fibrosis of the lung and other tissues: New concepts in pathogenesis and treatment: Clin Immunol, 2001; 99(3); 308-19
26.. Tomashefski JF, Pulmonary pathology of acute respiratory distress syndrome: Clin Chest Med, 2000; 21(3); 435-66
27.. Rai DK, Sharma P, Kumar R, Post-covid 19 pulmonary fibrosis. Is it a real threat?: Indian J Tuberc, 2021; 68(3); 330-33
28.. Zou JN, Sun L, Wang BR, The characteristics and evolution of pulmonary fibrosis in COVID-19 patients as assessed by AI-assisted chest HRCT: PLoS One, 2021; 16(3); e0248957
29.. Zhang P, Li J, Liu H, Long-term bone and lung consequences associated with hospital-acquired severe acute respiratory syndrome: A 15-year follow-up from a prospective cohort study: Bone Res, 2020; 14; 8
30.. Vadász I, Husain-Syed F, Dorfmüller P, Severe organising pneumonia following COVID-19: Thorax, 2021; 76(2); 201-4
31.. Ford DO, Holladay R, Sartawi T, Post-covid organizing pneumonia: Am J Respir Crit Care Med, 2021; 203(9) ID: covidwho-1277200
32.. de Oliveira Filho CM, Vieceli T, de Fraga Bassotto C, Organizing pneumonia: A late phase complication of COVID-19 responding dramatically to corticosteroids: Braz J Infect Dis, 2021; 25(1); 101541
33.. Drakopanagiotakis F, Polychronopoulos V, Judson MA, Organizing pneumonia: Am J Med Sci, 2008; 335(1); 34-39
34.. Ajlan AM, Ahyad RA, Jamjoom LG, Middle East respiratory syndrome coronavirus (MERS-CoV) infection: Chest CT findings: Am J Roentgenol, 2014; 203(4); 782-87
35.. Zanetti G, Hochhegger B, Marchiori E, Organizing pneumonia as a pulmonary sequela of swine flu: Lung India, 2013; 30(2); 171
36.. Torrego A, Pajares V, Mola A, Influenza A (H1N1) organising pneumonia: BMJ Case Rep, 2010; 2010; bcr12.2009.2531
37.. Kim I, Lee JE, Kim KH, Successful treatment of suspected organizing pneumonia in a patient with Middle East respiratory syndrome coronavirus infection: A case report: J Thorac Dis, 2016; 8(10); E1190-94
38.. Cottin V, Cordier JF, Cryptogenic organizing pneumonia: Semin Respir Crit Care Med, 2012; 33(5); 462-75
39.. Bazdyrev E, Rusina P, Panova M, Lung fibrosis after COVID-19: Treatment prospects: Pharmaceuticals (Basel), 2021; 14(8); 807
40.. Wollin L, Wex E, Pautsch A, Mode of action of nintedanib in the treatment of idiopathic pulmonary fibrosis: Eur Respir J, 2015; 45(5); 1434-45
41.. Ogata H, Nakagawa T, Sakoda S, Nintedanib treatment for pulmonary fibrosis after coronavirus disease 2019: Respirol Case Rep, 2021; 9(5); e00744
42.. Umemura Y, Mitsuyama Y, Minami K, Efficacy and safety of nintedanib for pulmonary fibrosis in severe pneumonia induced by COVID-19: An interventional study: Int J Infect Dis, 2021; 108; 454-60
43.. Lam E, Sayed N, Anjum F, Corticosteroid therapy in post-COVID-19 pulmonary fibrosis: Am J Respir Crit Care Med, 2021; 203(9)
44.. : Short-term low-dose corticosteroids for management of post COVID-19 pulmonary fibrosis; Identifier NCT04551781, 2020, Qena, Egypt, South Valley University
45.. Hanekom S, Gosselink R, Dean E, The development of a clinical management algorithm for early physical activity and mobilization of critically ill patients: Synthesis of evidence and expert opinion and its translation into practice: Clin Rehabil, 2011; 25(9); 771-87
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133