08 February 2023: Articles 
Cannabis-Associated Myocardial Infarction with Non-Obstructive Coronary Arteries in a Young Patient with Underlying Myocardial Bridge
Unusual clinical course, Challenging differential diagnosis, Management of emergency care
Mahlatse MankgeleDOI: 10.12659/AJCR.938175
Am J Case Rep 2023; 24:e938175
Abstract
BACKGROUND: Myocardial infarction (MI) is one of the most ominous medical emergencies because it carries significant morbidity and mortality. A myocardial bridge is an anomaly previously perceived as benign. However, with a better understanding, it is considered a risk factor for angina and MI in some cases. Certain precipitating factors potentiate the coronary artery within the myocardial bridge to having vasospasms. Cannabis is one of many potential precipitants for vasospasm in the setting of a myocardial bridge because it increases vascular tone and increases sympathetic hormone secretion in the form of noradrenaline.
CASE REPORT: We report a case of a 31-year-old man presenting with myocardial infarction, without any known traditional risk factors for cardiovascular disease. Upon investigation, we discovered that he had an underlying myocardial bridge, which in the setting of significant cannabis consumption, precipitated myocardial infarction with non-obstructive coronary arteries (MINOCA) disease on invasive angiography.
CONCLUSIONS: In a setting with an underlying risk factor or cardiac anomaly, cannabis can induce an MI and potentially other adverse cardiac complications. With the increasing use of cannabis in several regions of the world, more adverse events outside of the well-documented psychotropic effects of cannabis are anticipated. Therefore, clinicians need to bear in mind the multifaceted effects of adverse events from cannabis in the various organ systems and be prepared to ensure prompt treatment as needed. Given the nature of the pathophysiology of MINOCA in a patient with a myocardial bridge, it is paramount to be aware that the cause of infarction is a reversible one.
Keywords: Cannabis, Coronary Artery Disease, Myocardial bridging, Non-ST Elevated Myocardial Infarction, Male, Humans, Adult, MINOCA, Coronary Vessels, Coronary Angiography, Risk Factors, Vascular Diseases
Background
The entity termed myocardial infarction with non-obstructive coronary arteries (MINOCA) has variable pathophysiological mechanisms termed endotypes [1,2]. The prevalence of MINOCA varies geographically, ranging between 1% to 15% in patients with myocardial infarction (MI) [3]. MINOCA and obstructive coronary artery disease from atherosclerosis decreases blood supply to the myocardium, leading to angina [1,2].
MINOCA endotypes include microvascular angina and epicardial vasospastic angina [2]. A myocardial bridge is one of the causes of MINOCA classified under the epicardial vasospastic angina category [4]. A myocardial bridge forms when part of the coronary vessel traverses within the myocardium at varying depths and lengths instead of at the usual epicardial location [5]. The myocardial muscle overlying the coronary vessel is termed a myocardial bridge [5]. Previously deemed benign, it has now been shown to play a role in vasospastic infarction in the setting of certain precipitants, especially those that increase heart rate and vascular tone [4].
The use of cannabinoids is on the rise in various geographical regions, in medically controlled settings and recreationally [6]. The anticipated increase is due to newly approved legislations that enable and decriminalize the use of cannabis [7]. Furthermore, there has been an increase in the concentration of cannabinoid-active compounds in cannabis products [7].
There are growing concerns about the effects of cannabis outside the neurological system, specifically the cardiovascular system [7]. These include MI, arrhythmias, cardiomyopathies, and strokes [6].
Case Report
The patient was a 31-year-old previously healthy man. He was originally from Nigeria and had been residing in South Africa for the past 10 years, with no recent travel history. He had 7 siblings, who were all healthy. There was no family history of cardiovascular disease or sudden cardiac-related death.
The patient notably smoked marijuana recreationally. He smoked 5 to 10
He presented with intermittent chest pain of a 2-week duration. The pain was central and crushing (typical ischemic chest pain). He had no aggravating factors and had reported the pain to be spontaneous at times. He did not report any flu-like symptoms in the preceding weeks.
On clinical examination, he had no peripheral edema, elevated jugular venous pressure, S3 gallop rhythm, or bilateral basal crackles noted. His pain did not change in character with positions like leaning forward, and he had no pericardial friction rub. The rest of his physical examination was unremarkable. He had no documented fever, features of sepsis, or thyrotoxicosis. He had no suggestive features of a pulmonary embolism. He had a lean body phenotype.
He initially presented to a secondary level hospital with severe typical chest pain, graded 9/10. His admission electrocardiogram (ECG) had features of benign early repolarization with minimal ST-segment elevation in leads II and III, V2-V6 of 1 mm with no reciprocal changes, and PR segment elevation in lead aVR (Figure 1). However, upon admission, his high-sensitivity troponin-T was 551 ng/L (rule-in for acute coronary syndrome >100 ng/L). Therefore, a working diagnosis of a non-ST elevation MI (NSTEMI) was made. He also had a modestly raised C-reactive protein level, which was attributed to his myocar-dial necrosis [8].
He was subsequently initiated on the acute coronary syndrome management protocol, which included aspirin, clopidogrel, atorvastatin, and carvedilol.
He was transferred to the Cardiology Department in a tertiary academic hospital within 12 h after his index episode of chest pain. On admission to the Coronary Care Unit, his chest pain was graded 5/10. His COVID-19 PCR was negative on admission. At this point, his repeat ECG remained unchanged compared to the one from the first medical contact.
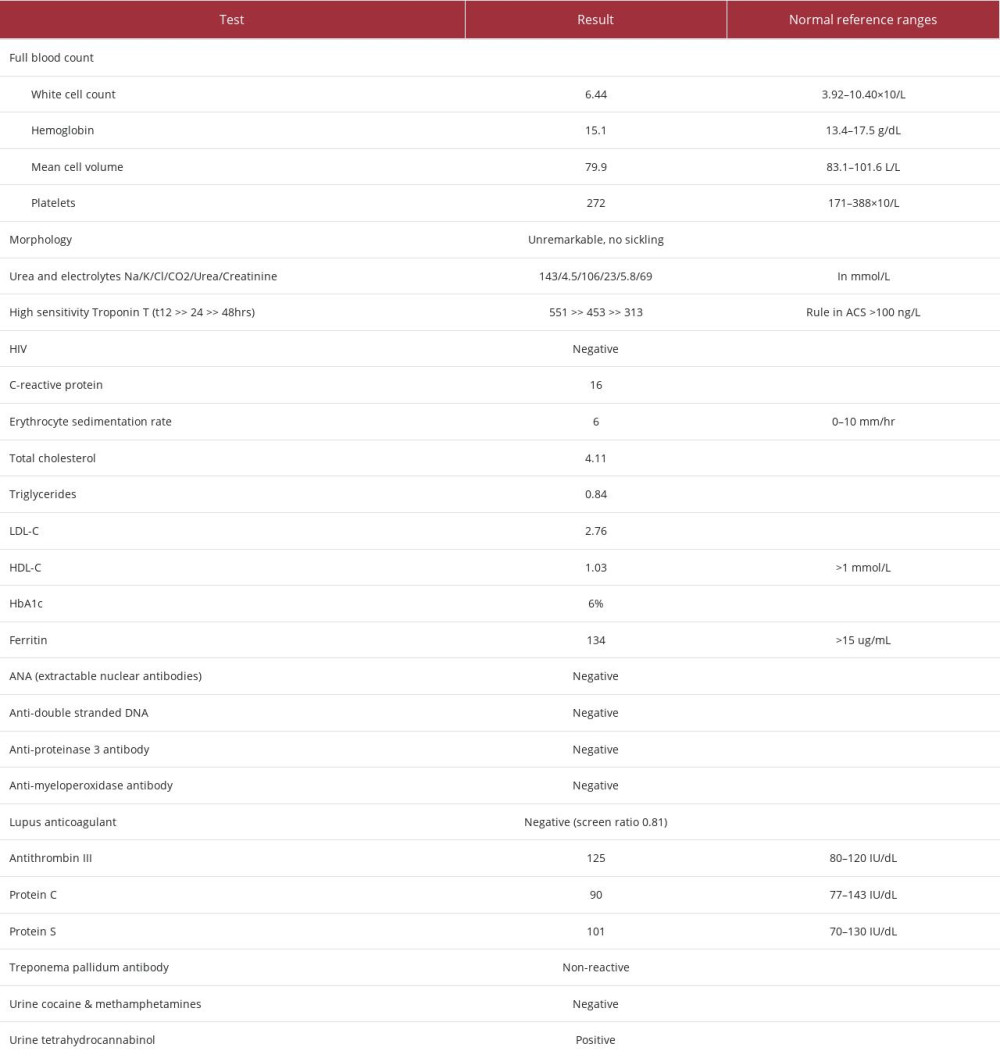
His laboratory biochemical investigations revealed that he was negative for HIV. In addition, his triglycerides and low-density lipoprotein cholesterol levels were not elevated, and he did not have diabetes. The rest of the biochemical findings are reported in Table 1. Of note, his urine drug screen was positive for only cannabis and negative for other recreational drugs, particularly amphetamines (Table 1).
His echocardiogram was within normal limits, without regional wall motion abnormalities or pericardial fluid. He had an ejection fraction of 55% to 60%.
A diagnostic coronary angiogram was performed at 12 h. The angiogram revealed no occlusive atherosclerotic lesions. However, a myocardial bridge was noted over the mid-left anterior descending coronary artery (Figures 2, 3).
One day following the diagnostic coronary angiogram, the patient underwent a modified Bruce protocol exercise stress test, during which he reported no chest pain, and no ischemic changes were noted on the ECG. Furthermore, a 24-h Holter ECG was performed and did not identify any significant rhythm abnormalities.
A diagnosis of a type 2 MI was made; the myocardial bridge was postulated as the etiology of his MINOCA, precipitated by a cannabis-induced increase in heart rate and vascular tone. Differential diagnoses of pericarditis and myocarditis were taken into consideration. The absence of a pericardial friction rub was not in keeping with pericarditis, particularly on leaning forward. However, the patient did have a mild elevation of his C-reactive protein level, which could also be accounted for by myocardial death/ necrosis in an MI. Pericarditis can also be a complication of MI.
The patient was counseled and advised on cannabis use cessation. He was subsequently discharged from the hospital on verapamil 40 mg 3 times a day and nitroglycerin as needed. At follow-up 3 months after admission and cannabis use cessation, he remained free of chest pain.
Discussion
In the absence of traditional risk factors for MI, other plausible alternative etiologies must be sought to prevent or anticipate any other cardiovascular events and most importantly, treat them accordingly [1].
Our patient had an underlying predisposition for his presentation with MINOCA, namely the underlying myocardial bridge. The noted myocardial bridge pointed out in the mid-left anterior descending coronary artery is the most common location for this anomaly [4]. As previously mentioned, this congenital anomaly is not necessarily benign [4]. It can lead to perfusion abnormalities and chest pain syndromes in specific clinical settings [5].
A myocardial bridge is present in roughly one-third of the adult population [5]. Myocardial bridges have been commonly found more in male individuals who do not have diabetes or elevated body mass indices [6,7], which fits our patient’s demographics and phenotype. The prevalence of a myocardial bridge can vary depending on the method of diagnostic imaging used [5]. The vulnerability toward MI and sudden cardiac death has been described [4]. In a study of individuals with a myocardial bridge, Teragawa et al indicated that the coronary vasospasm is usually noted on provocation, meaning there is a need for other risk factors to come into play in eliciting the vasospasm [4].
Cannabis denotes the generic term for the wide range of formulations of the cannabis plant, which contains cannabinoids [6]. Cannabinoids are a vast group of synthetic and naturally occurring compounds [6]. Humans are responsive to cannabinoids via the endocannabinoid system of mainly 2 receptors [9]. Cannabinoid receptor 1 is mainly isolated in nervous tissues, smooth muscle, gastrointestinal tract, and cardiac muscle [10]. Cannabinoid receptor 2 is found mainly in hemopoietic cells, immune cells, and cardiac myocytes [10].
Several case reports have defined severe cardiovascular complications, including acute coronary syndromes and strokes in cannabis users [11]. Three main mechanisms are suggested as pathophysiological effects of cannabis-induced major cardiovascular events, namely hemodynamic changes via catecholaminergic drive, vasospasm induction, and possible thromboembolic occlusive disease [10].
A prospective study of 1913 adults, followed up over 3.8 years, demonstrated a dose-response relationship between cannabis use and cardiovascular mortality [12]. Furthermore, the World Health Organization has highlighted specific case reports and case series signaling an increased risk of cardiovascular disease due to cannabis use in younger, otherwise relatively low-risk individuals [6]. However, isolating and identifying specific adverse effects can be challenging owing to the high incidence of concomitant drug use with cannabis [6].
In a myocardial bridge, the most significant compression of the coronary vessel occurs during systole when the myocardial bridge over the coronary vessel constricts flow [5]. Following the narrowing, relaxation of the artery can be delayed during early diastole when myocardial perfusion ensues [5]. This delay worsens specifically with states of increased vascular tone (adrenergic states) [1,5]. Cannabis, explicitly increasing adrenergic hormone secretion, is a potential cause of type 2 MI, namely myocardial supply-demand mismatch in some MINOCA endotypes [1,10].
In our case study, we hypothesize that the negative exercise stress test results suggest that there was a precipitant of the ischemic event that was not present at the time of the stress test, that is, the cannabis. Furthermore, we propose that with his recently increased consumption of cannabis, there was a potential elevation in the adrenergic tone leading to a type 2 MI, demonstrating the dose-effect relationship described by Mukamal et al [12].
Limitations in this clinical scenario arose from our inability to further define the patient’s coronary vasculature’s luminal anatomy with optical coherence tomography or intravascular ultrasound to exclude an atherosclerotic plaque. Also, we could not perform vasoreactivity testing to elicit the vaso-spasm and precipitate angina. We also have limited access to cardiac magnetic resonance imaging and limited expertise to carry out this investigation in our local setting.
Conclusions
With the reported increasing use and legalization of cannabis in some regions of the world, we anticipate more adverse cardiovascular events outside of the well-documented psychotropic effects. Therefore, clinicians treating cannabis users need to be aware of the multifaceted adverse effects of cannabis use and the implications of cannabis use on management.
Figures
References:
1.. Kunadian V, Chieffo A, Camici PG, An EAPCI Expert Consensus Document on Ischaemia with Non-Obstructive Coronary Arteries in Collaboration with European Society of Cardiology Working Group on Coronary Pathophysiology & Microcirculation Endorsed by Coronary Vasomotor Disorders International Study Group: Eur Heart J, 2020; 41(37); 3504-20
2.. Mukherjee D, Myocardial infarction with nonobstructive coronary arteries: A call for individualized treatment: JAMA, 2019; 8(14); e013361
3.. Abdu FA, Mohammed AQ, Liu L, Xu Y, Che W, Myocardial infarction with nonobstructive coronary arteries (MINOCA): A review of the current position: Cardiology, 2020; 145(9); 543-52
4.. Teragawa H, Fukuda Y, Matsuda K, Myocardial bridging increases the risk of coronary spasm: Clin Cardiol, 2003; 26(8); 377-83
5.. Sternheim D, Power DA, Samtani R, Myocardial bridging: Diagnosis, functional assessment, and management: JACC state-of-the-art review: J Am Coll Cardiol, 2021; 78(22); 2196-212
6.. : The health and social effects of nonmedical cannabis use, 2016, Geneva, WHO Available at: https://www.who.int/publications/i/item/9789241510240
7.. Chetty K, Lavoie A, Deghani P, A literature review of cannabis and myocardial infarction-what clinicians may not be aware of: CJC Open, 2021; 3(1); 12-21
8.. Dedobbeleer C, Melot C, Renard M, C-reactive protein increase in acute myocardial infarction: Acta Cardiol, 2004; 59(3); 291-96
9.. Mittleman MA, Lewis RA, Maclure M, Triggering myocardial infarction by marijuana: Circulation, 2001; 103(23); 2805-9
10.. Latif ZGN, The impact of marijuana on the cardiovascular system: A review of the most common cardiovascular events associated with marijuana use: J Clin Med, 2020; 9(6); 1925
11.. Jouanjus E, Lapeyre-Mestre M, Micallef J, Cannabis use: Signal of increasing risk of serious cardiovascular disorders: J Am Heart Assoc, 2014; 3(2); e000638
12.. Mukamal KJ, Maclure M, Muller JE, Mittleman MA, An exploratory prospective study of marijuana use and mortality following acute myocardial infarction: Am Heart J, 2008; 155(3); 465-70
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133