31 August 2022: Articles 
Posterior Myocardial Infarction in a 45-Year-Old Javanese Woman with a 1-Month History of COVID-19-Related Guillain-Barré Syndrome: A Challenging Emergency Diagnosis
Challenging differential diagnosis
Ivan Satria PratamaDOI: 10.12659/AJCR.937105
Am J Case Rep 2022; 23:e937105
Abstract
BACKGROUND: Guillain-Barré syndrome (GBS) is an autoimmune demyelinating disease that affects peripheral nerves and may be associated with nerve pain in the upper limbs and chest. Autonomic dysfunction in GBS can result in electrocardiography (ECG) changes that include T wave inversion, ST segment depression, or ST segment elevation. Recently, GBS was been recognized as a neurological consequence of COVID-19. This report describes the challenge of emergency diagnosis of posterior myocardial infarction (MI) in a 45-year-old Javanese woman who was known to have a 1-month history of COVID-19-related Guillain-Barré syndrome.
CASE REPORT: We report the case of a 45-year-old patient who presented to the Emergency Department (ED) with atypical angina. She had a history of GBS that started 2 weeks after she developed COVID-19. Since then, she frequently had pain in both legs and occasionally in the chest. Her electrocardiogram revealed subtle ST segment depression in the anteroseptal leads (V1-V4), along with ST segment elevation in the posterior leads (V7-V9). Cardiac marker (troponin I) was elevated and posterior regional wall motion abnormality was present on an echocardiogram. Coronary angiography revealed total occlusion of the first diagonal branch of the LAD, followed by deployment of drug-eluting stents to achieve good angiographic results. The patient was diagnosed with GBS and isolated posterior ST segment elevation myocardial infarction.
CONCLUSIONS: This report shows the importance of performing standard cardiac investigations for myocardial ischemia or infarction in patients known to have Guillain-Barré syndrome so that the patient can be treated appropriately and urgently to ensure the best possible outcome.
Keywords: Electrocardiography, Guillain-Barre Syndrome, ST elevation myocardial infarction, Arrhythmias, Cardiac, COVID-19, COVID-19 Testing, Female, Humans, Indonesia, Middle Aged
Background
Guillain-Barré syndrome is an immune-mediated polyneuropathy that affects the peripheral nervous system. It is caused by a cross-reaction of an immunological response to an earlier nonspecific respiratory or gastrointestinal infection that shares epitopes on peripheral nerves. The reported incidence worldwide varies from 0.4 to 3.25 per 100 000 people per year [1]. GBS was recently included as one of the neurological complications of COVID-19. As of July 2020, there were 73 reported cases of COVID-19-associated cases of GBS. However, the etiologies of these 2 diseases remain unclear [2]. The classic clinical manifestations of GBS are limb weakness and areflexia, but sensory disturbances such as paresthesia or neuropathic pain and autonomic nervous system disorders such as loss of vascular tone and tachy- or brady-arrhythmia can also occur [1]. These autonomic nervous system changes may result in cardiogenic complications and electrocardiography (ECG) abnormalities [1,3].
Myocardial infarction (MI) is defined as cardiac injury in the setting of evidence of acute myocardial ischemia. Posterior wall MI occurs when there is an obstruction of blood flow to the posterior circulation. It frequently co-occurs with inferior or inferolateral MI, but when seen alone, posterior myocardial infarction poses a diagnostic challenge. The clinical symptoms of posterior MI are similar to the clinical symptoms of MI in other regions, but in females, older age, history of diabetes mellitus (autonomic neuropathy), dementia, or chronic renal disease atypical presentation (angina equivalent) often occur, thus blurring the diagnosis of MI [4,5].
Concurrent GBS and MI are rare, with only a few cases previously reported. GBS-related chest pain and ECG abnormalities may conceal the presence of MI. Furthermore, ECG abnormalities in isolated posterior MI are difficult to detect, complicating the diagnosis of MI. Delays or failure to recognize MI symptoms and ECGs can result in a poor prognosis or even death. As a result, better recognition of individuals with atypical MI presentations may reduce the medical mistakes associated with a missed or incorrect diagnoses. This report describes the challenge of emergency diagnosis of posterior myocardial infarction in a 45-year-old Javanese woman who was known to have a 1-month history of COVID-19-associated Guillain-Barré syndrome.
Case Report
A 45-year-old Javanese woman came to the Emergency Department (ED) with atypical chest pain and a burning sensation in the left chest, radiating to both arms, back, and legs. The pain suddenly started 2 hours before her visit to the ER. She had a long history of uncontrolled hypertension and was hospitalized for GBS 1 month ago.
Six weeks before this visit, the patient had been diagnosed with mild COVID-19. Two weeks later, she reported weakness in both legs accompanied by tingling and stabbing pain in the legs, abdomen, and chest. She denied any difficulty breathing or speaking or weakness of facial muscles. Electromyography and a nerve conduction study showed signs of lower motor neuron demyelination, a lumbar puncture showed cytoalbumin dissociation, and a diagnosis of GBS was made. She received supportive treatment and the weakness of the lower limbs improved. However, since then, she frequently had pain in both arms and legs, and occasionally in the chest.
Vital sign examination results were as follow: blood pressure was 160/90 mmHg, heart rate was 105 beats/min, respiratory rate was 20 breaths/min, and peripheral oxygen saturation was 99% with 4 L/min oxygen from nasal cannula. Physical examination revealed a single S1 and S2 heart sound without extrasystole, gallop, or murmur. No rales or wheezing were detected in either lung. Upper- and lower-extremity paraplegia and proprioceptive pain were found.
A 12-lead ECG revealed horizontal ST segment depression in leads V1–V4 with an increase in cardiac marker (troponin I level=4.37 ng/L; reference value: 0.1 ng/L) (Figure 1). These findings indicated an infarct in the patient’s myocardium. The posterior LV wall appeared hypokinetic on echocardiography, with a left ventricular ejection fraction (EF) of 46% (normal value ≥50%) by Biplane. Due to these findings, we performed a posterior ECG to rule out isolated posterior LV wall infarction. The posterior ECG showed ST segment elevation in leads V7–V9 and occasional PVC (Figure 2). Subsequently, we diagnosed the patient with isolated posterior ST segment elevation myocardial infarction (STEMI) and prepared her for percutaneous coronary intervention (PCI).
Coronary angiography revealed a normal left main coronary artery (LMCA), right coronary artery (RCA), and left circumflex coronary artery (LCX) with small LCx diameter. The left anterior descending (LAD) artery appeared normal, but its first diagonal branch (D1) was completely occluded (Figure 3). A 2.25×16 mm drug-eluting stent (DES) was deployed in the D1 branch, achieving acceptable angiographic results (TIMI grade III flow) (Figure 4).
The patient’s chest pain was greatly reduced after stent placement. She was discharged after 5 days of therapy with dual antiplatelet, statin, beta blocker, and angiotensin receptor blocker medicines. Because the patient’s GBS had reached a plateau, she was given a neuroprotectant medicine.
Discussion
This case report demonstrates COVID-19-associated GBS masking the presentation of posterior myocardial infarction. GBS is mostly diagnosed based on clinical symptoms. Its clinical features include progressive symmetrical paralysis of the arms or legs, cranial nerve weakness (facial weakness, bulbar, and external ophthalmoplegia), respiratory muscle weakness, hyporeflexia or areflexia of the affected limb, and autonomic disturbances. Supportive features include mild sensory dysfunction (paresthesia, pain, or numbness), prodromal symptoms of febrile respiratory or gastrointestinal infection, and cytoalbumin dissociation (elevated protein and normal to mildly elevated leucocyte level in cerebrospinal fluid). Initial symptoms may appear within a few days to a week and typically progress over a 2-week period. More than 90% of patients have reached the disease’s nadir within 4 weeks after onset of symptoms [1,6,7].
COVID-19 has recently been associated with GBS. As of July 2020, 73 cases of COVID-19 associated GBS have been reported. The GBS symptoms started around a median of 14 days after onset of COVID-19, and the clinical picture usually resembled classic sensorimotor GBS [2]. Most of these cases were reported as case reports, and direct causality has not been established. The mechanism of this association is not yet clear but may be caused by autoimmunity triggered by the viral peptide [8]. Similar autoimmune demyelinating diseases have been reported to be associated with other coronavirus infections [9].
Cardiovascular complications in GBS are attributable to autonomic dysfunction and are observed in around two-thirds of affected patients [10]. These complications can be explained in part by catecholamine-associated myocardial injury theories, which include a disturbance of catecholamine uptake surrounding myocytes [11], redistribution of coronary blood flow [12], and myocardium denervation hypersensitivity [3]. Cardiovascular complications that can be caused by GBS include heart rate variability, hypertension or hypotension, cardiomyopathy, acute coronary syndrome, and electrocardiographic changes [3,13]. These changes are thought to be related to catecholamine imbalance during GBS and may cause coronary vasospasm and aggravate myocardial ischemia in patients who are at risk [7,14].
Myocardial infarction should be considered in GBS patients, especially if they have concurrent chest pain and ECG abnormalities. In the present case, the patient had atypical chest discomfort in the form of a burning sensation in the left chest that spread to the back and both the arms and legs. Complaints of burning sensations in the hands, feet, and chest were noticed since the patient’s GBS diagnosis. These symptoms may have obscured the presence of an acute MI because they resemble atypical an-gina symptoms. A prospective cohort study reported that neuropathic pain is a common problem in GBS, affecting 66% of patients in the acute phase (the first 3 weeks), mainly including muscle pain, painful paresthesia/dysesthesias, radicular pain, arthralgia, and meningism. During the acute phase, the pain is usually felt in the extremities (70%), back or low back (35%), interscapular area (28%), neck (28%), and trunk (11%) [15].
According to an observational study, complications of acute coronary syndrome occurred in 2.08% of GBS patients. Furthermore, 50% of patients showed ECG abnormalities, with sinus tachycardia (58.33%) being the most prevalent, followed by ST-T segment abnormality (25%). T wave inversion, ST segment depression, and ST segment elevation were found in 4.17%, 6.25%, and 4.17% of the patients, respectively [16]. There is also a 3.12% chance of increased troponin T and an 8.33% chance of abnormal 2D echocardiography findings. In several case reports, concurrent GBS and coronary occlusion were associated with intravenous immunoglobulin therapy, which could be attributed to an elevated risk of prothrombotic condition caused by immunoglobulin medication [17–19]. In one report, cardiac catheterization revealed normal coronary artery morphology, whereas another report showed coronary spasm due to dysautonomia and catecholamine elevation [12,20].
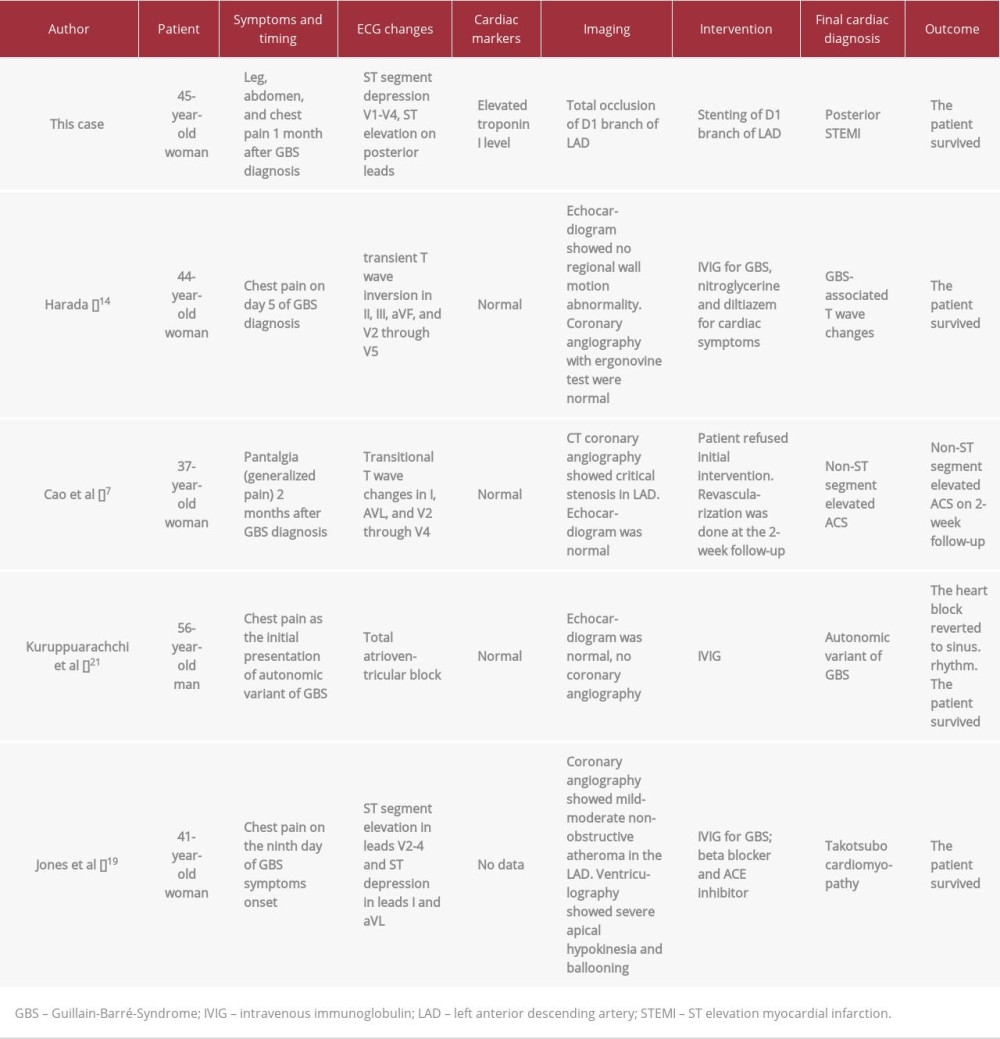
There were also case reports of signs of myocardial ischemia in GBS patients, showing that GBS may mimic or induce myocardial ischemia. Three out of 4 of these case reports described chest pain within 9 days of GBS onset, while 1 case report showed chest pain as the initial symptom of GBS. The final cardiac diagnosis of each case was GBS-associated T wave changes [14], takotsubo cardiomyopathy [19], total AV block associated with autonomic variant of GBS [21], and non-ST segment elevated acute coronary syndrome [7]. One case report noted that T wave changes without signs of MI can occur in GBS, but this diagnosis can only be made after exclusion of significant pathology in the heart and coronary arteries [14]. These findings are summarized in Table 1.
Acute posterior MI is frequently associated with lateral or inferior MI (15–21%). However, isolated posterior MI is rare, accounting for 3.3% of all MI. Unlike the anterior, lateral, and inferior left ventricle walls, none of the typical 12-lead ECG leads cover the posterior wall, and initially posterior MI is detected by reciprocal changes in the standard anterior leads (V1–V3). According to the European Society of Cardiology, isolated posterior MI ECG changes are defined as isolated ST segment depression greater than or equal to 0.5 mm in leads V1–V3 and ST segment elevation greater than or equal to 0.5 mm in posterior chest wall leads V7–V9. Due to milder ECG abnormalities, the detection of an isolated posterior MI is more likely to be missed. In the present patient, 12-lead ECG demonstrated ST depression in the V1–V4 segments and ST segment elevation in the V7–V9 segments. Moreover, she had elevated cardiac marker (Troponin I=4.37) and posterior wall motion abnormality on echocardiography examination. Subsequently, the patient was diagnosed as isolated posterior STEMI and was scheduled for PCI [4].
Conclusions
This report has highlighted the importance of performing standard cardiac investigations for myocardial ischemia or infarction in patients with known Guillain-Barré syndrome so that the patient can be treated appropriately and urgently to ensure the best possible outcome.
Figures
References:
1.. Shahrizaila N, Lehmann HC, Kuwabara S, Guillain-Barré syndrome: Lancet, 2021; 397(10280); 1214-28
2.. Abu-Rumeileh S, Abdelhak A, Foschi M, Guillain-Barré syndrome spectrum associated with COVID-19: An up-to-date systematic review of 73 cases: J Neurol, 2021; 268(4); 1133-70
3.. Mukerji S, Aloka F, Farooq MU, Cardiovascular complications of the Guillain-Barré syndrome: Am J Cardiol, 2009; 104(10); 1452-55
4.. Lizzo JM, Chowdhury YS, Posterior Myocardial infarction. [Updated 2022 May 2]: StatPearls [Internet] Jan, 2022, Treasure Island (FL), StatPearls Publishing Available from: https://www.ncbi.nlm.nih.gov/books/NBK553168/
5.. Collet JP, Thiele H, Barbato E, 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: Supplementary data: Eur Heart J, 2021; 42(14); 1289-367
6.. Fokke C, van den Berg B, Drenthen J, Diagnosis of Guillain-Barré syndrome and validation of Brighton criteria: Brain, 2014; 137(Pt 1); 33-43
7.. Cao X, Tang M, Liu H, Yue X, Case report: Abnormal ECG and pantalgia in a patient with Guillain-Barré syndrome: Front Cardiovasc Med, 2021; 8; 742740
8.. Khan F, Sharma P, Pandey S, COVID-19-associated Guillain-Barré syndrome: Postinfectious alone or neuroinvasive too?: J Med Virol, 2021; 93(10); 6045-49
9.. Whittaker A, Anson M, Harky A, Neurological manifestations of COVID-19: A systematic review and current update: Acta Neurol Scand, 2020; 142(1); 14-22
10.. Flachenecker P, Wermuth P, Hartung HP, Reiners K, Quantitative assessment of cardiovascular autonomic function in Guillain-Barré syndrome: Ann Neurol, 1997; 42(2); 171-79
11.. Iga K, Himura Y, Izumi C, Reversible left ventricular dysfunction associated with Guillain-Barré syndrome – an expression of catecholamine cardiotoxicity?: Jpn Circ J, 1995; 59(4); 236-40
12.. Dagres N, Haude M, Baumgart D, Assessment of coronary morphology and flow in a patient with Guillain-Barré syndrome and ST-segment elevation: Clin Cardiol, 2001; 24(3); 260-63
13.. Chakraborty T, Kramer CL, Wijdicks EFM, Rabinstein AA, Dysautonomia in Guillain-Barré syndrome: Prevalence, clinical spectrum, and outcomes: Neurocrit Care, 2020; 32(1); 113-20
14.. Harada Y, Transient T-wave changes in Guillain-Barré syndrome: J Gen Fam Med, 2017; 18(6); 460-61
15.. Ruts L, Drenthen J, Jongen JLM, Pain in Guillain-Barré syndrome: A long-term follow-up study: Neurology, 2010; 75(16); 1439-47
16.. Gupta S, Verma R, Sethi R, Cardiovascular complications and its relationship with functional outcomes in Guillain-Barré syndrome: QJM, 2020; 113(2); 93-99
17.. Barsheshet A, Marai I, Appel S, Zimlichman E, Acute ST elevation myocar-dial infarction during intravenous immunoglobulin infusion: Ann NY Acad Sci, 2007; 1110; 315-18
18.. Vinod KV, Kumar M, Nisar KK, High dose intravenous immunoglobulin may be complicated by myocardial infarction: Indian J Crit Care Med, 2014; 18(4); 247-49
19.. Jones T, Umaskanth N, De Boisanger J, Penn H, Guillain-Barré syndrome complicated by takotsubo cardiomyopathy: An under-recognised association: BMJ Case Rep, 2020; 13(2); e233591
20.. Hiraga A, Nagumo K, Suzuki K, [A patient with Guillain-Barré syndrome and recurrent episodes of ST elevation and left ventricular hypokinesis in the anterior wall.]: No To Shinkei, 2003; 55(6); 517-20 [in Japanese]
21.. Kuruppuarachchi A, Sundaresan K, Thivakaran T, Rare presentation of Guillain-Barré syndrome with complete heart block and neuropathic chest pain mimicking acute coronary syndrome: A case report: Ceylon Med J, 2018; 63(3); 149-50
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133