11 September 2022: Articles 
A Rare Case of Negative Serum Calcitonin in Metastatic Medullary Thyroid Carcinoma: Diagnosis, Treatment, and Follow-Up Strategy
Challenging differential diagnosis, Diagnostic / therapeutic accidents, Rare disease, Educational Purpose (only if useful for a systematic review or synthesis)
Lorena Licata1AE, Cristina A. Di Fidio1BCE, Marco Vacante1F, Francesco Basile1D, Antonio Biondi1F, Pierina Richiusa2AB, Alessandra Gurrera3D, Roberto Ciuni1ADE*DOI: 10.12659/AJCR.935207
Am J Case Rep 2022; 23:e935207
Abstract
BACKGROUND: Medullary thyroid carcinoma is a rare tumor and represents less than 5% of all thyroid cancers. Tumor size and the presence of possible metastases are strictly related to serum calcitonin level, which also suggests prognosis. A serum calcitonin level above the normal range is highly suspicious for medullary cancer. This tumor has no characteristic patterns on ultrasound and fine-needle aspiration, so serum calcitonin level is the only significant element for diagnosis. The literature contains 47 cases of serum calcitonin-negative medullary thyroid cancer with heterogeneous behavior, ranging from non-aggressive to aggressive form.
CASE REPORT: We report a rare case of a young man with negative serum calcitonin but with lateral cervical node metastases from medullary thyroid carcinoma. At the beginning, diagnosis was not achieved due to normal range of serum calcitonin and a negative thyroid ultrasonography for large nodules. Nevertheless, in another medical center he underwent a neck biopsy of the lateral cervical mass. The histologic findings showed metastases of medullary thyroid cancer. Therefore, a total thyroidectomy plus cervical lymphadenectomy was performed. During the follow-up, due to unusefulness of serum markers, and as CEA levels were negative pre- and post-operatively, an ¹⁸F-DOPA PET scan was performed. In 3 years of follow-up, there is still no evidence of disease relapse.
CONCLUSIONS: Our case shows how a normal serum level of calcitonin cannot exclude medullary thyroid cancer. Early diagnosis is crucial because it influences appropriate surgical therapy and patient outcome. The current lack of an optimal strategy for the diagnosis and postoperative follow-up for patients with negative serum calcitonin tumor presents a diagnostic challenge for physicians.
Keywords: Calcitonin, procalcitonin, Thyroid Cancer, Medullary, RET Protein, Human, Carcinoma, Neuroendocrine, Follow-Up Studies, Humans, Male, Thyroid Neoplasms
Background
Medullary thyroid carcinoma (MTC) represents 3–5% of all differentiated thyroid tumors [1] and 14% of all deadly thyroid cancers [2,3]. Diagnosis is based on serum tumor markers (calcitonin, CEA, chromogranin A, Ca 19-9 and pro-calcitonin), on ultrasound imaging of the neck, and on cytology.
Regarding medullary thyroid carcinoma markers, serum calcitonin (s-CT) has the highest sensitivity (100%) [1], allowing the correct diagnosis. Other MTC markers helpful in evaluating prognoses and follow-up are carcino-embryonic antigen (CEA), chromogranin A (CgA), Ca 19-9, and pro-calcitonin (PCT) [4]
MTC has no specific patterns at ultrasonography [3,4]: many samples are indeterminate with variable appearances on fine-needle aspiration cytology [5,6]. Thus, ultrasound and cytology support MTC diagnosis in patients with positive s-CT, but they are not sufficient by themselves for diagnosis. Another supportive diagnostic tool is immunohistochemical (IHC) analysis of FNA specimens, which might reduce indeterminate diagnoses, but it is not routinely available in most institutions.
s-CT level is directly proportional to tumor volume and potential metastases. Nevertheless, MTC patients with normal s-CT level, called CT-negative MTC, are diagnosed at an advanced stage [7,8]. In s-CT-negative MTC patients, ultrasonography and FNA may not help in establishing a diagnosis. The biological mechanism of negative serum is still unknown; CT- negative status may depend on the undifferentiation of the tumor cells, which is related to poorer prognosis [3]. There are many hypotheses: one considers that negative serum can be related to alteration of process of storage/secretion, while others suggest that it can be linked to protein-misfolding [5,9,10]. The literature contains 47 cases of CT-negative MTC. We report a case of a patient with an advanced MTC, s- CT-negative with RET mutation (positive in exon 14 with a heterozygous mutation c.2410 G>A, p.Val804Met). In this paper, we discuss the hypotheses of this biological presentation and the difficulties related to establishing the diagnosis of medullary thyroid carcinoma in s-CT-negative patients, which is essential to determine the best therapeutic strategy.
Case Report
A 39-year-old man with a slightly painful 2.5-cm left laterocervical mass at level III presented in April 2017, at which time the maxillofacial surgeon service report showed normal thyroid function, physiological range of antibodies against thyroid peroxidase (Ab TPO), and antibodies against thyroglobulin (Ab Tg); CT: 4 pg/L, CEA: 3.70 ng/mL (range 0–5 ng/mL). Thyroid ultrasonography demonstrated a hypoechoic nodule measuring 0.7×0.38 cm in the left middle lobe. Ultrasonography, computed tomography (CT), and MRI of the neck and fine-needle aspiration (FNA) of the latero- cervical mass finding were compatible with diagnosis of schwannoma, and all findings were incompatible with a diagnosis of metastatic MTC. The patient also had bilateral congenital neurosensory deafness.
Lateral cervical mass biopsy was performed as indicated. Histologic findings showed metastases of MTC; the immunohistochemistry (IHC) showed CT+ (calcitonin +), CK7+ (cytokeratin 7+), CgA+, synaptophysin (SPH) +, CEA+, Vimentin+, thyroglobulin-, and this IHC profile was compatible with MTC metastases.
Therefore, the patient was referred to the Endocrinology Unit of the Paolo Giaccone Hospital in Palermo, where he was screened for multiple neuroendocrine malignancies using neuroendocrine marker dosage (NSE: measured by a Roche Cobas e 602 automatic electrochemical luminescence immune-analyzer and its matched reagents [Roche Diagnostics GmbH]). The NSE was slightly above the normal range, but not significantly. Plasma metanephrine and fractionated 24-h urinary metanephrines were performed to exclude pheochromocytoma. Parathyroid hormone was normal (PTH: 44.6 pg/ml). The family history was negative for adrenal and parathyroid tumors. Finally, a genetic study for RET mutation was performed and was positive for heterozygous mutation in exon 14 (c.2410 G>A, p.Val804Met), confirmed on 2 independent samples.
The patient has been referred to us at Vittorio Emanuele Hospital of Catania. Preoperative thyroid ultrasonography revealed a single hypoechoic nodule, 0.4 cm in diameter in the right lobe and a single nodule in the left lobe, measuring 0.7×0.38 cm in diameter. After collecting all data and excluding MEN syndrome (multiple endocrine neoplasia), a total thyroidectomy with bilateral central neck and lateral left compartments dissection and lymphadenectomy were recommended. On the morning of surgery, blood samples were drawn 3 times, ensuring that the patient was drug-free so as not to alter the CT serum level. The samples were carried to the laboratory within 30 min, and they were centrifuged at 3000 r/min for 10 min, then serum was separated and analyzed. For analysis of serum calcitonin, our laboratory used a Siemens IMMULITE® 2000 automatic chemiluminescence immunoassay analyzer (reference range <8 pg=L) using a standard assay kit for in-vitro diagnostic (Siemens Healthcare Diagnostics Products Limited). Carbohydrate-associated antigen 19-9 (CA19-9), and CEA were analyzed using a Siemens Centaur® XP automatic chemiluminescence immunoassay analyzer (Siemens Healthcare Diagnostics, Inc). It was also dosed Pro-CT and was in normal range.
Surgical specimen histological findings (Figures 1, 2) showed bilateral medullary thyroid cancer with an amyloid stroma; the immunohistochemical analysis was positive for CT and CEA. Both tumors measured 3 mm and all remnants of thyroid tissue revealed C cell hyperplasia and venous vascular invasion but no extra-thyroidal extension. Two lymph nodes of the central compartment were positive for metastases, but no other metastases were found at the left lateral cervical level (only 1 was excised during the previous biopsy), so the final staging was pT1a(m) N1b, stage IVa.
A month and a half after surgery, blood tests showed Tg<1 ng/ml, CT 1.8 pg/mL, and CEA 1.14 ng/mL. Three months after surgery, blood tests showed chromogranin A 96.2 ng/ml (0–100) and NSE 9.4 mcg/L (0–18.3). The patient’s mother and son tested positive for RET mutation with a heterozygous mutation c.2410 G>A, p.Val804Met. During the follow-up, serum CT level was normal (1.7 pg/mL), as were the other markers. Ultrasound of the neck and of the liver were negative for metastases, as was the RX-chest. The patient underwent 18F-DOPA PET, as he was CT-negative, to be sure he was metastases-free. Thus, to date (February 2022) there is no evidence of disease relapse.
Discussion
The medullary thyroid tumor markers used for diagnosis and follow-up are Ct, Pro-CT, chromogranin A, CEA, and Ca19-9. Normally, a high level of serum CT is strongly suggestive for MTC. Serum concentration of CT is related to the mass of the tumor, to parafollicular C cell hyperplasia, and to presence of loco-regional cervical lymph nodes or distant-organ metastases [3]. Calcitonin is the most accurate marker for the diagnoses of MTC, but it is subjected to degradation if the test is not performed properly. Calcitonin is a 32-amino acid monomeric peptide resulting from cleavage of the post-translational processing of Pro-CT; it has a half-life of 15–40 min in physiologic condition and 3–30 h if in high concentrations [11]. The serum range reflects the pulsating secretion of the CT, which can be influenced by food intake and by temperature. At low temperature, CT decays by 23% after 12 h, by 35% after 24 h, and by 65% after 7 days, causing false-negative test results [11,12]. Pro-CT has been reported as a promising alternative marker for diagnosis and follow-up in uncertain cases of MTC without a high CT concentration, whereas chromogranin A is elevated in more than 50% of MTC cases, mainly at advanced disease stage, so it cannot be considered a useful marker for MTC diagnosis [1]. CgA is stored in neuroendocrine cells into aggregates; its serum level is directly proportional to tumor mass size and to cellular secretory activity [1]. Intracellular concentration CgA and its low serum level validate the hypothesis of impaired secretion function. CEA is not a specific marker for diagnosis of MTC, with high levels associated with other malignancies, but it can be useful in MTC patient follow-up [1]. Patients with aggressive MTC may have a disproportionately high CEA/CT ratio and a rapid CEA doubling time [13], so it might be irrelevant for the diagnosis, but it can be significant in follow-up. Carbohydrate antigen 19.9 (Ca 19.9) is a serum marker for gastrointestinal tumor, present in approximately 6% of MTC patients [5,14,15]. Further, the association between MTC and Ca 19.9 serum level has been demonstrated [4]. Based on these last observations, Ca19.9 level identifies a subgroup of patients with aggressive disease and with high mortality risk. All these tumor markers can be useful for diagnosis and follow-up of MTC, but against this background, dedifferentiation of the tumoral cells determines a reduction of CT secretion, known as CT-negative MTC. Several cases of non-secretory CT MTC, also called CT- negative MTC, are reported in the literature; since normal serum tumor markers are useless, their diagnosis is given only by surgical specimen IHC analysis. CT-negative MTC is very rare, with an estimated prevalence of 0.83%, as reported by Franck-Raue et al [5]. To date, the biological mechanism of normal or reduced CT levels in MTC patients has not yet been clarified. It has been assumed that low CT serum level could be linked to impaired production of CT, as shown by CT immunostaining. This hypothesis is related to dedifferentiation of tumor cells, as tumors with only 25% cells IHC-positive for CT are related to a poor prognosis [12]. Frank-Roue et al reported that prognoses of these patients are heterogeneous, varying from slow-progression disease to aggressive biological behavior [5]. Another hypothesis supports the hook effect: false-negative serum CT with high-level tumor marker at immunoassay analysis identified by exceeding antibody binding capacity [15]. The diagnosis in CT-negative MTC, which would allow selection of an optimal surgery strategy, is difficult to obtain. Relating to our patient, negative CT serum and imaging diagnosis of schwannoma of the lateral mass led to a wrong diagnosis. The histologic and immunohistochemical analysis findings after lateral cervical mass biopsy showed metastases by medullary thyroid carcinoma. Subsequently, at our center, we dosed all tumor markers related to MCT, and to prevent errors related to the hook effect, we performed 1: 10 and 1: 100 dilutions of the patient’s serum; findings were within normal ranges. In accordance with thyroid ultrasound imaging (showing a left lobe nodule measuring 0.7×0.38 cm) and immune-histological findings (mts of MTC), the patient underwent total thyroidectomy and left lateral cervical lymph nodes dissection. At histological examination, metastatic lymph nodes of about 1 cm were found in the central compartment, so it was staged as pT1a(m) N1a. However, the final TNM staging (AJCC staging criteria, 8th edition), adding the lymph node metastases of MTC biopsied at the beginning of all patient’s journey, was pT1a(m) N1b.
Conventional follow-up with neck and liver ultrasound were negative for persistence/relapse of disease; RX-chest was negative for pulmonary metastases. However, as it was a non-secretory MTC, so we focused on finding a different follow-up approach compared to the conventional one.
In the literature, the use of positron emission tomography (PET) with radiolabeled amino acid dihydroxyphenylalanine (DOPA), usually performed for gastro-pancreatic neuroendocrine tumors [17,18], has been proposed for CT-negative MTC patient follow-up [16]. 18F-DOPA PET for MTC has a sensitivity of 72% and a specificity of >90%, but limitations of this test are its high cost and its unavailability in many medical centers; consequently, it is not a regularly performed procedure [16]. In our case, 18F-DOPA PET [26,27] was performed, with a negative result for distant metastases.
Only 47 cases of CT-negative MTC are described in the literature (Table 1): 16 had a preoperative diagnosis and 29 had a postoperative incidental diagnosis; only 3 of them had lateral lymph node metastases. A retrospective analysis of 19 cases by Zhou et al [19] evaluated the immunohistochemical examination of the surgical specimens of 158 patients with a diagnosis of medullary thyroid carcinoma. Frank-Roue [5] reported 7 cases with preoperative diagnoses of MTC CT-negative, finding for the first time the prevalence of non-secretory MTC patients with sporadic diseases (prevalence of 0.83%). RET mutation analysis was not performed in all patients reported in the literature, and this is a critical point for collection and analysis of MTC CT-negative patient data.
It is also possible to classify CT-negative MTCs into single-negative, double-negative, and triple-negative. In the single-negative group, only serum CT is negative. In the double-negative group, MTC markers are negative at the time of diagnosis and in case of relapse [28]. Triple-negative patients have a typical medullary thyroid carcinoma histopathological pattern with high negative serum calcitonin, no calcitonin expression on immunohistochemistry, and absence of mRNA in situ hybridization [29]. The patient that we report in this paper belongs to the double-negative group.
As shown in Table 2, CT-negative MTC has a large spectrum of biological behavior, from indolent to life-threatening tumors [20,21]. Moreover, CT-negative and CT-positive patients show no difference in histologic characteristics and RET mutation (Table 3).
The reliability of FNA in CT-negative MTC patients is compromised by intracellular calcitonin concentration. Different MTC intracellular markers could be investigated to be helpful in FNA examination, but this can be difficult due to economic reasons and limited cellular sample availability.
Conclusions
In conclusion, we present a rare clinical case of double-negative, stage IVA MTC. CT-negative MTC is a very rare disease, showing a non-clear biological mechanism crucial for negative serum calcitonin; therefore, CT-negative MTCs, due to their biological characteristics, are usually diagnosed at an advanced stage. To date, due to the rarity of this pathology diagnosis, there are no protocols for CT-negative MTC management that can help physicians in these patients’ clinical journey. Further studies are needed to determine the optimal diagnostic, therapeutic, and follow-up pathways for these patients.
Figures
Tables
Table 1.. Cases of CT- negative MTC reported in the literature, with their time of diagnosis and surgical specimen characteristics.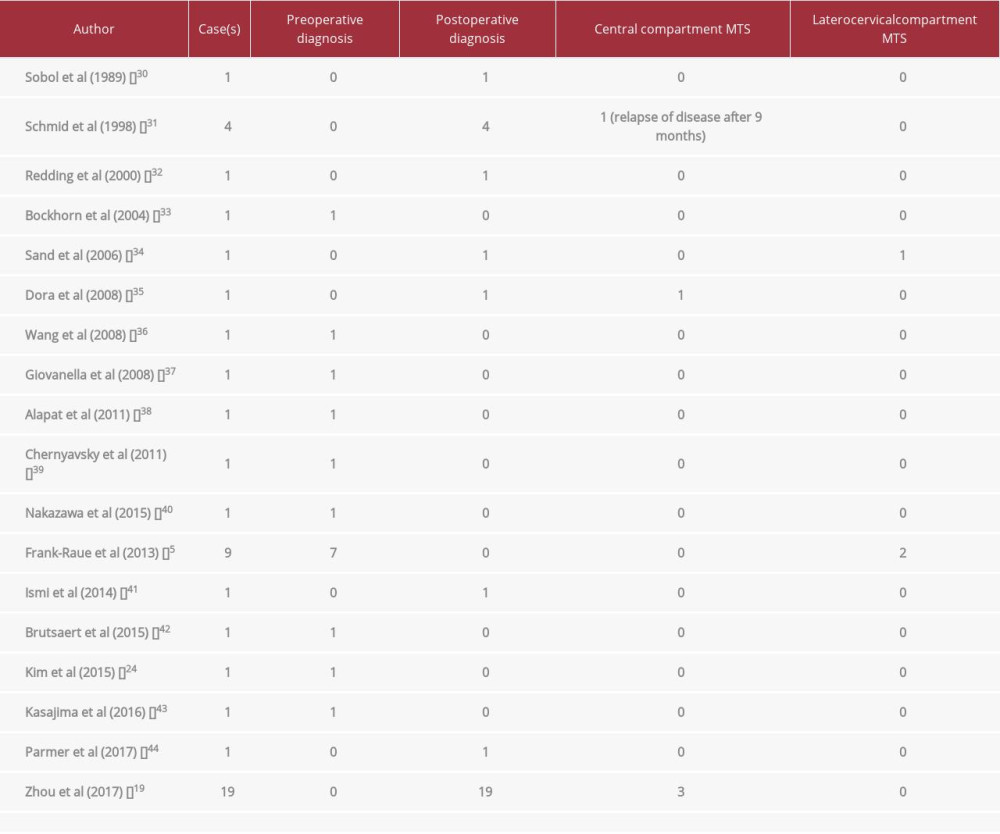 Table 2.. CT-negative patients’ outcome in relation to preoperative and postoperative tumor marker concentrations.
Table 2.. CT-negative patients’ outcome in relation to preoperative and postoperative tumor marker concentrations.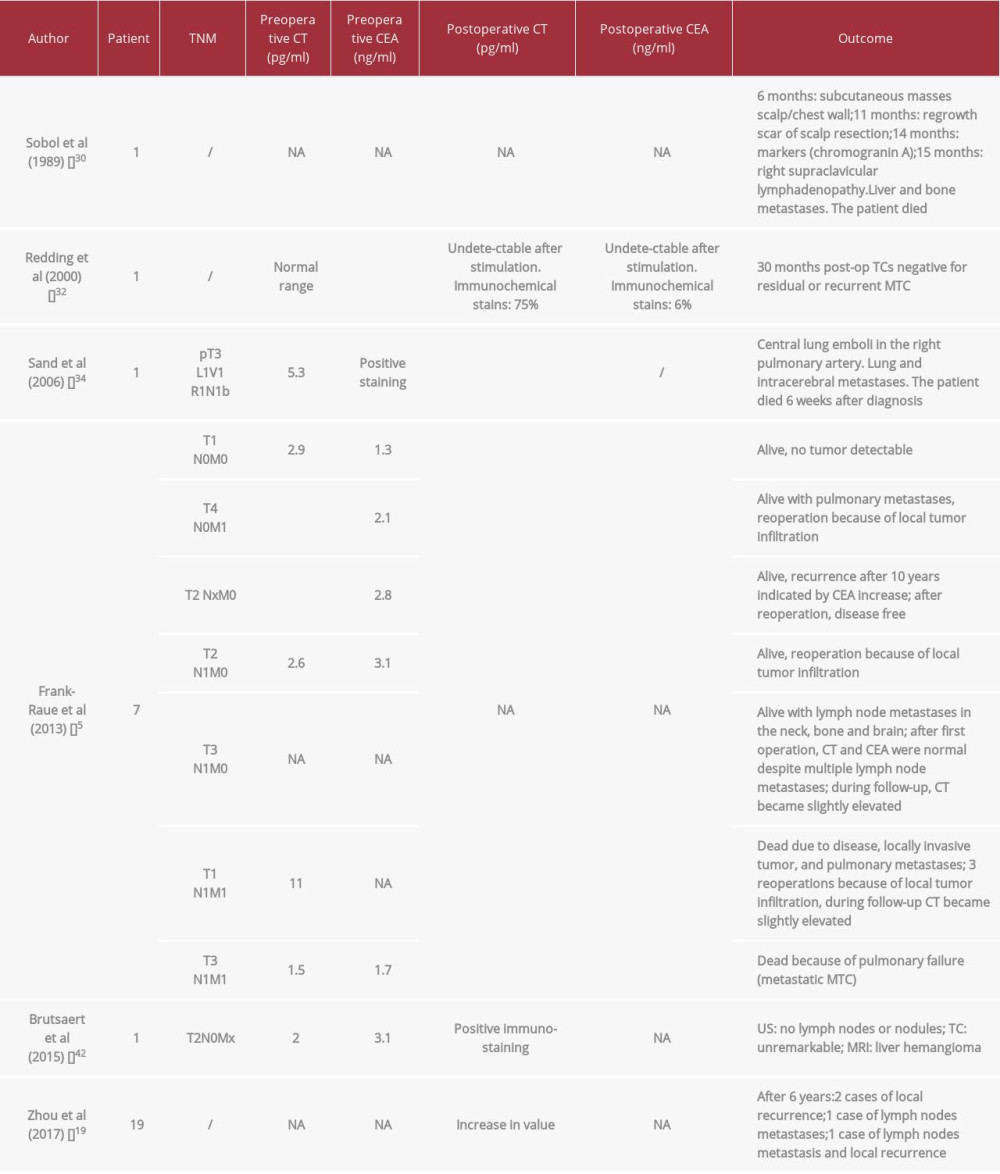 Table 3.. Histologic characteristics and RET mutation in CT-negative and CT-positive patients.
Table 3.. Histologic characteristics and RET mutation in CT-negative and CT-positive patients.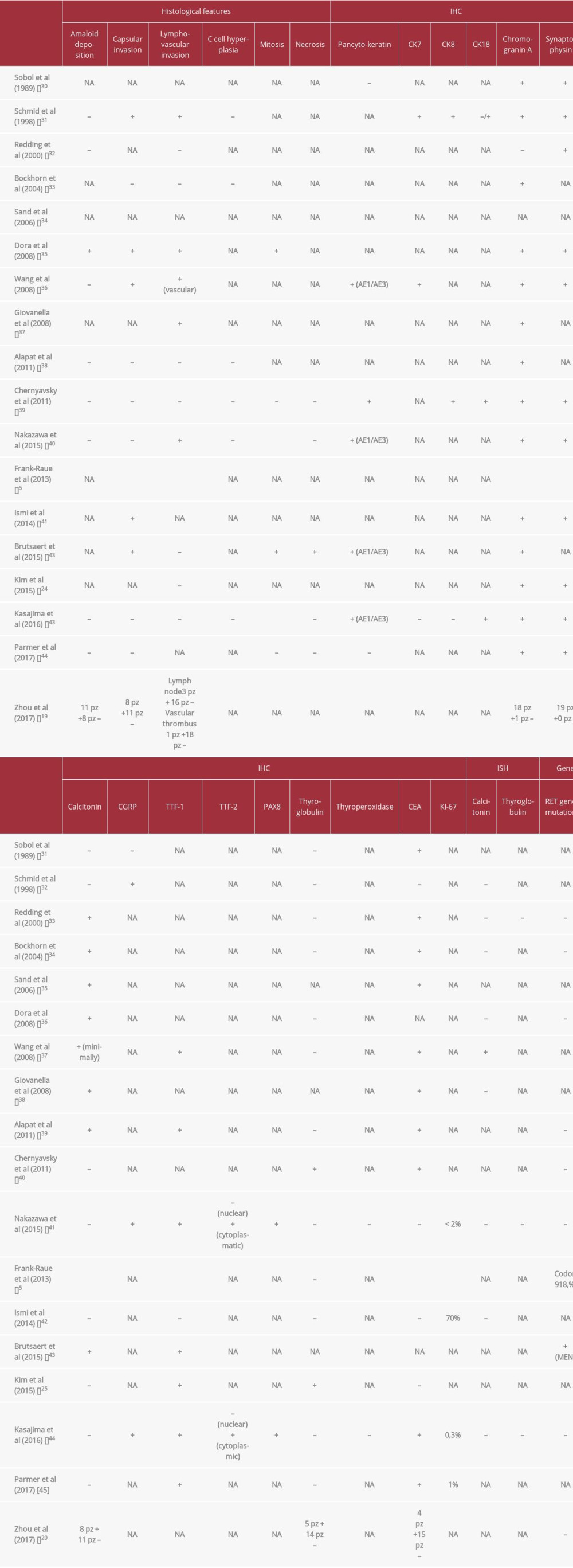
References:
1.. Trimboli P, Giovanella L, Serum calcitonin negative medullary thyroid carcinoma: A systematic review of the literature: Clin Chem Lab Med, 2015; 53(10); 1507-14
2.. Miguel Dora JM, Canalli MH, Capp C, Normal perioperative serum calcitonin levels in patients with advanced medullary thyroid carcinoma: Case report and review of the literature: Thyroid, 2008; 18(8); 895-99
3.. Wells SA, Asa SL, Dralle H, Revised American Thyroid Association guidelines for the management of medullary thyroid carcinoma: Thyroid, 2015; 25(6); 567-610
4.. Elisei R, Lorusso L, Piaggi P, Elevated level of serum carbohydrate antigen 19.9 as predictor of mortality in patients with advanced medullary thyroid cancer: Eur J Endocrinol, 2015; 173(3); 297-304
5.. Frank-Raue K, Machens A, Leidig-Bruckner G, Prevalence and clinical spectrum of nonsecretory medullary thyroid carcinoma in a series of 839 patients with sporadic medullary thyroid carcinoma: Thyroid, 2013; 23(3); 294-300
6.. Trimboli P, Seregni E, Treglia G, Procalcitonin for detecting medullary thyroid carcinoma: A systematic review: Endocr Relat Cancer, 2015; 22(3); R157-64
7.. Kloos RT, Eng C, Evans DB, Medullary thyroid cancer: Management guidelines of the American Thyroid Association: Thyroid, 2009; 19; 565-612
8.. Gharib H, Papini E, Paschke R, American Association of Clinical Endocrinologists, Associazione Medici Endocrinologi, and European Thyroid Association medical guidelines for clinical practice for the diagnosis and management of thyroid nodules: Endocr Pract, 2010; 16; 468-75
9.. Gelos M, Sand D, Bechara FG, Serum calcitonin negative medullary thyroid carcinoma: S World J Surg Oncol, 2006; 4; 97
10.. Brutsaert EF1, Gersten AJ, Tassler AB, Surks MI, Medullary thyroid cancer with undetectable serum calcitonin: J Clin Endocrinol Metab, 2015; 100(2); 337-41
11.. Costante G, Meringolo D, Durante C, Predictive value of serum calcitonin levels for preoperative diagnosis of medullary thyroid carcinoma in a cohort of 5817 consecutive patients with thyroid nodules: J Clin Endocrinol Metab, 2007; 92; 450-55
12.. Cheung K, Roman SA, Wang TS, Calcitonin measurement in the evaluation of thyroid nodules in the United States: A cost-effectiveness and decision analysis: J Clin Endocrinol Metab, 2008; 93; 2173-80
13.. Machens A, Ukkat J, Hauptmann S, Dralle H, Abnormal carcinoembryonic antigen levels and medullary thyroid cancer progression: A multivariate analysis: Arch Surg, 2007; 142; 289-93
14.. Larena A, Vierbuchen M, Fischer R, Blood group antigen expression in malignant tumors of the thyroid: A parallel between medullary and nonmedullary carcinomas: Langenbecks Arch Chir, 1995; 380; 269-72
15.. Leboeuf R, Langlois MF, Martin M, “Hook effect” in calcitonin immunoradiometric assay in patients with metastatic medullary thyroid carcinoma: Case report and review of the literature: J Clin Endocrinol Metab, 2006; 91(2); 361-64
16.. Treglia G1, Castaldi P, Villani MF, Comparison of 18F-DOPA, 18F-FDG and 68Ga-somatostatin analogue PET/CT in patients with recurrent medullary thyroid carcinoma: Eur J Nucl Med Mol Imaging, 2012; 39(4); 569-80
17.. de Groot JW, Links TP, Jager PL, Impact of 18F-fluoro-2-deoxy-D-glucose positron emission tomography (FDG-PET) in patients with biochemical evidence of recurrent or residual medullary thyroid cancer: Ann Surg Oncol, 2004; 11; 786-94
18.. Beuthien-Baumann B, Strumpf A: Eur J Nucl Med Mol Imaging, 2007; 34; 1604-9
19.. Zhou Q, Yue S, Cheng Y, Clinical and pathological analysis of 19 cases of medullary thyroid carcinoma without an increase in calcitonin: Exp Toxicol Pathol, 2017; 69(8); 575-79
20.. Samà MT, Rossetto Giaccherino R, Gallo M, Clinical challenges with calcitonin-negative medullary thyroid carcinoma: J Cancer Res Clin Oncol, 2016; 142(9); 2023-29
21.. Gambardella C, Offi C, Patrone R, Calcitonin negative medullary thyroid carcinoma: A challenging diagnosis or a medical dilemma?: BMC Endocr Disord, 2019; 19(Suppl. 1); 45
22.. Pusztaszeri MP, Bongiovanni M, Faquin WC, Update on the cytologic and molecular features of medullary thyroid carcinoma: Anat Pathol, 2014; 21; 26-35
23.. Essig GF, Porter K, Schneider D, Fine needle aspiration and medullary thyroid carcinoma: The risk of inadequate preoperative valuation and initial surgery when relying upon FNAB cytology alone: Endocr Pract, 2013; 19; 920-27
24.. Kim SH, Kim BS, Jung SL, Ultrasonographic findings of medullary thyroid carcinoma: A comparison with papillary thyroid carcinoma: Korean J Radiol, 2009; 10; 101-5
25.. Trimboli P, Giovanella L, Valabrega S, Ultrasound features of medullary thyroid carcinoma correlate with cancer aggressiveness: A retrospective multicenter study: J Exp Clin Cancer Res, 2014; 33; 87
26.. Brammen L, Niederle MB, Riss P, Medullary thyroid carcinoma: Do ultrasonography and F-DOPA-PET-CT influence the initial surgical strategy?: Ann Surg Oncol, 2018; 25(13); 3919-27
27.. Rasul S, Hartenbach S, Rebhan K, [18F]DOPA PET/ceCT in diagnosis and staging of primary medullary thyroid carcinoma prior to surgery: Eur J Nucl Med Mol Imaging, 2018; 45(12); 2159-69
28.. Gambardella C, Offi C, Clarizia G, Medullary thyroid carcinoma with double negative calcitonin and CEA: A case report and update of literature review: BMC Endocr Disord, 2019; 19(1); 103
29.. Murphy DC, Johnson SJ, Aspinall S, Calcitonin-negative medullary thyroid carcinoma: The ‘triple-negative’ phenotype: Ann R Coll Surg Engl, 2020; 102(3); e63-e66
30.. Sobol RE, Memoli V, Deftos LJ, Hormone-negative, chromogranin A – positive endocrine tumors: N Engl J Med, 1989; 320(7); 444-47
31.. Schmid KW, Ensinger C, “Atypical” medullary thyroid carcinoma with little or no calcitonin expression: Virchows Arch, 1998; 433(3); 209-15
32.. Redding AH, Levine SN, Fowler MR, Normal preoperative calcitonin levels do not always exclude medullary thyroid carcinoma in patients with large palpable thyroid masses: Thyroid, 2000; 10(10); 919-22
33.. Bockhorn M, Frilling A, Rewerk S, Lack of elevated serum carcinoembryonic antigen and calcitonin in medullary thyroid carcinoma: Thyroid, 2004; 14(6); 468-70
34.. Sand M, Gelos M, Sand D, Serum calcitonin negative medullary thyroid carcinoma: World J Surg Oncol, 2006; 4; 97
35.. Dora JM, Canalli MH, Capp C, Normal perioperative serum calcitonin levels in patients with advanced medullary thyroid carcinoma: Case report and review of the literature: Thyroid, 2008; 18(8); 895-99
36.. Wang TS, Ocal IT, Sosa JA, Medullary thyroid carcinoma without marked elevation of calcitonin: A diagnostic and surveillance dilemma: Thyroid, 2008; 18(8); 889-94
37.. Giovanella L, Crippa S, Cariani L, Serum calcitonin-negative medullary thyroid carcinoma: Role of CgA and CEA as complementary markers: Int J Biol Markers, 2008; 23(2); 129-31
38.. Alapat DV, Ain KB, Sloan DA, Disparity between tissue and serum calcitonin and carcinoembryonic antigen in a patient with medullary thyroid carcinoma: Endocrine, 2011; 39(2); 148-52
39.. Chernyavsky VS, Farghani S, Davidov T, Calcitonin-negative neuroendocrine tumor of the thyroid: A distinct clinical entity: Thyroid, 2011; 21(2); 193-96
40.. Nakazawa T, Cameselle-Teijeiro J, Vinagre J, C-cell-derived calcitonin-free neuroendocrine carcinoma of the thyroid: The diagnostic importance of CGRP immunoreactivity: Int J Surg Pathol, 2014; 22(6); 530-35
41.. Ismi O, Arpaci RB, Berkesoglu M, Calcitonin-negative neuroendocrine tumor of thyroid gland mimicking anaplastic carcinoma: An unusual entity: Gland Surg, 2015; 4(4); 344-49
42.. Brutsaert EF, Gersten AJ, Tassler AB, Surks MI, Medullary thyroid cancer with undetectable serum calcitonin: J Clin Endocrinol Metab, 2015; 100(2); 337-41
42.. Kasajima A, Cameselle-Teijeiro J, Loidi L, A calcitonin non-producing neuroendocrine tumor of the thyroid gland: Endocr Pathol, 2016; 27(4); 325-31
44.. Parmer M, Milan S, Torabi A, Calcitonin-negative neuroendocrine tumor of the thyroid: Int J Surg Pathol, 2017; 25(2); 191-94
Figures
Tables
 Table 1.. Cases of CT- negative MTC reported in the literature, with their time of diagnosis and surgical specimen characteristics.
Table 1.. Cases of CT- negative MTC reported in the literature, with their time of diagnosis and surgical specimen characteristics. Table 2.. CT-negative patients’ outcome in relation to preoperative and postoperative tumor marker concentrations.
Table 2.. CT-negative patients’ outcome in relation to preoperative and postoperative tumor marker concentrations. Table 3.. Histologic characteristics and RET mutation in CT-negative and CT-positive patients.
Table 3.. Histologic characteristics and RET mutation in CT-negative and CT-positive patients. Table 1.. Cases of CT- negative MTC reported in the literature, with their time of diagnosis and surgical specimen characteristics.
Table 1.. Cases of CT- negative MTC reported in the literature, with their time of diagnosis and surgical specimen characteristics. Table 2.. CT-negative patients’ outcome in relation to preoperative and postoperative tumor marker concentrations.
Table 2.. CT-negative patients’ outcome in relation to preoperative and postoperative tumor marker concentrations. Table 3.. Histologic characteristics and RET mutation in CT-negative and CT-positive patients.
Table 3.. Histologic characteristics and RET mutation in CT-negative and CT-positive patients. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








