16 December 2022: Articles 
Cardiac Arrest Following Torsades de Pointes Caused by Hypokalemia and Catecholamines in a Patient with Congenital Long QT Syndrome Type 1 After Surgical Aortic Valve Replacement: A Case Report
Unusual setting of medical care, Patient complains / malpractice, Rare disease, Adverse events of drug therapy, Educational Purpose (only if useful for a systematic review or synthesis)
Atsuhiro Kitaura1ABCDEFG*, Shinichi Nakao2CDEF, Haruyuki Yuasa1EF, Shota Tsukimoto1CDF, Yasuhumi Nakajima1FGDOI: 10.12659/AJCR.938609
Am J Case Rep 2022; 23:e938609
Abstract
BACKGROUND: Prevention of lethal arrhythmias in congenital long QT syndrome type 1 (LQT1) requires avoidance of sympathoexcitation, drugs that prolong QT, and electrolyte abnormalities. However, it is often difficult to avoid all these risks in the perioperative period of open heart surgery. Herein, we report hypokalemia-induced cardiac arrest in a postoperative cardiac patient with LQT1 on catecholamine.
CASE REPORT: A 79-year-old woman underwent surgical aortic valve replacement for severe aortic stenosis. Although the initial plan was not to use catecholamine, catecholamine was used in the Postoperative Intensive Care Unit with attention to QT interval and electrolytes due to heart failure caused by postoperative bleeding. Serum potassium levels were controlled above 4.5 mEq/L, and no arrhythmic events occurred. On postoperative day 4, the patient was started on insulin owing to hyperglycemia. Cardiac arrest occurred after the first insulin dose; the implantable cardioverter defibrillator was activated, and the patient’s own heartbeat resumed. Subsequent examination revealed that a marked decrease in serum potassium level had occurred after insulin administration. The electrocardiogram showed obvious QT prolongation and ventricular fibrillation following R on T. Thereafter, under strict potassium management, there was no recurrence of cardiac arrest events.
CONCLUSIONS: A patient with LQT1 who underwent open heart surgery developed ventricular fibrillation after Torsades de Pointes, probably due to hypokalemia after insulin administration in addition to catecholamine. It is important to check serum potassium levels to avoid the onset of Torsades de Pointes in patients with long QT syndrome. In addition, the impact of insulin administration was reaffirmed.
Keywords: Arrhythmias, Cardiac, Catecholamines, Death, Sudden, Cardiac, Diabetes Mellitus, Female, Humans, Aged, Torsades de Pointes, Romano-Ward Syndrome, Hypokalemia, Ventricular Fibrillation, Aortic Valve, Long QT Syndrome, Electrocardiography, Heart Arrest, Potassium, Insulins
Background
Congenital long QT syndrome (LQTS) is one of the genetic disorders characterized by prolonged QT interval on electrocardiogram (ECG), lethal arrhythmias such as Torsades de Pointes (TdP), ventricular fibrillation (VF), and a high possibility of sudden cardiac death [1]. More than 15 genes have been reported to be associated with LQTS [1]. Type 1 (LQT1) accounts for about 42% of cases of LQTS [2]. LQT1 is caused by mutations in the KCNQ1 gene, which causes dysfunction of slow delayed rectifier potassium current (Iks) [1]. As adrenergic stimuli are associated with cardiac events in patients with LQT1 syndrome, β blockers are often given to such patients [3]. Herein, we report a patient with LQT1 who had from cardiac arrest following TdP due to hypopotassemia and catecholamines.
Case Report
A 79-year-old woman (height: 160 cm, body weight: 59 kg) was scheduled for surgical aortic valve replacement owing to her severe aortic valve stenosis. Her past medical history included diabetes mellitus and LQTS. She was taking 8 mg/day bisoprolol and 40 mg/day verapamil, and had an implantable cardioverter defibrillator (ICD) because of her history of cardiac arrests and syncope. She had had 2 episodes of ICD actuation in the 3 months before admission. LQT1 syndrome with KCNQ1 gene mutation had been diagnosed. Her ECG showed remarkable QTc interval prolongation (Bazett formula, 644 ms) and left ventricular hypertrophy (SV1+RV5 >35 mm, strain pattern on V5–6) (Figure 1). Echocardiography revealed a left ventricular ejection fraction of 0.65 (Teichholz method), Dd/Ds of 43/28, left ventricular posterior wall thickness of 12 mm, aortic valve area of 0.7 cm2 (equation of continuity), and peak velocity of 4.1 m/s. The Society of Thoracic Surgeons risk score, European System for Cardiac Operative Risk Evaluation 2, and clinical frailty scale score were 3.56%, 2.25%, and 4, respectively.
She took bisoprolol (5 mg) in the morning on the day of surgery. In the operating room, standard monitors were used for the blood pressure (BP) cuff, pulse oximetry, and ECG (II and V5 leads). An arterial pressure line was inserted in her right radial artery before anesthesia induction. Three milligram per hour of diltiazem was given intravenously. Anesthesia induction was performed with 3 ug/mL target-controlled infusion propofol, 0.3 ug/kg/min remifentanil, and 0.6 mg/kg rocuronium and was maintained with 2 to 2.3 ug/mL target-controlled infusion propofol, 0.3 ug/kg/min remifentanil, and 0.2 mg/kg/h rocuronium. To maintain the patient’s BP, 0.25 to 0.4 µg/kg/min of phenylephrine was administered. A pulmonary artery catheter and a transesophageal echocardiograph probe were placed. An inflow duct for the cardiopulmonary bypass was placed in the ascending aorta, and an outflow duct (2-stage) was inserted into the right atrium. After the surgery, she was weaned from the cardiopulmonary bypass without problems. After cardio-pulmonary bypass, her hemodynamics, including BP, heart rate, and cardiac index (CI), were stable, without catecholamines. However, phenylephrine was administered to raise her BP. The operation time was 5 h and 23 min, anesthesia time was 7 h and 10 min, and cardiopulmonary bypass duration was 2 h and 34 min. The patient’s serum potassium ion level was kept in the range of 4.5 to 5.0 mEq/L, serum glucose level under 200 mg/dL, and serum calcium level in the normal range.
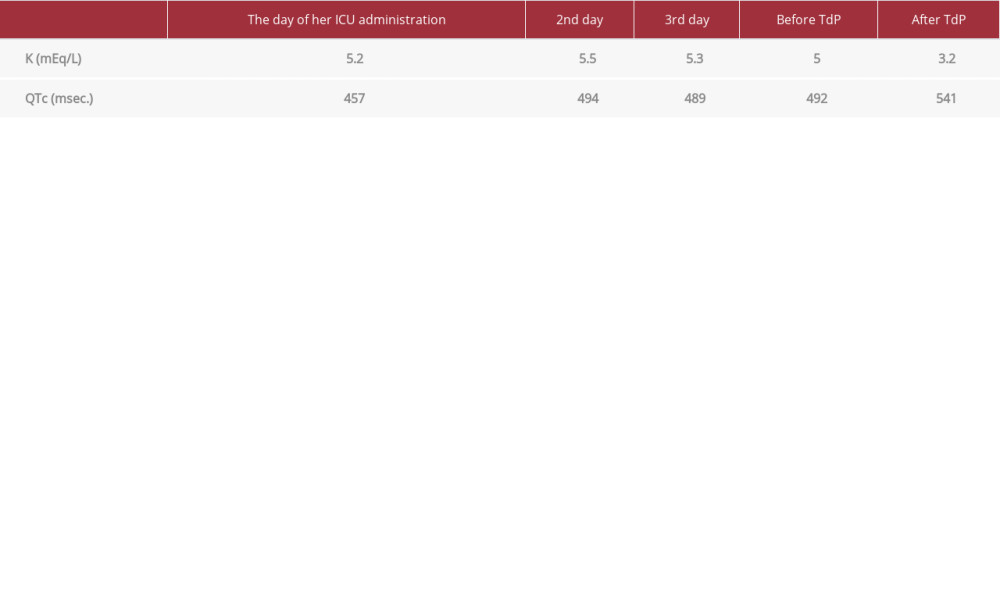
In the Intensive Care Unit (ICU), she was maintained under mechanical ventilation. The serum potassium level was kept in the range of 5.0 to 5.5 mEq/L (Table 1). Two milligram per hour of phenylephrine and 3 mg/h diltiazem were administered intravenously, and 8 mg bisoprolol tape was applied. Two hours after ICU admission, she showed signs of hemodynamic collapse, because of the decrease of cardiac preload due to hemorrhage from the surgical wound. Her deteriorated hemo-dynamics were not improved after intensive fluid replacement. The pulmonary artery pressure increased from 40 mmHg to 50 mmHg, and the central venous pressure was elevated from 14 mmHg to 20 mmHg, without an increase in BP. Furthermore, the cardiac index (CI) fell from 1.7 L/m2 to 1.0 L/m2. We decided to use catecholamines, namely 5 mcg/kg/min dobutamine and 0.03 mcg/kg/min noradrenaline. The phenylephrine was discontinued. The CI increased to 2.2 to 2.7 L/m2. On postoperative day 4, the patient was started on insulin sliding therapy by a diabetes physician for hyperglycemia. Early in the morning of the postoperative day 4, 10 units of rapid-acting insulin were administered intravenously. One hour after insulin administration, according to her ICD records, the patient had VF, and received ICD electric shocks. The VF was changed to sinus rhythm immediately. Her serum potassium level immediately after recovery was 3.2 mEq/L. The monitored ECG, which was checked after the event, showed a prolonged QTc interval of 643 m/s, and the VF occurred as R on T from the intrusion of premature ventricular contraction (Figure 2). The hypokalemia was thought to have been caused by the insulin therapy initiated that day. In addition to potassium supplementation, bisoprolol was restarted. She was discharged from the ICU on postoperative day 5 without event. Later, the RR interval and QT interval (from the onset of the QRS complex to the end of the T wave) were measured manually. The QT interval was adjusted for the patient’s heart rate using the Bazett formula (QTc=QT/[RR/1,000]1/2).
Discussion
In the present case, VF following TdP occurred, probably due to hypokalemia and catecholamines, in a patient with LQT1 after surgical aortic valve replacement. LQT1 is characterized by mutations in the KCNQ1 gene causing a loss of Iks.
There are 2 key points in the perioperative management of patients with LQT1 syndrome: (1) adrenergic stimuli, especially β1 activation, should be avoided or β blockers should be used if needed, and (2) drugs that can prolong action potential duration and consequently the QT interval on ECG or electrolyte disorders, particularly hypokalemia, should be avoided.
Cardiac events in patients with LQT1 syndrome are frequently triggered by adrenergic stimuli, such as physical or emotional stress. Cardiac repolarization is controlled by the outward current of rapidly (Ikr) and slowly (Iks) activating delayed rectifier potassium current channels and the inward current of L-type Ca2+ channel current Icap; thus, they are the most important determinants of action potential duration. Unlike at rest, when Ikr, but not Iks, plays an important role in controlling the action potential, Iks plays a dominant role in regulating an action potential after adrenergic stimuli. In a state of activated adrenergic stimuli, including exercise, inward currents of Ica increase due to L-type Ca2+ channel activation by cAMP-dependent kinase phosphorylation; however, the excessive inward currents can be suppressed by the activation of phosphorylated Iks channels. However, in a patient with LQT1 syndrome, dysfunctional Iks channels are unable to suppress excessive inward currents, resulting in prolonged action potential duration [4]. On the other hand, it has been reported that hypokalemia, a reduction in extracellular potassium, acutely decreases the amplitude of Ikr in cardiomyocytes and induces reduction in the cell surface Ikr channels [5].
In the present case, the patient had an ICD because of several episodes of TdP and VF, and was administered bisoprolol and verapamil before surgery. In the operating room, we did not use β blockers but continuously administered diltiazem, a calcium channel blocker. In addition, we decided not to use β stimulant drugs and, instead, used phenylephrine, a pure α stimulant, in order to maintain BP. Furthermore, we maintained the serum potassium ion level in the range of 4.5 to 5.0 mEq/mL. However, in the ICU, after the patient was precipitated to hypovolemic shock due to hemorrhage from the surgical wound, the patient’s hemodynamics deteriorated: the BP decreased; pulmonary artery and central venous pressure increased; and there was a marked CI decrease. Therefore, we administered dobutamine and noradrenaline. However, after insulin administration to correct the hyperglycemia, VF suddenly occurred following TdP, probably due to hypokalemia (3.2 mEq/L) in addition to the catecholamines. Even though poorly controlled diabetes also increases the risk of fatal arrhythmias in patients with prolonged QT syndrome [6], more attention should have been paid to insulin use. Specifically, to reduce potassium fluctuations, insulin boluses should have been avoided, or the number of units should have been reduced, and continuous administration of insulin should have been considered. Serum potassium levels should have been monitored immediately after insulin administration. After correction of the hypokalemia and bisoprolol use, lethal arrhythmias were not observed. It was imperative to use catecholamines because the patient had heart failure after hypovolemic shock; however, we should have used a β blocker as well and carefully monitored serum potassium levels.
General anesthetics also affect the QT interval [7]. In general, volatile anesthetics, such as sevoflurane, isoflurane, and desflurane, inhibit Ikr and prolong the QT interval. In contrast, propofol does not inhibit Ikr and does not induce QT prolongation. Theoretically, propofol is safer than volatile anesthetics in terms of anesthetic management of patients with LQTS. We used propofol in the present case. A review reported that 128 of 158 (81%) patients with LQTS were anesthetized with volatile anesthetics and there was no correlation between general anesthetics and the chance of TdP, and 76% (120/158) of patients took β blockers in the morning of surgery [8]. Based on this report, β blocker use is important for the anesthetic management of patients with LQTS [8].
Transcatheter aortic valve replacement is a more reasonable option for the patient because, in most cases, patients do not need catecholamines during and after surgery and there is little chance of surgical bleeding [9,10]. At the time of our experience with this case, our facility could not perform transcatheter aortic valve replacement on this patient quickly enough, so we performed surgical aortic valve replacement. Today, we would have opted for transcatheter aortic valve replacement.
Conclusions
A patient with LQT1 who underwent open heart surgery had VF following TdP, probably due to hypokalemia after insulin administration in addition to catecholamines. It is important to check serum potassium levels to avoid the onset of TdP in patients with LQTS.
Figures
References:
1.. Mizusawa Y, Horie M, Wilde AAM, Genetic and clinical advances in congenital long QT syndrome: Circ J, 2014; 78; 2827-33
2.. Drake E, Preston R, Douglas J, Brief review: Anesthetic implications of long QT syndrome in pregnancy: Can J Anesth, 2007; 54; 561-72
3.. Moss AJ, Shimizu W, Wilde AA, Clinical aspects of type-1 long-QT syndrome by location, coding type, and biophysical function of mutations involving the KCNQ1 gene: Circulation, 2007; 115; 2481-89
4.. Wu J, Ding WG, Horie M, Molecular pathogenesis of long QT syndrome type 1: Arrhythm, 2016; 32; 381-88
5.. Guo J, Massaeli H, Xu J, Extracellular K+ concentration controls cell surface density of IKr in rabbit hearts and of the HERG channel in human cell lines: J Clin Invest, 2009; 119; 2745-57
6.. Thiruvenkatarajan V, Osborn KD, Van Wijk RM, Torsade de pointes in a patient with acute prolonged QT syndrome and poorly controlled diabetes during sevoflurane anaesthesia: Anaesth Intensive Care, 2010; 38; 555-59
7.. Kang J, Reynold WP, Chen XL, Mechanisms underlying the QT interval-prolonging effects of sevoflurane and its interactions with other QT-prolonging drugs: Anesthesiology, 2006; 104; 1015-22
8.. Whyte SD, Nathan A, Myers D, The safety of modern anesthesia for children with long QT syndrome: Anesth Analg, 2014; 119; 932-38
9.. Villablanca PA, Mohananey D, Nikolic K, Comparison of local versus general anesthesia in patients undergoing transcatheter aortic valve replacement: A meta-analysis: Catheter Cardiovasc Interv, 2018; 91(2); 330-42
10.. Leon MB, Smith CR, Mack MJ, Transcatheter or surgical aortic-valve replacement in intermediate-risk patients: N Engl J Med, 2016; 374; 1609-20
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133