07 September 2022: Articles 
A Case of Segmental Arterial Mediolysis of Multiple Visceral Arteries Following Anti-COVID-19 Vaccination: Late Complication or Rare Coincidence?
Unusual clinical course, Challenging differential diagnosis, Diagnostic / therapeutic accidents, Management of emergency care, Unexpected drug reaction, Clinical situation which can not be reproduced for ethical reasons
Akihiro Takeda1ABCDEF*, Wataru Koike2BCD, Fumihiro Okumura3BCDDOI: 10.12659/AJCR.937505
Am J Case Rep 2022; 23:e937505
Abstract
BACKGROUND: Segmental arterial mediolysis (SAM) is a rare noninflammatory, nonatherosclerotic vascular disorder characterized by arterial media disruption. In conjunction with the SARS-CoV-2 infection or anti-COVID-19 vaccination, vascular disorders have been recognized as organ-specific immune-mediated complications, and the number of reported cases is gradually increasing.
CASE REPORT: A 68-year-old man presented with severe upper abdominal pain and nausea 58 days after a third injection of Pfizer-BioNTech anti-COVID-19 mRNA vaccination. An abdominal dynamic computed tomography angiography showed stenosis and dilatation of multiple visceral arteries, including the middle and right colic arteries. In the omental arteries, spindle-shaped dilatation and stenosis were identified. The left epiploic artery was not visualized, suggesting the development of occlusion due to arterial dissection. Based on these findings, SAM of multiple visceral arteries was diagnosed. Because the patient’s vital condition was stable, treatment by observation, with restriction of daily living, was chosen. Seventy-five days later, the pathological lesions in the affected vessels spontaneously resolved.
CONCLUSIONS: While coincidence could not be completely excluded in this case, anti-COVID-19 mRNA vaccination should be noted for its potential association with SAM as a possible late complication.
Keywords: case reports, computed tomography angiography, COVID-19 vaccine, Vascular Diseases, Aged, COVID-19, Constriction, Pathologic, Dilatation, Pathologic, Humans, Male, Mesenteric Artery, Superior, RNA, Messenger, SARS-CoV-2, Vaccination
Background
Segmental arterial mediolysis (SAM) is a rare noninflammatory, nonatherosclerotic vascular disorder characterized by arterial media disruption [1–4]. The most common imaging findings via computed tomography (CT) include dissection, aneurysm, beading, occlusion, and arterial wall thickening, which can cause ischemia/infarction of the organs involved and sometimes a life-threatening hemorrhage [1–4].
Frequently involved sites of SAM are the visceral and renal arteries, while the coronary and cerebral arteries could be involved to a lesser extent [1–4]. SAM requires surgical or endovascular management if the condition worsens due to rupture; otherwise, conservative management with or without medical therapy could be a feasible option [1–4].
The outbreak of COVID-19, which is caused by SARS-CoV-2, began in December 2019 in Wuhan, Hubei, China, and then, it became a pandemic [5]. Among the morbidities associated with COVID-19, vascular complications, including vasculopathy, thrombotic microangiopathy, and intravascular coagulopathy, are of increasing concern because devastating vital organ injuries could be induced at various stages of the disease progression [5,6].
With the advent of promising anti-COVID-19 vaccines, the SARS-CoV-2 pandemic was expected to quickly subside [6]. In line with this concept, the national vaccination program in Japan started on February 21, 2021, mainly by using the approved messenger RNA (mRNA) Comirnaty concentrate for dispersion of injection (Pfizer-BioNTech: Mainz, Germany). However, the rapid development and application of the novel mRNA vaccines come with a setback because studies on long-term unpredictable adverse effects remain insufficient [5]. Furthermore, reports on the potential risk of developing late adverse effects, including visceral vascular disorders, after receiving vaccination remain limited, particularly for the anti-COVID-19 mRNA vaccine [6–8].
Here, a case of SAM after the third dose of Pfizer-BioNTech anti-COVID-19 mRNA vaccination is presented as a potential late complication or rare coincidence.
Case Report
This is the case of a 68-year-old male gynecologic surgeon working at a regional tertiary referral center in a suburban city in Japan. Previous disease history includes gastric ulcer, which was treated with
The patient received the first dose of intramuscular BNT 162b2 anti-COVID-19 mRNA vaccine (EP2163, Pfizer-BioNTech) on March 9, 2021. He then experienced mild symptoms of soreness and redness at the injection site and myalgia lasting for more than 24 h. He had a fever of approximately 38°C, which was managed through the oral intake of acetaminophen. On March 30, 2021, a second intramuscular injection of the anti-COVID-19 mRNA vaccine (EP2163, Pfizer-BioNTech) was administered. This time, the fever was higher than the first, and frequent intake of acetaminophen was necessary. On December 10, 2021, a third dose of the anti-COVID-19 mRNA vaccine (FJ5929, Pfizer-BioNTech) was administered intramuscularly. On that occasion, symptoms including swelling and redness at the injection site and fever were milder than those on the previous 2 occasions, and were followed by an uneventful recovery.
Fifty-eight days after the third injection, discomfort from the upper abdomen to the right side of the abdomen was felt. Because the pain worsened and nausea emerged, the patient visited the Emergency Department. The systemic and neurological examinations were unremarkable except for persistent epigastric pain. The patient had stable vital signs.
An abdominal dynamic CT angiography showed stenosis and dilatation of multiple visceral arteries, including the middle and right colic arteries (Figure 1A, green circle). In the omental arteries, spindle-shaped dilatation and stenosis were identified (Figure 1A, red circle). The left epiploic artery was not visualized, suggesting the development of occlusion due to arterial dissection (Figure 1A, arrow).
Blood counts and the metabolic panel at the patient’s presentation showed no remarkable findings except for a slight elevation of C-reactive protein (CRP) (Table 1). The GeneXpert Xpress SARS-CoV-2 polymerase chain reaction assay (Beckman Coulter, Tokyo, Japan) yielded a negative result for SARS-CoV-2 infection (Table 1). A subsequent examination of vasculitis, thrombosis, and collagen disease screen showed no significant abnormal findings except for some minor changes (Table 2). Further genetic diagnosis for collagen vascular disorders was not performed because no physical or laboratory findings indicated such a background. On magnetic resonance imaging angiography of the brain, abnormal findings of the blood vessels were unidentified.
Based on these findings, we diagnosed SAM of the multiple visceral arteries [9]. Because the patient’s vital condition was stable, treatment by observation with restriction of daily living was chosen after the patient denied the recommendation to admit for observation. Then, the abdominal discomfort gradually lessened, without secondary attack.
On follow-up CT imaging, 21 days later, spontaneous resolution of dilatation in the affected vessels was observed. Fifty-two days after symptom onset, a spindle-shaped aneurysmal structure was observed in the left gastric artery (Figure 1B, arrow). From this point on, the restrictions on daily activities were stopped. Seventy-five days after symptom onset, aneurysmal dilatation of the left gastric artery spontaneously subsided (Figure 1C, arrow). Subsequently, the disease course was uneventful, and observation was discontinued.
Discussion
SAM is a rare vascular disorder resulting from degeneration of the arterial media; its exact developmental mechanism is unknown [1–4]. Serological examination does not usually show elevated inflammatory markers, including CRP and erythrocyte sedimentation rate [1]. However, acute dissection or intra-abdominal hemorrhage in cases of SAM can cause a temporary increase in inflammatory markers, which would normalize quickly [1]. In the present case, it was assumed that a slight CRP value elevation at triage could have been due to acute dissection and subsequent occlusion of the left epiploic artery.
In a surgically obtained SAM specimen [1], the characteristic pathological findings of the affected vessel include lysis and vacuolization of the outer media originating from the arterial gaps and patchy transmural loss of the external elastic lamina, followed by separation at the adventitial medial junction. Then, it is speculated that these changes can form aneurysms and dissecting hematomas due to weakening of the arterial wall, as was observed in the present case.
With the recent accumulation of conservatively managed cases without surgical excision [1], the findings on CT imaging to characterize SAM are now becoming more important [9]. They include arterial dilation and stenosis showing a “beaded” appearance, dissecting hematoma, pseudoaneurysm, single or multiple aneurysms, end-organ ischemia or infarct, and rarely arteriovenous fistula [9], some of which were observed in the present case.
Because of the low recognition and characterization of SAM, its true incidence has not yet been clearly demonstrated [1]. SAM affects middle-aged and elderly patients, with a median age at onset of 55 years, and tends to be slightly more common in men. Abdominal pain, as observed in the present case, accounts for approximately 10% of presenting symptoms of SAM in the emergency department [1].
However, SAM presents diagnostic and therapeutic challenges because various infectious, inflammatory, neoplastic, obstructive, and idiopathic disorders must be excluded. Among vascular disorders, the principal differential diagnoses of SAM, fibromuscular dysplasia, which usually lacks symptoms, is more common in young patients and predominantly involves the renal arteries and polyarteritis nodosa [1].
Although new antivirals, including nirmatrelvir plus ritonavir (Paxlovid; Pfizer, New York, NY, USA), have been approved now for COVID-19 infection, it is still believed that the best strategy to terminate the pandemic depends on timely and appropriate immunizations worldwide [5]. Because the efficacy and safety of anti-COVID-19 vaccines were shown in the registration trials, their benefits are believed to outweigh the potential risk of complications [5,6].
Nevertheless, the anti-COVID-19 mRNA vaccine was developed faster than any vaccine in the past (thanks to the collaboration between researchers, scientists, manufacturers, and distributors in a never-before-seen approach), and it should be cautioned that immunization with limited experiences such as this can induce unexpected de novo vaccine-related disorders, including autoimmune diseases, particularly in predisposed individuals [5,6]. Furthermore, in addition to complications with a relatively clear causal relationship occurring in the early period after vaccination, associated morbidities manifesting in later periods with less unclear causal relationships should be carefully monitored.
In conjunction with the SARS-CoV-2 infection or anti-COVID-19 vaccination, vascular disorders have been recognized as organ-specific immune-mediated complications, and the number of reported cases is increasing [5,6].
A recent report has shown that spontaneous dissection of visceral arteries without aortic involvement, which shows a similar clinical presentation to SAM, has occurred after anti-COVID-19 mRNA vaccination (Table 3) [7]. Furthermore, spontaneous pseudoaneurysm of the visceral artery with rupture after anti-COVID-19 mRNA vaccination has been presented (Table 3) [8]. In these cases, the potential association of vascular disorders with anti-COVID-19 mRNA has been discussed, although rare coincidences are undeniable [7,8].
Vascular complications can occur early after the onset of COVID-19 or anti-COVID-19 vaccination, at less than 2 weeks apart [7,8]. Alternatively, the complications can manifest later during the course of the disease or vaccination, as in the present case, and can be associated with significant morbidity. Although the determination of the causality or coincidence between a COVID-19 infection or vaccination and vascular disorders is extremely difficult to ascertain, it has been suggested that both possibilities should be included as potential risk factors causing or promoting systemic vascular disorders [5,6].
The developmental mechanisms of COVID-19-related vascular disorders remain unelucidated [5,6]. It has been shown that SARS-CoV-2 binds to the angiotensin-converting enzyme 2 receptors expressed on endothelial cells of the arterial wall by using surface spike proteins. Despite the controversy about whether this binding of the spike protein can lead to direct viral infection of the endothelial cells, the binding can eventually result in the dysfunction of the endothelial cells through damage. Furthermore, a suggestion has been made that SARS-CoV-2 infection can also induce giant cell arteritis [10].
Because all these mechanisms can also play a similar role in the development of anti-COVID-19 mRNA vaccine-associated vascular disorders [6], it could be wondered whether the anti-COVID-19 mRNA vaccine, which allows for the endogenous production of the viral spike protein, has induced any type of arterial wall injuries. Alternatively, it could be hypothesized that bystander hyperactivation of the immune surveillance system, molecular mimicry, neoantigen production, and disturbance of immunological tolerance play a substantial role in triggering and developing vascular disorders [6].
As an analogy to this potential mechanism of anti-COVID-19 mRNA vaccine-associated vascular disorders [8], possible mechanisms accounting for SAM after anti-COVID-19 mRNA vaccination could be postulated. Given the rare manifestation of SAM and its chronological relationship to the third dose of anti-COVID-19 mRNA vaccination in the present case, we still wonder whether SAM or mimics might have been related to the anti-COVID-19 mRNA vaccination in our patient’s case. However, to date, such reported cases are limited in literature (Table 3).
It is understood that there is the possibility that the patient in the present case may have had a previously undiagnosed SAM by chance, which subsequently resulted in the eventual formation of arterial dissection of the visceral arteries followed by occlusion by coincidence, without an unknown trigger at present.
Current information on the risk of vascular disorders due to anti-COVID-19 mRNA vaccination remains controversial. Thus, our experience concerning the development of SAM after anti-COVID-19 mRNA vaccination was presented in this case report. In fact, it is not our intention to deny the benefit of mass anti-COVID-19 vaccinations in preventing potential morbidity and mortality due to COVID-19.
We hope that this case of the relationship between anti-COVID-19 vaccination and SAM will add to the literature on the various potential manifestations and complications either in patients with COVID-19 or in patients who have received anti-COVID-19 mRNA vaccines.
Conclusions
SAM is a potentially life-threatening rare condition characterized by the disruption of visceral arterial media. While it could all be a rare coincidence, reporting such adverse events regardless of any potential causality will help add to the body of literature that is important for phase IV post-market surveillance to monitor for any late adverse complications related to anti-COVID-19 vaccines.
Tables
Table 1.. Blood counts, metabolic panel, and GeneXpert Xpress SARS-CoV-2 polymerase chain reaction assay at presentation. Table 2.. Vasculitis, thrombosis, and collagen disease screen.
Table 2.. Vasculitis, thrombosis, and collagen disease screen.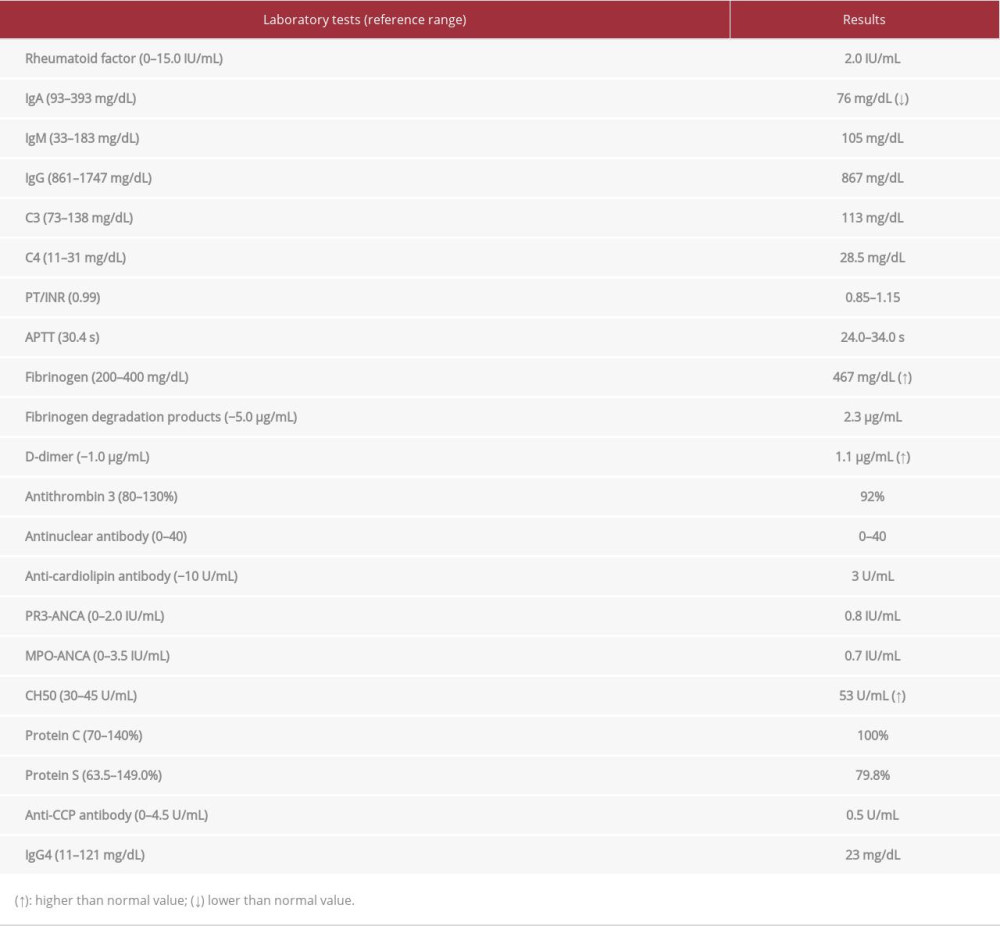 Table 3.. The reported cases of visceral arterial lesions suspected to be related to anti-COVID-19 mRNA vaccination.
Table 3.. The reported cases of visceral arterial lesions suspected to be related to anti-COVID-19 mRNA vaccination.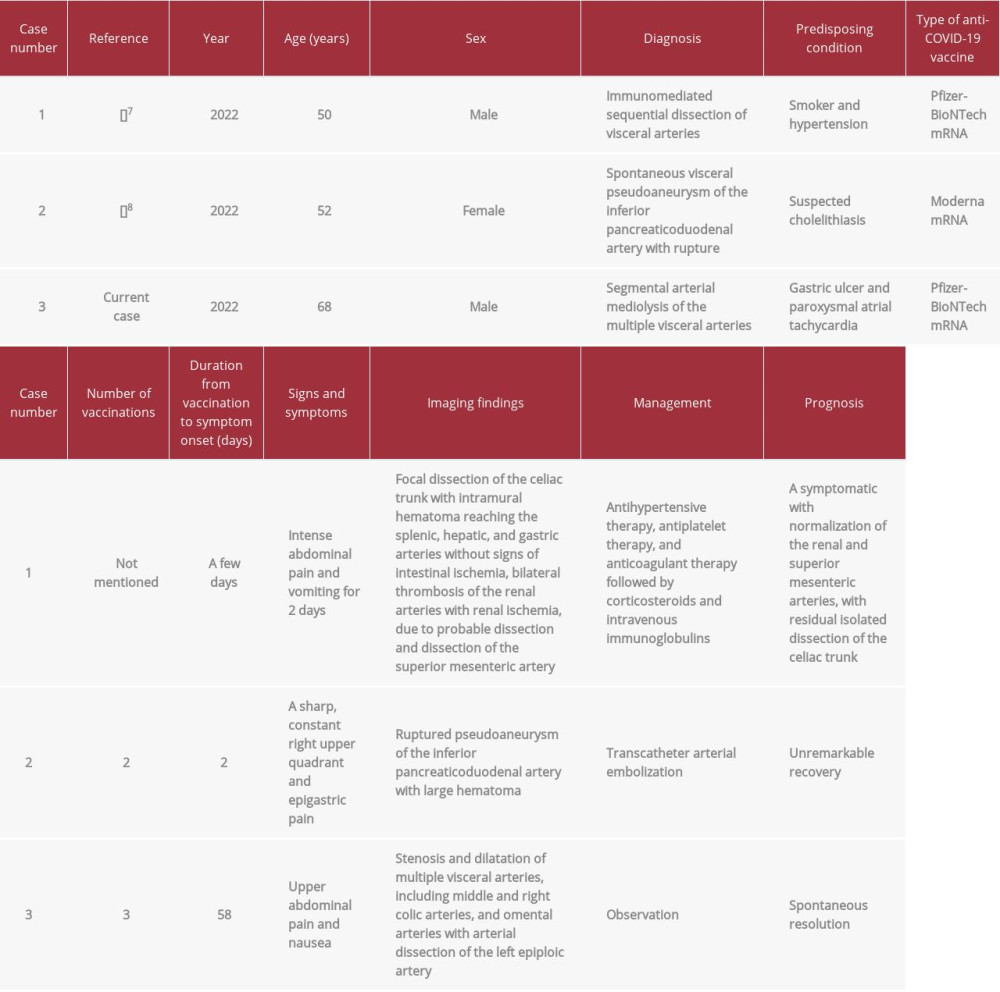
References:
1.. Peng KX, Davila VJ, Stone WM, Natural history and management outcomes of segmental arterial mediolysis: J Vasc Surg, 2019; 70(6); 1877-86
2.. Chatterjee T, Stephens J, Roy M, Segmental arterial mediolysis: An under-recognized cause of chronic abdominal pain: Eur J Case Rep Intern Med, 2020; 7(10); 001830
3.. Kennedy CA, Toomey DP, Segmental arterial mediolysis: A rare cause of an acute abdomen: J Surg Case Rep, 2021; 2021(10); rjab370
4.. Borde P, Vilgrain V, Segmental arterial mediolysis: Radiology, 2022; 302(3); 515
5.. Humayun L, Smith C, Li W, SARS-CoV-2-related vascular injury: Mechanisms, imaging and models: Microphysiol Syst, 2021; 5; 1
6.. Hočevar A, Simonović Z, Rotar Ž, Tomšič M, Vasculitis as temporally associated with COVID-19 infection or vaccination: A single-center experience: J Rheumatol, 2022; 49(2); 232-33
7.. Pina A, Farina A, Savonitto S, Immunomediated sequential dissection of visceral arteries: Dramatic improvement after steroid therapy: Am J Med, 2022; 135(6); e130-31
8.. Chue KM, Tok NWK, Gao Y, Spontaneous rare visceral pseudoaneurysm presenting with rupture after COVID-19 vaccination: ANZ J Surg, 2022; 92(4); 915-17
9.. Michael M, Widmer U, Wildermuth S, Segmental arterial mediolysis: CTA findings at presentation and follow-up: Am J Roentgenol, 2006; 187(6); 1463-69
10.. Mursi AM, Mirghani HO, Elbeialy AA, A case report of post COVID19 giant cell arteritis and polymyalgia rheumatica with visual loss: Clin Med Insights Case Rep, 2022; 15; 11795476221088472
Tables
 Table 1.. Blood counts, metabolic panel, and GeneXpert Xpress SARS-CoV-2 polymerase chain reaction assay at presentation.
Table 1.. Blood counts, metabolic panel, and GeneXpert Xpress SARS-CoV-2 polymerase chain reaction assay at presentation. Table 2.. Vasculitis, thrombosis, and collagen disease screen.
Table 2.. Vasculitis, thrombosis, and collagen disease screen. Table 3.. The reported cases of visceral arterial lesions suspected to be related to anti-COVID-19 mRNA vaccination.
Table 3.. The reported cases of visceral arterial lesions suspected to be related to anti-COVID-19 mRNA vaccination. Table 1.. Blood counts, metabolic panel, and GeneXpert Xpress SARS-CoV-2 polymerase chain reaction assay at presentation.
Table 1.. Blood counts, metabolic panel, and GeneXpert Xpress SARS-CoV-2 polymerase chain reaction assay at presentation. Table 2.. Vasculitis, thrombosis, and collagen disease screen.
Table 2.. Vasculitis, thrombosis, and collagen disease screen. Table 3.. The reported cases of visceral arterial lesions suspected to be related to anti-COVID-19 mRNA vaccination.
Table 3.. The reported cases of visceral arterial lesions suspected to be related to anti-COVID-19 mRNA vaccination. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








