22 July 2022: Articles 
Multidrug-Resistant in a Patient with SARS-Cov-2 Pneumonia in an Intensive Care Unit in Guayaquil, Ecuador: A Case Report
Unusual clinical course
Betty J. Pazmiño Gómez1ACDE*, Jennifer P. Rodas PazmiñoDOI: 10.12659/AJCR.936498
Am J Case Rep 2022; 23:e936498
Abstract
BACKGROUND: Multi-resistant microorganisms are a public health problem. Their incidence has risen due to COVID-19, indiscriminate antibiotics use, corticosteroid treatments, and higher admissions to intensive care units (ICUs) of patients requiring invasive mechanical ventilation. These are risk factors for bacterial over-infection. The present case study that is relevant because of the multiple isolated strains with a resistance pattern: Klebsiella pneumoniae carbapenemases (KPC), extended-spectrum beta lactamases (ESBL) and New Delhi metallo-β-lactamase (NDM) in a patient without comorbidities.
CASE REPORT: A 53-year-old Ecuadorian man with no past medical history arrived at the Emergency Department (ED) with dyspnea, nasopharyngeal swab with a positive reverse transcription polymerase chain reaction (RT-PCR) test for SARS-CoV2, and a chest computed tomography (CT) scan showing bilateral ground-glass pulmonary infiltrates with 40% involvement. On day 10 in the ICU, the presence of Klebsiella pneumoniae KPC strain was reported in an axillary swab culture. Consequently, the antibiotic was rotated to vancomycin 1 g intravenously (i.v.) every 12 h and meropenem 1 g i.v. every 8 h. On day 15 in the ICU, a tracheal secretion culture was reported with the presence of Klebsiella pneumoniae ESBL and a blood culture with Klebsiella pneumoniae NDM.
CONCLUSIONS: The COVID-19 pandemic is a perfect scenario for superinfection with multi-resistant pathogens such as carbapenem-resistant Klebsiella pneumoniae (CRKP), due to the increase in patients admitted to ICUs requiring invasive mechanical ventilation, the use of corticosteroids, and empirical broad-spectrum antibiotic management based on guidelines. The emergence of combined multidrug-resistant strains is a challenge for laboratory detection and the selection of antimicrobial treatment.
Keywords: Klebsiella pneumoniae, COVID-19, Pandemics, Respiration, Artificial, Anti-Bacterial Agents, COVID-19, Ecuador, Humans, Intensive Care Units, Klebsiella Infections, Male, Microbial Sensitivity Tests, Middle Aged, RNA, Viral, SARS-CoV-2
Background
The world is going through a public health crisis that took science and research by surprise: the spread of the SARS-CoV-2 virus, which causes COVID-19 disease. Another alarming situation is multi-resistance to antimicrobials, which is responsible for 700 000 deaths per year worldwide. Carbapenem-resistant
A meta-analysis of 16 studies that involved 3627 participants identified the following risk factors associated with CRPK infection: longer length of hospital stay, admission to an ICU, prior hospitalization, more days in an ICU, transplant recipient, steroid use, and exposure to carbapenems, aminoglyco-sides, glycopeptides, quinolones, and penicillin [7].
It is challenging to differentiate between SARS-CoV-2 and bacterial pneumonia based on clinical signs and symptoms. An accurate microbiological diagnosis is essential to identify bacterial superinfection pneumonia in patients with severe COVID-19 on mechanical ventilation. Bronchoalveolar lavage (BAL) samples are the criterion standard for detecting respiratory pathogens by bronchoscopy and confirming their superiority over nasopharyngeal swabs or endotracheal aspirates [8,9]. However, in many hospitals, BAL procedures were avoided in patients with SARS-CoV-2 pneumonia, as this is an invasive procedure with high risk for the operator [10].
Case Report
A 53-year-old Ecuadorian man with no past medical history presented with 9 days of myalgia, headache, and general malaise. On day 3 after the onset of his symptoms, he had a fever of 38.2°C and polyarthralgia and was given a RT-PCR SARS-CoV-2 test, with a negative result. For symptom management, he received acetaminophen, antihistamines, hydration, and vitamin C. He self-medicated with two 6 mg doses of ivermectin. On day 7, he presented anosmia, dysgeusia, and dyspnea, so he took a second RT-PCR SARS-CoV-2 test, with a positive result. He arrived at the Emergency Department (ED) with a chest computed tomography (CT) scan showing bilateral ground-glass pulmonary infiltrates, with 40% involvement (Figure 1).
His vital signs in the ED showed a respiratory rate of 36 breaths per min, and SaO2 of 90% on room air. His heart rate was 80 beats per minute, his blood pressure was 135/85 mmHg, and his weight 100 kg. A high-flow nasal cannula was placed at 30 L/min with 50% FiO2 with a subsequent respiratory rate of 26 breaths per min and SaO2 of 99%. He received treatment based on dexamethasone, enoxaparin, acetaminophen, acetylcysteine, and bronchodilators. During his second day of hospitalization, the fever persisted with a temperature of 39°C, a respiratory rate of 38 rpm, SpO2 92% with high-flow parameters at 50 L/min and FiO2 of 100%. Therefore, he was admitted to the ICU and endotracheal intubation was carried out. His laboratory findings were leukocytes 10 500/mm3, neutrophils 73.4%, lymphocytes 19.2%, hemoglobin 13.3 g/dl, hematocrit 39.4%, and platelets 196 000/mm3. The first blood, urine, and sputum samples were taken for culture with subsequent initiation of broad-spectrum antibiotics based on piperacillin-tazobactam 4.5 g i.v. every 6 h. Subsequently, the cultures were reported negative. On day 9, axillary, inguinal, and rectal swab samples were taken for the rescue of multi-resistant microorganisms as part of the ICU’s hospital protocol. On day 10 of his stay in the ICU, the presence of
On day 11 in the ICU, due to an increase in leukocytes and inflammatory markers plus lung involvement in a control chest CT scan and isolation of
On day 15 of his stay in the ICU, a tracheal secretion culture was reported with the presence of 10 000 000 CFU/ml
On day 16 of his stay in the ICU, a blood culture was reported with the presence of
Samples of blood cultures, axillary swabs, and tracheal secretions were taken from a patient from Guayaquil, Ecuador. They were inoculated in selective enriched culture media of blood agar, MacConkey, and supplemented chocolate, and placed in an incubator at 37°C for 48 h. Bacterial colonies were recovered, on which the Gram stain and oxidase test were performed. For the sensitivity test, the automated platform BD Phoenix 100 was used. A result was shown in 18 to 24 h with the identification and MIC of the isolate. As complementary tests, an assay was performed of the induction method with ethylenediaminetetraacetic acid (EDTA) and boronic acid (APB) disk to presumptively detect resistance mechanisms, showing a reaction with the EDTA disk (NDM) (Figure 2). The IMP and VIM enzymes have a hydrolytic profile that includes all beta-lactam antibiotics except aztreonam and they are not inhibited by clavulanic acid, sulbactam, or tazobactam. However, they are inhibited by divalent cation chelating agents such as EDTA and thiol compounds such as 2-mercaptopropionic acid or dipicolinic acid. Recently, the NDM-1 enzyme has affected the world due to the multi-resistant or broad-resistant profile generated by these microorganisms.
A rapid test was carried out to detect OXA-48, KPC and NDM in bacterial isolates (RESISTET-3 O.K.N. K-Set/Coris BioConcept). Positive feedback was shown for KPC (Figure 3). As an additional test, a rapid test was performed to detect OXA-48, KPC, and NDM in bacterial isolates (RESISTET-3 O.K.N. K-Set/Coris BioConcept). A positive reaction for NDM (Figure 4) was shown, which was then sent to the reference laboratory, CRN-RAM of the National Institute of Public Health Research (INSPI), to confirm the diagnosis.
Tracheal aspirate showed
Aerobic and anaerobic blood culture showed
A blood culture confirmed at the National Institute of Public Health was performed using the Kirby Bauer manual identification method. The following sensitivity was obtained: sensitive to amikacin and gentamicin and resistant to ceftazidime, cefotaxime, cefepime, ciprofloxacin, aztreonam, meropenem, imipenem, ertapenem, and intermediate colistin MDC 0.5 ug/ ml. CIM-BMD+DD susceptibility tests were carried out and CLSI interpretation standards were met. The results showed a carbapenemase-positive, ABP-IMI synergy-negative, EDTA-IMI synergy-positive, MDM-type carbapenemase-producing micro-organism (Table 3).
Discussion
We presented the case of a patient without comorbidities with severe pneumonia due to COVID-19. The case was complicated by superinfection with CRKP associated with invasive mechanical ventilation in an ICU in Guayaquil, Ecuador.
Two other case studies have been presented of patients in the ICU of a tertiary hospital in Spain. The patients had COVID-19 and developed superinfection by multi-resistant microorganisms associated with risk factors such as the use of mechanical ventilation, corticosteroids, broad-spectrum antibiotic treatment, and increased length of hospital stay [11,12].
An outbreak of multi-resistant
It is assumed that there was non-compliance with standard and contact biosafety regulations. NDM-1K is thought to have been transmitted in the environment due to invasive procedures and the administration of immunomodulatory therapy in patients with COVID-19. Failure to change gloves between patient check-ups is possibly one of the causes of the propagation of NDM-1K-producing bacteria. Non-compliance with biosafety standards occurred worldwide as ICUs were overwhelmed and understaffed, and the lack of sanitary implements to control these multi-resistance outbreaks aggravated the patients’ situations and raised the risk of mortality [13].
In a study, patients with a positive result in the reverse transcriptase polymerase chain reaction (RT-PCR) test for the SARSCoV-2 virus were included in the COVID-19 group, and more than 1 species of MMR was detected. The positivity of the analyzed samples was made in axillary (2%), pharyngeal (2.4%), nasal (0.9%), and rectal (8.2%) exudates. The latter were the most significant samples for the detection of ESBL-producing Enterobacteriaceae [11].
Clinical teams in Chicago’s ICUs routinely obtained BAL fluid from mechanically ventilated patients at the time of intubation and when ventilator-associated pneumonia (VAP) was clinically suspected to administer antibiotics. With the initial surge of COVID-19 in Chicago, the standard bronchoscopy and bronchoalveolar lavage (BAL) technique was modified to minimize operator exposure to infectious aerosols [14]. Based on ICU hospital protocol, axillary, inguinal, and rectal swab samples are obtained for screening multi-resistant microorganisms, similar to a studio of optimization of multiple muco-cutaneous site sampling method for screening MRSA colonization in ICUs [15].
In Michigan in 2018,
The INEI-ANLIS antimicrobial service “Dr Carlos Malbrán” (National Reference Laboratory LNR) in Argentina confirmed during the first wave of the COVID-19 pandemic the health emergency and dissemination by combined carbapenemase-producing
In Latin America, 11.6% of hospitalized patients develop nosocomial infections caused by different procedures. In Ecuador, mortality due to multi-resistant microorganisms is approximately 50%. Complete surveillance of CRKP in Ecuador has not been reported previously, although some cases have been described (23 cases reported in 2016) [18,19].
The clinical case described here presents the same risk factors as the studies carried out in Spain on patients with COVID-19. Some case reports of combined multi-resistant strains were published in years prior to the COVID-19 pandemic. In contrast, during the first wave of COVID-19 cases in Argentina, co-expression of classes of carbapenemases in
Conclusions
The COVID-19 pandemic generated a perfect scenario for the superinfection of multi-resistant pathogens such as CRKP, due to the increase in patients admitted to ICUs who required invasive mechanical ventilation, the use of corticosteroids, and empirical broad-spectrum antibiotic management based on guidelines. The emergence of combined multidrug-resistant strains constitutes a challenge for laboratory detection and selection of specific antimicrobial treatment.
Biosafety measures and immediate communication with the infectious diseases service must be carried out once a patient with CRKP has been identified for decision making to prevent the spread of life-threatening multi-resistant microorganisms.
Figures
Tables
Table 1.. Tracheal aspirate MIC Klebsiella pneumoniae, ESBL strain.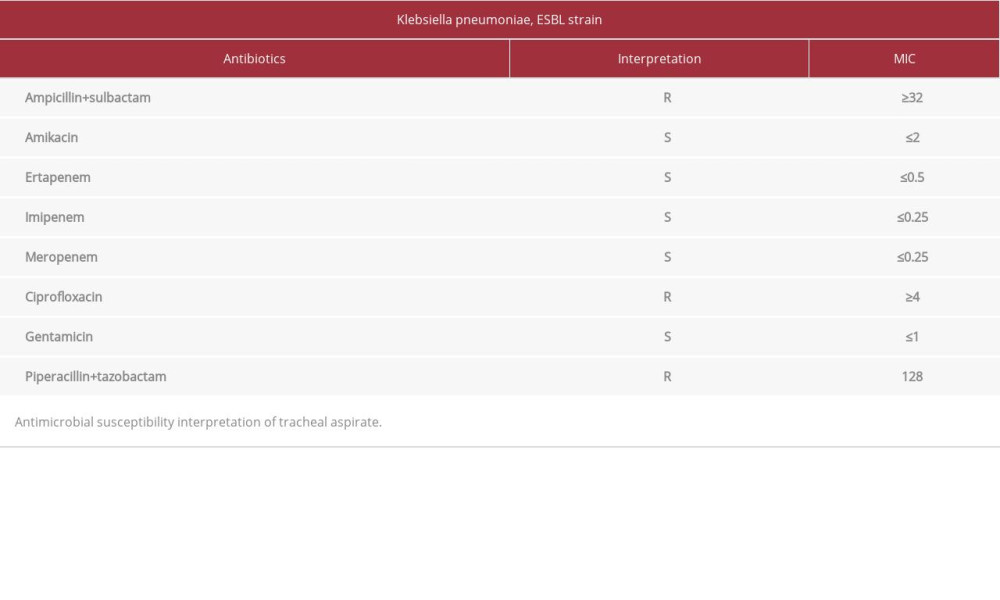 Table 2.. Blood culture and axillar swab MIC Klebsiella pneumoniae, NDM strain.
Table 2.. Blood culture and axillar swab MIC Klebsiella pneumoniae, NDM strain.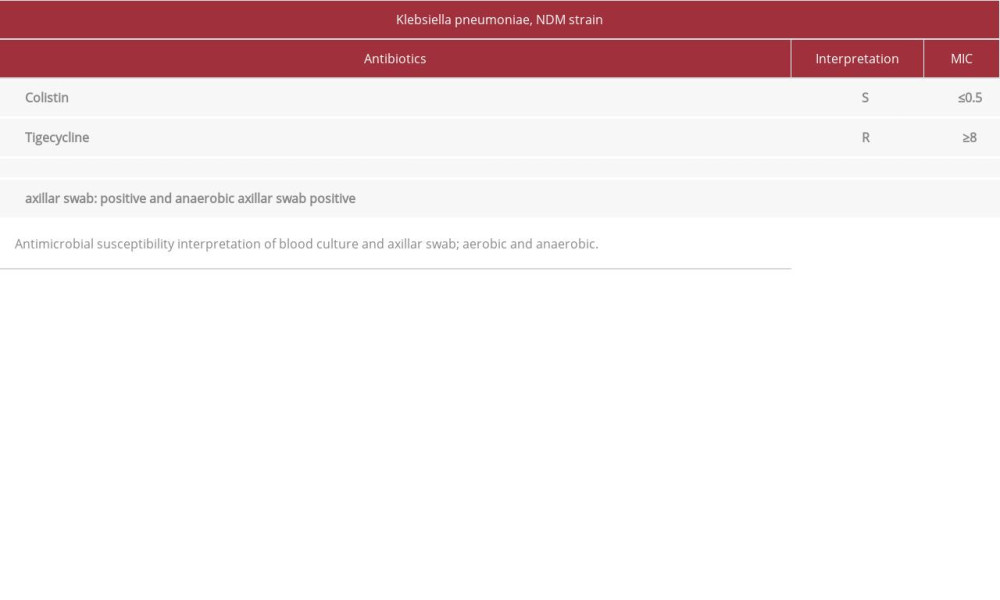 Table 3.. Blood culture confirmation Klebsiella pneumoniae, NDM strain (confirmed at the INSPI Guayaquil, Ecuador). Microbiological findings.Klebsiella pneumoniae ss. Pneumonia.
Table 3.. Blood culture confirmation Klebsiella pneumoniae, NDM strain (confirmed at the INSPI Guayaquil, Ecuador). Microbiological findings.Klebsiella pneumoniae ss. Pneumonia.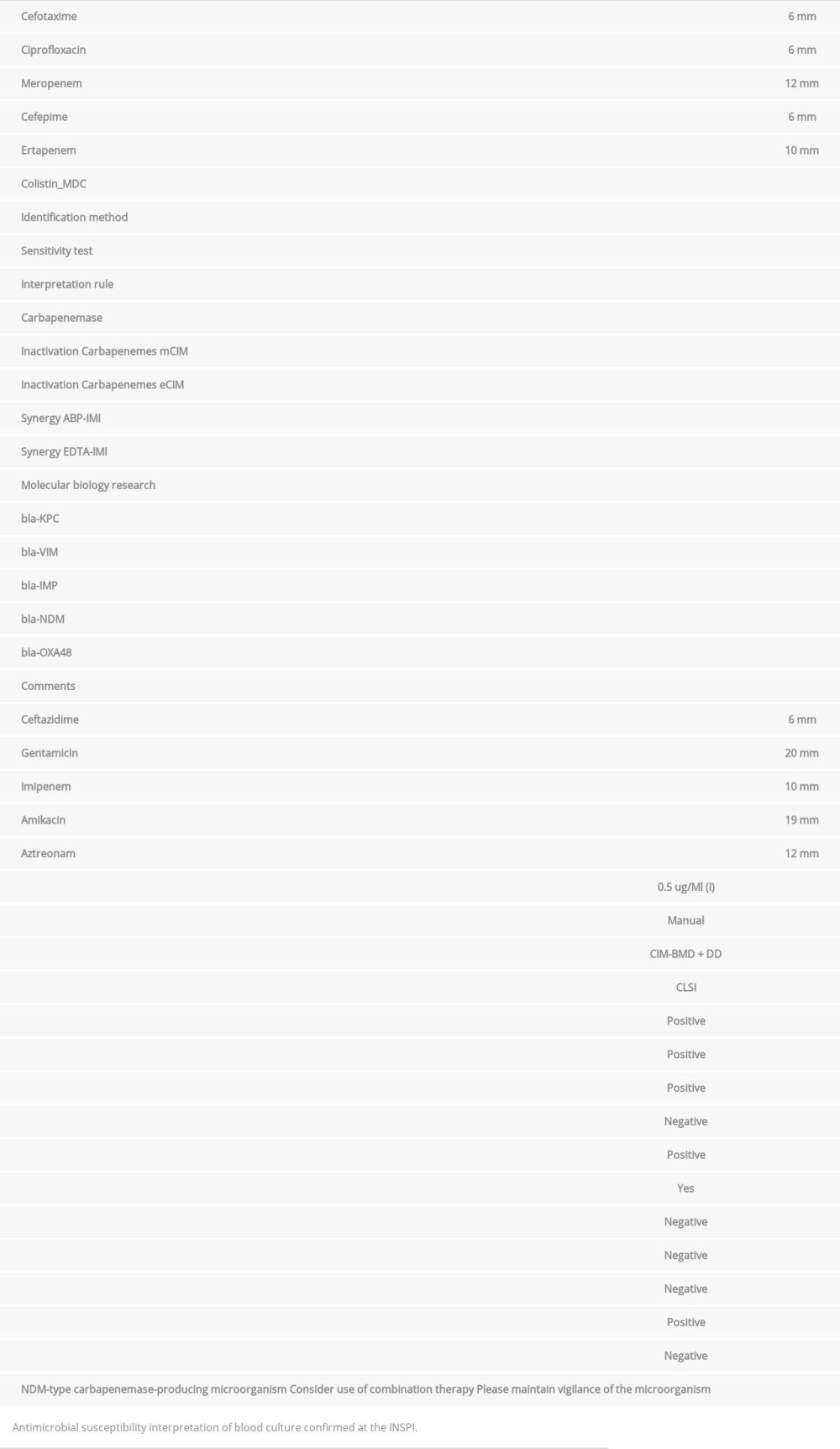
References:
1.. Lee C-R, Lee JH, Park KS: Front Microbiol, 2016; 7; 895
2.. da Silva JB, Espinal M, Ramón-Pardo P, Resistencia a los antimicrobianos: Tiempo para la acción: Rev Panam Salud Pública, 2020; 44; e122 [in Spanish]
3.. Yauri M, Rodríguez M, Alcocer I: Infectio, 2019; 24(1); 42-49
4.. Rojo V, Vázquez P, Reyes S: Rev Española Quimioter, 2018; 31(5); 427 [in Spanish]
5.. Prats GU de B, Microbiología clínica prats: Panamericana, 2005, España editor.
6.. Kazmierczak KM, Karlowsky JA, de Jonge BLM, Epidemiology of carbapenem resistance determinants identified in meropenem-nonsusceptible enterobacterales collected as part of a Global Surveillance Program, 2012 to 2017: Antimicrob Agents Chemother, 2021; 65(7); e0200020
7.. Liu P, Li X, Luo M: Microb Drug Resist, 2018; 24(2); 190-98
8.. Torres A, Niederman MS, Chastre J, International ERS/ESICM/ESCMID/ ALAT guidelines for the management of hospital-acquired pneumonia and ventilator-associated pneumonia: Guidelines for the management of hospital-acquired pneumonia (HAP)/ventilator-associated pneumonia (VAP) of the European Respiratory Society (ERS), European Society of Intensive Care Medicine (ESICM), European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and Asociación Latinoamericana del Tórax (ALAT): Eur Respir J, 2017; 50(3); 17000582
9.. Gao CA, Cuttica MJ, Malsin ES, Comparing nasopharyngeal and BAL SARS-CoV-2 assays in respiratory failure: Am J Respir Crit Care Med, 2021; 203(1); 127-29
10.. Wahidi MM, Shojaee S, Lamb CR, The use of bronchoscopy during the coronavirus disease 2019 pandemic: CHEST/AABIP Guideline and Expert Panel Report: Chest, 2020; 158(3); 1268-81
11.. Fernández P, Moreno L, Yagüe G, Colonización por microorganismos multirresistentes en pacientes de UCI durante la pandemia de la COVID-19: Elsevier [Internet], 2021; 45; 313-19
12.. Ramos R, de la Villa S, García-Ramos S, COVID-19 associated infections in the ICU setting: A retrospective analysis in a tertiary-care hospital: Enferm Infecc Microbiol Clin, 2021 [Online ahead of print]
13.. Amarsy R, Jacquier H, Munier AL: Am J Infect Control, 2021; 49(10); 1324-26
14.. Pickens CO, Gao CA, Cuttica MJ, Bacterial superinfection pneumonia in patients mechanically ventilated for COVID-19 pneumonia: Am J Respir Crit Care Med, 2021; 204(8); 921-32
15.. Datta P, Vasdeva HR, Chander J, Optimization of multiple muco-cutaneous site sampling method for screening MRSA colonization in ICU: Indian J Crit Care Med, 2013; 17(4); 243
16.. Zhang Y, Gu D, Yang X: Front Microbiol, 2022; 12; 3920
17.. Pasteran F, Corso A, Programa nacional de control de calidad en bacteriología boletín informativo Nro. 4-abril 2021: Programa Nac Control Calid en Bacteriol, 2021; 4; 12 [in Spanish]
18.. Lespada MI, Córdova E, Roca V: Estudio comparativo y evolución en 7 años, 2019; 32(1); 15-21
19.. Prado-Vivar MB, Ortiz L, Reyes J, Molecular typing of a large nosocomial outbreak of KPC-producing bacteria in the biggest tertiary-care hospital of Quito, Ecuador: J Glob Antimicrob Resist [Internet], 2019; 19; 328-32
Figures
Tables
 Table 1.. Tracheal aspirate MIC Klebsiella pneumoniae, ESBL strain.
Table 1.. Tracheal aspirate MIC Klebsiella pneumoniae, ESBL strain. Table 2.. Blood culture and axillar swab MIC Klebsiella pneumoniae, NDM strain.
Table 2.. Blood culture and axillar swab MIC Klebsiella pneumoniae, NDM strain. Table 3.. Blood culture confirmation Klebsiella pneumoniae, NDM strain (confirmed at the INSPI Guayaquil, Ecuador). Microbiological findings.Klebsiella pneumoniae ss. Pneumonia.
Table 3.. Blood culture confirmation Klebsiella pneumoniae, NDM strain (confirmed at the INSPI Guayaquil, Ecuador). Microbiological findings.Klebsiella pneumoniae ss. Pneumonia. Table 1.. Tracheal aspirate MIC Klebsiella pneumoniae, ESBL strain.
Table 1.. Tracheal aspirate MIC Klebsiella pneumoniae, ESBL strain. Table 2.. Blood culture and axillar swab MIC Klebsiella pneumoniae, NDM strain.
Table 2.. Blood culture and axillar swab MIC Klebsiella pneumoniae, NDM strain. Table 3.. Blood culture confirmation Klebsiella pneumoniae, NDM strain (confirmed at the INSPI Guayaquil, Ecuador). Microbiological findings.Klebsiella pneumoniae ss. Pneumonia.
Table 3.. Blood culture confirmation Klebsiella pneumoniae, NDM strain (confirmed at the INSPI Guayaquil, Ecuador). Microbiological findings.Klebsiella pneumoniae ss. Pneumonia. In Press
06 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.942937
12 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943244
13 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943275
13 Mar 2024 : Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.943411
Most Viewed Current Articles
07 Mar 2024 : Case report 
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133
10 Jan 2022 : Case report 
DOI :10.12659/AJCR.935263
Am J Case Rep 2022; 23:e935263
19 Jul 2022 : Case report 
DOI :10.12659/AJCR.936128
Am J Case Rep 2022; 23:e936128
23 Feb 2022 : Case report 
DOI :10.12659/AJCR.935250
Am J Case Rep 2022; 23:e935250








