13 March 2023: Articles 
Successful Surgical Management of Gastric Antral Vascular Ectasia in a Patient with End-Stage Renal Disease: A Case Report and Literature Review
Unknown etiology, Management of emergency care, Rare disease, Educational Purpose (only if useful for a systematic review or synthesis), Rare coexistence of disease or pathology
Bader H. Alsaeed1BDE, Ayesha A. AlAbdulqader1BDE, Ali A. Al-QadhiDOI: 10.12659/AJCR.938543
Am J Case Rep 2023; 24:e938543
Abstract
BACKGROUND: Gastric antral vascular ectasia (GAVE) is a rare clinical entity that presents with acute upper-gastrointestinal bleeding or chronic anemia. It is characterized by endoscopic watermelon appearance of the stomach. It is usually associated with other comorbidities; however, few articles have previously described GAVE in patients with end-stage renal disease. Its management is controversial, and endoscopic management is considered the treatment of choice.
CASE REPORT: A middle-age female patient, on regular hemodialysis for ESRD, was referred to the surgical out-patient clinic as a refractory GAVE after failure of endoscopic management as she became blood transfusion-dependent. She underwent laparoscopic subtotal gastrectomy with a Billroth II reconstruction of gastrojejunostomy. She had a smooth postoperative course and was followed up in the clinic for 12 months with no complications. Her hemoglobin level was stable at 9.4 g/dL without further blood transfusion.
CONCLUSIONS: Gastric antral vascular ectasia is usually associated with other comorbidities; however, an association between GAVE and CKD is rare. Its management is controversial, and endoscopic management is considered the preferred method of treatment. Laparoscopic subtotal gastrectomy is an effective management modality for GAVE, with dramatic improvement and good outcomes in terms of bleeding, blood transfusion requirements, and nutritional status.
Keywords: Blood Transfusion, Gastric Antral Vascular Ectasia, Gastric Bypass, Gastrointestinal Hemorrhage, Renal Insufficiency, Chronic, Middle Aged, Humans, Female, Kidney Failure, Chronic, Anemia, Renal Dialysis
Background
Gastric antral vascular ectasia (GAVE) is a rare clinical entity accounting for 4% of non-variceal upper-gastrointestinal bleeding. It is characterized by endoscopic watermelon appearance of the stomach [1–3]. Typically, it is presented by upper-gastrointestinal bleeding leading to chronic anemia [4,5]. GAVE is usually associated with various comorbidities. However, few articles have previously described GAVE in patients with end-stage renal disease (ESRD).
Herein, we describe a successful surgical management of a middle-aged woman on regular hemodialysis for ESRD who became a blood transfusion-dependent and was found to have GAVE.
Case Report
A 54-year-old female patient was referred to the upper-gastrointestinal surgery clinic as a case of GAVE refractory to endoscopic management. She was known to have ESRD, diabetes mellitus, and hypertension. She had been on hemodialysis for 6 years. A year prior to presentation, she became blood transfusion-dependent with a baseline hemoglobin level of 4.5–6.1 g/dL, receiving 1 unit of packed RBCs during each dialysis session.
She underwent esophagogastroduodenoscopy and colonos-copy, which revealed a typical watermelon appearance of the stomach, with prominent erythematous folds in the antrum, features suggestive of GAVE (Figure 1). She was managed by endoscopic argon plasma coagulation (APC) that was repeated 3 times, with no noticeable clinical or endoscopic improvement.
Upon presentation to the surgical clinic, she was pale and undernourished, with bilateral lower-limb edema. Laboratory test results were significant for normocytic normochromic anemia and positive occult blood in the stool. Renal function tests were typical for ESRD. Other lab results, including liver function test and coagulation profile, were unremarkable.
After thorough counseling, she underwent a laparoscopic subtotal gastrectomy with a Billroth II reconstruction of gastrojejunostomy. Under general anesthesia, she was placed in a supine anti-Trendelenburg position. Diagnostic laparoscopy showed small amounts of ascites, with a huge fatty liver. The stomach was dissected from the mid-body downward to the pylorus using a sealing device, starting with the greater omentum followed by the lesser omentum. The stomach was transected 1 cm proximal to the pylorus and at the level of the mid-body. Gastrojejunostomy was then performed 60 cm distal to the duodeno-jejunal junction. The blood loss was minimal (around 50 milliliters). The total procedure duration was 115 minutes.
The patient had a smooth postoperative course. A water-soluble study on the third postoperative day excluded leakage. Gradually, she started oral intake and was eventually discharged home on the seventh postoperative day in good condition. She was followed up in the clinic for 1 year with no concerns and good nutritional status. Her hemoglobin level was stable at 9.4 g/dL without further blood transfusions. Her albumin level increased from 1.9 g/dL preoperatively to 3.7 g/dL 1 year postoperatively.
Discussion
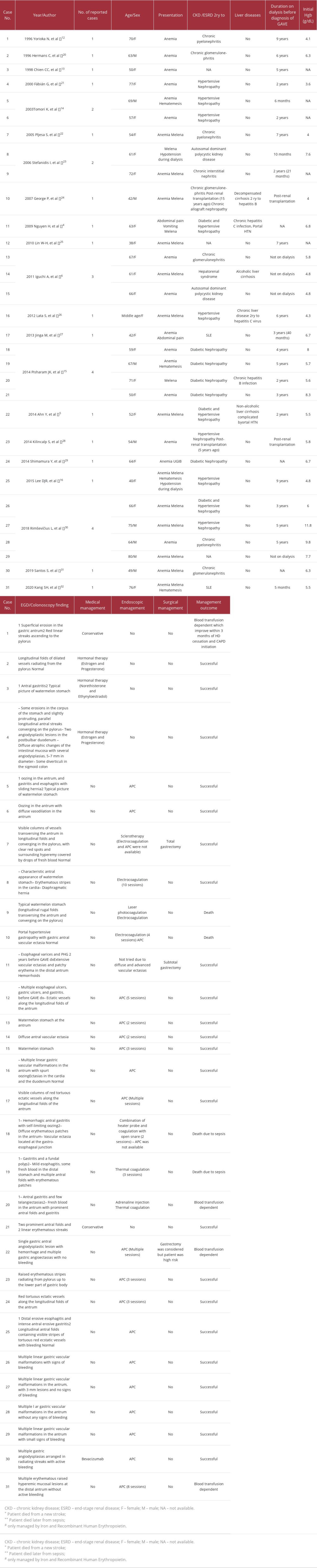
GAVE is a rare clinical entity with unknown etiology [2,3]. GAVE is usually associated with various systemic diseases, especially autoimmune diseases. Around 30% of patients with GAVE have liver cirrhosis [2]. Other associated systemic diseases include Sjogren’s syndrome, systemic lupus syndrome, systemic sclerosis, Raynaud’s disease, DM, hypertension, acute myeloid leukemia, and bone marrow transplant [6–9]. Rarely, GAVE has been reported in patients with chronic kidney diseases. We performed a literature review using the terms “gastric antral vascular ectasia,” “GAVE,” “watermelon stomach,” “ESRD,” “chronic kidney disease,” “uremia,” and “hemodialysis” in databases of PubMed, Scopus, Medline, and Google scholar and identified 31 cases of GAVE in patients with chronic kidney diseases (Table 1).
GAVE is a disease of elderly people, with a female predominance in hepatic patients and male predominance in all other patients. This can be explained by the age and sex distribution of associated comorbidities [10].
Diagnosis of GAVE depends mainly on its characteristic endoscopic features: parallel red stripes of tortuous ectatic vessels, usually located at the antral mucosal folds [11]. Moreover, it may extensively involve the whole stomach, giving a picture of honeycomb stomach, especially in cirrhotic patients [2,10]. However, the absence of the characteristic watermelon appearance does not entirely exclude the presence of GAVE, as the literature review shows that many patients required more than 1 endoscopy to confirm the presence of GAVE. Moreover, 7 cases (22.5%) in the literature review did not report this finding and were diagnosed based on clinical picture [12–16].
Management of GAVE is controversial, including various medical, endoscopic, and surgical management modalities. Several drugs have been tried in such patients, including estrogen and progesterone analogs, octreotide, and pulse steroids [17]. The effectiveness of medical management is questionable, as only 16.1% of patients with GAVE and CKD in our literature review were successfully managed using only a medical approach.
Nowadays, endoscopic management is considered as the plan of choice. Several techniques have been used, including APC, laser, and sclerotherapy. APC is more easily tolerated, with fewer adverse effects, than other endoscopic techniques. Usually, multiple sessions of APC are required to control bleeding.
However, repeated sessions may lead to stenosis, gastric outlet obstruction, or perforation [17].
In general, surgical management for GAVE is considered as a last option after the failure of other management modalities [18]. However, the clinical improvement of patients was much more noticeable after surgical management vs medical or endoscopic management, in the form of a stable hemoglobin level without blood transfusion. Multiple procedures have been reported as partial vs total gastrectomy and gastroesophagectomy [17,19]. In our literature review, only 2 patients (6.5%) underwent surgical management owing to extensive disease or after the failure of non-operative modalities [4,19].
Our patient was dependent on blood transfusion, with an average transfusion of 3 packed RBCs weekly. Initially, she was managed with 3 sessions of endoscopic APC, which failed to improve her anemia or reduce her transfusion requirements. The gastroenterologist referred her to us as a refractory case for endoscopic management. Therefore, surgical management was considered after multidisciplinary meetings owing to failure of the endoscopic management as well as fear of repeated blood transfusion complications. The patient underwent laparoscopic subtotal gastrectomy with Billroth II reconstruction. The laparoscopic approach has well-known advantages over the open approach. The histopathological examination was consistent with GAVE. During close follow-up visits, the patient had a stable hemoglobin level and did not require any blood transfusion.
Herein, we highlight the efficacy of surgical management in patients with GAVE. Indeed, the laparoscopic subtotal gastrectomy approach may be considered as an effective modality of management, along with endoscopic management, owing to its favorable outcomes, in terms of bleeding, blood transfusion requirements, and nutritional status, with low complication rates if performed in a well-equipped center by an expert laparoscopic surgeon. Further well-designed studies are required to assess the efficacy and outcome of the laparoscopic subtotal gastrectomy approach as first-line management for GAVE.
Conclusions
Gastric antral vascular ectasia is a rare clinical entity that presents with acute upper-gastrointestinal bleeding or chronic anemia. It is usually associated with other comorbidities; however, an association between GAVE and CKD is rare. Its management is controversial, and endoscopic management is considered the preferred method of treatment. Laparoscopic subtotal gastrectomy is an effective management modality for GAVE, with dramatic improvement and good outcomes in terms of bleeding, blood transfusion requirements, and nutritional status.
References:
1.. Gretz J, The watermelon stomach: Clinical presentation, diagnosis, and treatment: Am J Gastroenterol, 1998; 93(6); 890-95
2.. Kar P, Mitra S, Resnick JM, Gastric antral vascular ectasia: Case report and review of the literature: Clin Med Res, 2013; 11(2); 80-85
3.. Jin T, Fei B-Y, Zheng W-H, Successful treatment of refractory gastric antral vascular ectasia by distal gastrectomy: A case report: World J Gastroenterol, 2014; 20(38); 14073
4.. Nguyen H, Le C, Nguyen H, Gastric antral vascular ectasia (watermelon stomach) – an enigmatic and often-overlooked cause of gastrointestinal bleeding in the elderly: Perm J, 2009; 13(4); 46-49
5.. Ahn Y, Wang T, Dunlop J, Treatment resistant gastric antral vascular ectasia in a patient undergoing haemodialysis: J Ren Care, 2014; 40(4); 263-65
6.. Iguchi A, Kazama JJ, Komatsu M, Three cases of gastric antral vascular ectasia in chronic renal failure: Case Rep Nephrol Urol, 2011; 1(1); 15-19
7.. Liberski S, McGarrity T, Hartle R, YAG laser therapy: Gastrointest Endosc, 1994; 40(5); 584-87
8.. Takahashi T, Miya T, Oki M, Severe hemorrhage from gastric vascular ectasia developed in a patient with AML: Int J Hematol, 2006; 83(5); 467-68
9.. Kopylov U, Yung DE, Engel T, Diagnostic yield of capsule endoscopy versus magnetic resonance enterography and small bowel contrast ultra-sound in the evaluation of small bowel Crohn’s disease: Systematic review and meta-analysis: Dig Liver Dis, 2017; 49(8); 854-63
10.. Selinger CP, Ang YS, Gastric antral vascular ectasia (GAVE): An update on clinical presentation, pathophysiology and treatment: Digestion, 2008; 77(2); 131-37
11.. Jabbari M, Cherry R, Lough JO, Gastric antral vascular ectasia: The watermelon stomach: Gastroenterology, 1984; 87(5); 1165-70
12.. Yorioka N, Hamaguchi N, Taniguchi Y, Gastric antral vascular ectasia in a patient on hemodialysis improved with CAPD: Perit Dial Int, 1996; 16(2); 177-78
13.. Chien C, Fang J, Huang C, Watermelon stomach – an unusual cause of recurrent upper gastrointestinal bleeding in a uremic patient receiving estrogen-progesterone therapy: Case report: Changgeng Yi Xue Za Zhi, 1998; 21(4); 458-62
14.. Tomori K, Nakamoto H, Kotaki S, Gastric angiodysplasia in patients undergoing maintenance dialysis: Adv Perit Dial, 2003; 19; 136-42
15.. Pisharam JK, Ramaswami A, Chong VH, Tan J, Watermelon stomach: A rare cause of anemia in patients with end-stage renal disease: Clinical Nephro, 2014; 81(1); 58-62
16.. Lee DJR, Fragata J, Pestana J, Erythropoietin resistance in end-stage renal disease patient with gastric antral vascular ectasia: J Bras Nefrol, 2015; 37; 271-74
17.. Hsu W-H, Wang Y-K, Hsieh M-S, Insights into the management of gastric antral vascular ectasia (watermelon stomach): Therap Adv Gastroenterol, 2018; 11; 1756283X17747471
18.. Belle J, Feiler M, Pappas T, Laparoscopic surgical treatment for refractory gastric antral vascular ectasia: A case report and review: Surg Laparosc Endosc Percutan Tech, 2009; 19(5); e189-e93
19.. Fuccio L, Mussetto A, Laterza L, Diagnosis and management of gastric antral vascular ectasia: World J Gastrointest Endosc, 2013; 5(1); 6
20.. Hermans C, Goffin E, Horsmans Y, Watermelon stomach. An unusual cause of recurrent upper GI tract bleeding in the uraemic patient: Efficient treatment with oestrogen-progesterone therapy: Nephrol Dial Transplant, 1996; 11(5); 871-74
21.. Fábián G, Szigeti N, Kovács T, An unusual multiplex cause of severe gastrointestinal bleeding in a haemodialysed patient: Nephrol Dial Transplant, 2000; 15(11); 1869-71
22.. Pljesa S, Golubovic G, Tomasevic R, “Watermelon stomach” in patients on chronic hemodialysis: Ren Fail, 2005; 27(5); 643-46
23.. Stefanidis I, Liakopoulos V, Kapsoritakis A, Gastric antral vascular ectasia (watermelon stomach) in patients with ESRD: Am J Kidney Dis, 2006; 47(6); e77-82
24.. George P, Pawar G, Pawar B, Gastric antral vascular ectasia in a renal transplant patient: Indian J Nephrol, 2007; 17; 23-25
25.. Lin WH, Cheng MF, Cheng HC, Watermelon stomach in a uremia patient: Kidney Int, 2010; 78(8); 821
26.. Lata S, Gupta V, Nandwani A, Watermelon stomach in end-stage renal disease patient: Indian J Nephrol, 2012; 22; 477-79
27.. Jinga M, Checheriţă IA, Becheanu G, A rare case of watermelon stomach in woman with continuous ambulatory peritoneal dialysis and systemic lupus erythematosus: Rom J Morphol Embryol, 2013; 54(3 Suppl.); 863-65
28.. Kilincalp S, Üstün Y, Karaahmet F, A new cause of severe anemia in renal transplant recipients: Watermelon stomach: Clin Exp Nephrol, 2015; 19(1); 150-51
29.. Shimamura Y, Koga H, Takizawa H, Watermelon stomach: Gastric antral vascular ectasia: Clin Exp Nephrol, 2015; 19(4); 753-54
30.. Rimševičius L, Galkauskas D, Lavinskas J, Gastric antral vascular ectasia should not be overlooked in erythropoietin resistance: A series of case reports: Acta Med Litu, 2018; 25(4); 219-25
31.. Santos S, Bernardes C, Borges V, Gastric antral vascular ectasia (GAVE) and hereditary hemorrhagic telangiectasia (HHT): Two different conditions, one treatment: Ann Hematol, 2020; 99(2); 367-69
32.. Kang SH, Kim AY, Do JY, Gastric antral vascular ectasia in a patient with lupus undergoing hemodialysis: A case report: BMC Nephrol, 2020; 21(1); 468
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133