02 April 2023: Articles 
A 30-Year-Old Woman with an 8-Week History of Anxiety, Depression, Insomnia, and Mild Cognitive Impairment Following COVID-19 Who Responded to Accelerated Bilateral Theta-Burst Transcranial Magnetic Stimulation Over the Prefrontal Cortex
Unusual setting of medical care
Chun-Hung Chang123ABCDEF, Shaw-Ji Chen45CD, Yu-Chi Chen1BCD, Hsin-Chi Tsai67ABCDEFG*DOI: 10.12659/AJCR.938732
Am J Case Rep 2023; 24:e938732
Abstract
BACKGROUND: This report is of a 30-year-old woman with an 8-week history of anxiety, depression, insomnia, and mild cognitive impairment following COVID-19 infection, who responded to accelerated bilateral theta-burst transcranial magnetic stimulation (TBS) over the prefrontal cortex.
CASE REPORT: A previously healthy 30-year-old woman visited our psychiatric clinic for symptoms including anxiety, depression, insomnia, and brain fog (mild cognitive impairment) for more than 8 weeks after being diagnosed with COVID-19 on May 9, 2022. Continuous TBS of the right dorsolateral prefrontal cortex (DLPFC), followed by intermittent TBS of the left DLPFC, was performed twice daily over 5 days for a total of 10 sessions. The Beck Depression Inventory (BDI), Hamilton Depression Rating Scale (HAMD), Beck Anxiety Inventory (BAI), Pittsburgh Sleep Quality Index (PSQI), and subsets of the Wechsler Memory Scale (WMS)-Third Edition were administered at baseline and at the end of treatment. After 10 sessions of treatment, her BAI, BDI, HAMD, PSQI, WMS-Logical Memory, WMS-Faces, WMS-Verbal Paired Associates, and WMS-Family Pictures scores had improved from 4, 18, 10, 14, 8, 10, 12, and 8, respectively, to 0, 7, 1, 10, 15, 15, 15, and 10, respectively.
CONCLUSIONS: Accelerated TBS over the bilateral DLPFC may ameliorate long-COVID-associated neuropsychiatric symptoms. Additional trials are warranted to evaluate the effect of neuropsychiatric symptoms following COVID-19.
Keywords: COVID-19, Transcranial Magnetic Stimulation, Female, Humans, Adult, Sleep Initiation and Maintenance Disorders, Depression, post-acute COVID-19 syndrome, COVID-19, Anxiety, Prefrontal Cortex
Background
The rapid worldwide spread of the SARS-CoV-2 coronavirus resulted in the COVID-19 pandemic [1]. Some patients have experienced long-term or delayed effects after recovering from the acute phase of COVID-19 [2]. The term “long COVID” is used to refer to symptoms that persist 4 to 12 weeks after the acute illness and recovery. Long COVID symptoms include fatigue, dyspnea, cardiac abnormalities, cognitive impairment, sleep disturbances, muscle pain, concentration problems, and headache [3]. In a study involving 1733 patients discharged after COVID-19, depression was diagnosed in 23% of the patients, and the median followup time was 186 days [4]. A systematic review of 66 studies showed that the incidence of depression and anxiety ranged from none to >30% at followup (up to 199 days after discharge) [5]. In a study of 208 patients without premorbid neurological conditions who were hospitalized for COVID-19, the rate of cognitive deficit after a 6-month followup period was 17.5% [6].
Theta-burst stimulation (TBS) is a novel repetitive transcranial magnetic stimulation (rTMS) treatment that involves short bursts of high-frequency (50-Hz) stimulation repeated at 200-ms intervals (5 Hz). TBS, including intermittent TBS (iTBS) and continuous TBS (cTBS) has been approved for use on patients with treatment-resistant depression and has also shown promising results for neuropsychiatric disorders. In addition to regular TBS (600 pulses/session), accelerated TBS generally refers to multiple sessions per day (more than 1 daily session) [7,8]. A pilot study found that accelerated TBS may be highly effective, safe, and capable of conferring a rapid response with short treatment duration (less than 10 minutes per session) [9]. Konstantinou and his colleagues reported a case of a 66-year-old female patient with a severe major depressive episode who reached remission after 2 modified accelerated intermittent TBS treatments during the COVID-19 pandemic [10].
This report is of a 30-year-old woman with an 8-week history of anxiety, depression, insomnia, and mild cognitive impairment following COVID-19 who responded to accelerated bilateral theta-burst transcranial magnetic stimulation over the prefrontal cortex.
Case Report
A 30-year-old woman visited our psychiatric clinic for symptoms including anxiety, depression, insomnia, and brain fog (mild cognitive impairment). She was experiencing fever and cough with off-white sputum. She did not have dyspnea, chest pain, or loss of smell or taste. Two of her colleagues had received COVID-19 diagnoses 2 days prior. She visited the emergency department and received a COVID-19 diagnosis on May 9, 2022 (day 1) in the form of a positive result from nasopharyngeal specimens tested for SARS-CoV-2 by real-time reverse-transcriptase-polymerase-chain-reaction (RT-PCR) (Roche, Manheim, Germany). In accordance with regulations, she isolated at home for 7 days. She received medications including dextromethorphan 90 mg/d, acetaminophen 1500 mg/d, levocetirizine 5 mg/d, benzonatate 300 mg/d, and fenoterol 2.5 mg/d for 7 days. Her fever subsided gradually within 3 days (day 3). However, she experienced mild anxiety, depression, insomnia, and brain fog during isolation. On day 7, she tested negative in an antigen self-test for SARS-CoV-2 and subsequently returned to work.
However, her anxiety, depression, insomnia, and brain fog (mild confusion) persisted for 8 weeks. She was easily fatigued and was making mistakes at work more frequently than she usually would. She visited a family clinic. Long COVID was suspected. She was referred to our long-COVID clinic on August 1, 2022. A biochemistry blood exam, chest X-ray, and electrocardiography were arranged to exclude organic causes. The results were within normal range. In addition to a depressed mood, she also experienced lack of energy, a loss of interest in activities, feelings of worthlessness, and psychomotor retardation. Depressive disorder was diagnosed by a psychiatrist according to the diagnostic and statistical manual of mental disorders-5 (DSM-5) criteria [11]. She did not want to receive medications for depression and anxiety. She was in good health before COVID-19 infection, and had no past psychiatric history prior to the COVID-19 infection. The Beck Depression Inventory (BDI) [12] and Hamilton depression rating scale (HAMD) were used to evaluate the severity of her depression. The Beck Anxiety Inventory (BAI) [13], and Pittsburgh Sleep Quality Index (PSQI) were used to measure her anxiety and sleep quality [14]. The severity of depression, anxiety, and poor sleep quality increases with the scores; that is, higher scores mean worse depression, anxiety, and sleep quality. We used the subsets of the Wechsler Memory Scale (WMS)–Third Edition to evaluate her cognitive functions [15,16]. Higher scores on the WMS mean better cognitive functions. Her BAI, BDI, HAMD, PSQI, WMS-Logical Memory, WMS-Faces, WMS-Verbal Paired Associates, and WMS-Family Pictures scores were 4, 18, 10, 14, 8, 10, 12, and 8, respectively.
After the patient provided written informed consent, we decided to try TBS, a novel rTMS treatment, and began accelerated TBS of the bilateral dorsolateral prefrontal cortex (DLPFC) using an Apollo TMS Therapy System (MAG & More, Munich, Germany) equipped with a figure-of-8 coil. First, we applied 20 trains of 10 cTBS bursts (short bursts of 3 stimuli at 50 Hz, repeated at 200-ms intervals) targeting the right DLPFC, with 600 pulses/session for 40 s at 80% of the resting motor threshold (RMT). RMT is defined as the lowest stimulus intensity required to induce a motor-evoked potential with a peak-to-peak amplitude of at least 50 microvolts in 5 out of 10 consecutive trials in the relaxed target muscle. Second, we applied 20 trains of 10 iTBS bursts (short bursts of 3 stimuli at 50 Hz, repeated at 200-ms intervals) targeting the left DLPFC with 8-s intervals, with 600 pulses/session for 200 s at 80% of the RMT. The coil localization was based on the algorithm developed by Beam et al [17]. The Beam-F4 position was used for the right DLPFC and the Beam-F3 position was used for the left DLPFC. Subsequent stimulation sessions were performed twice daily (around 9: 00 am and 3: 00 pm) over 5 days within 1 week, for a total of 10 sessions (Figure 1). The patient did not receive medications or psychotherapy for depression or anxiety during the TBS treatment process.
The patient’s symptoms gradually lessened. After 10 TBS sessions, her BAI, BDI, HAMD, PSQI, WMS-Logical Memory, WMS-Faces, WMS-Verbal Paired Associates, and WMS-Family Pictures scores had improved to 0, 7, 1, 10, 15, 15, 15, and 10, respectively (Table 1). She felt that activities and functions improved after the TBS treatment course. Adverse effects of the treatment, such as dizziness and headache, were transient and resolved after treatment. She did not receive medications or psychotherapy after TBS treatment. After the 3-month follow-up, her clinical improvement continued; at this timepoint, she scored 0, 5, 1, and 8 points on the BAI, BDI, HAMD, and PSQI.
Discussion
We present in this report a case of the application of accelerated TBS of the bilateral DLPFC in a patient with COVID-19-associated neuropsychiatric symptoms including depression and mild cognitive impairment. Our patient demonstrated significant clinical improvement after 10 treatment sessions, without severe adverse effects.
Our findings agreed with a previous report of accelerated intermittent TBS (aiTBS) used to treat late-life depression during the COVID-19 pandemic. To reduce the risk of COVID-19 infection, Konstantinou and his colleagues treated a 66-year-old female patient with a severe major depressive episode using aiTBS. Moderate to severe baseline depressive symptoms reached remission levels after 8 daily sessions over 5 consecutive days, followed by gradual tapering, using 1800 pulses per session pre-COVID-19 (first course), and 600 pulses per session during the pandemic (second course) [10]. In our case, we treated a 30-year-old woman with COVID-19-associated neuropsychiatric symptoms including depression and cognitive impairment. Because our patient hoped to improve her condition rapidly during the COVID-19 pandemic, we chose to use accelerated bilateral TBS treatment (cTBS over the right DLPFC followed by iTBS over the left DLPFC; 1200 pulses per session) twice daily. Her symptoms improved after 10 sessions of treatment.
After TBS treatment, our patient’s depressive symptoms were attenuated (BDI score decreased from 18 to 7). Previous studies have shown that TBS is not inferior to conventional (10-Hz) rTMS for unipolar depression [18,19]. In a randomized sham-controlled study, 60 treatment-refractory patients with recurrent major depressive disorder were separated randomly into 4 groups of 15 patients to receive cTBS, iTBS, a combination of cTBS and iTBS, or sham TBS. After 2 weeks of treatment, the patients who received a combination of cTBS and iTBS demonstrated the greatest antidepressant effect, after controlling for age and refractoriness scores [18]. A meta-analysis of 10 TBS studies revealed that the overall effect sizes for the response and remission rates were 0.38 (95% confidence interval [CI]: 0.29–0.48) and 0.20 (95% CI: 0.13–0.29), respectively [19]. Moreover, Stanford accelerated intelligent neuro-modulation therapy, an accelerated, high-dose resting-state functional connectivity MRI-guided iTBS treatment, is more effective than sham stimulation for treatment-resistant depression [9]. Further trials are required to determine the efficacy of TBS for long-COVID-associated depression.
In our case, we noted improved cognitive function after TBS treatment. A pilot study suggested that TBS could be used to improve associative memory and increase the resting-state functional connectivity of the hippocampus as well as the occipital fusiform gyrus, orbitofrontal cortex, putamen, posterior parahippocampal gyrus, and temporal pole along the inferior longitudinal fasciculus in patients with mild cognitive impairment [20]. A systematic review and meta-analysis showed that, with outlier studies removed, iTBS enhanced cognition (relative to control conditions; standardized mean difference=0.21, 95% CI: 0.003–0.42,
The link between COVID and neuropsychiatric symptoms, including depression and mild cognitive confusion, may result from several possible mechanisms. First, acute viral damage of the central nervous system (CNS) may cause the neuropsychiatric symptoms. This could occur in long COVID via hematogenous or neuronal retrograde neuro-invasive routes. Second, SARS-CoV-2 may also affect the permeability of the blood-brain barrier. This may enable peripheral cytokines and other blood-derived substances to enter the CNS and further drive neuro-inflammation. Third, psychosocial stress due to isolation and decreased physical activities may also contribute to psychiatric symptoms including depression and anxiety [3,22].
Several hypotheses may support the effectiveness of TBS over bilateral DLPFC in treating COVID-associated depression and cognitive impairment. First, previous animal and human studies have found that the stimulation of DLPFC can improve mood and cognitive function [21,23]. Second, the improvement in cognitive functioning could be associated with improvement in depression [24,25]. Depression may result in cognitive impairment. TBS may help to improve the severity of depression-associated cognitive impairment.
This case report has several limitations. First, only 1 patient was evaluated. We lacked controls for sham stimulation. Second, baseline scores prior to COVID-19 were unavailable; thus, we could not compare the severity of depression and cognitive impairment before and after COVID-19. However, neither this patient nor her family had a history of depression. In addition, no brain fog was noted before the COVID pandemic. Third, confounding factors may not be comprehensively excluded. She received medications including dextromethorphan, acetaminophen, levocetirizine, benzonatate, and fenoterol. However, these medications were taken for COVID-19 infection for only 7 days. Her neuropsychiatric diseases persisted after stopping these medications. Besides, she and her family had no psychiatric history before the COVID-19 infection. Therefore, these neuropsychiatric symptoms are likely to be associated with the COVID-19 infection.
Conclusions
In this case, we reported a 30-year-old woman with an 8-week history of anxiety, depression, insomnia, and mild cognitive impairment following COVID-19 illness, who responded to accelerated bilateral theta-burst transcranial magnetic stimulation over the prefrontal cortex. Studies with larger sample sizes and randomized, double-blind, placebo-controlled trials are required to verify our initial findings of the efficacy of this treatment.
References:
1.. Dong E, Du H, Gardner L, An interactive web-based dashboard to track COVID-19 in real time: Lancet Infect Dis, 2020; 20(5); 533-34
2.. Nalbandian A, Sehgal K, Gupta A, Post-acute COVID-19 syndrome: Nat Med, 2021; 27(4); 601-15
3.. Tang SW, Leonard BE, Helmeste DM, Long COVID, neuropsychiatric disorders, psychotropics, present and future: Acta Neuropsychiatr, 2022; 34(3); 109-26
4.. Huang C, Huang L, Wang Y, 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study: Lancet, 2021; 397(10270); 220-32
5.. Schou TM, Joca S, Wegener G, Bay-Richter C, Psychiatric and neuropsychiatric sequelae of COVID-19 – a systematic review: Brain Behav Immun, 2021; 97; 328-48
6.. Pilotto A, Cristillo V, Cotti Piccinelli S, Long-term neurological manifestations of COVID-19: Prevalence and predictive factors: Neurol Sci, 2021; 42(12); 4903-7
7.. Huang YZ, Edwards MJ, Rounis E, Theta burst stimulation of the human motor cortex: Neuron, 2005; 45(2); 201-6
8.. Lefaucheur JP, Aleman A, Baeken C, Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018): Clin Neurophysiol, 2020; 131(2); 474-528
9.. Cole EJ, Phillips AL, Bentzley BS, Stanford neuromodulation therapy (SNT): A double-blind randomized controlled trial: Am J Psychiatry, 2022; 179(2); 132-41
10.. Konstantinou GN, Downar J, Daskalakis ZJ, Blumberger DM, Accelerated intermittent theta burst stimulation in late-life depression: A possible option for older depressed adults in need of ECT during the COVID-19 pandemic: Am J Geriatr Psychiatry, 2020; 28(10); 1025-29
11.. Du YL, Hu JB, Huang TT, Psychometric properties of the Clinically Useful Depression Outcome Scale supplemented with DSM-5 Mixed sub-type questionnaire in Chinese patients with mood disorders: J Affect Disord, 2021; 279; 53-58
12.. Beck AT, Ward CH, Mendelson M, An inventory for measuring depression: Arch Gen Psychiatry, 1961; 4; 561-71
13.. Beck AT, Epstein N, Brown G, Steer RA, An inventory for measuring clinical anxiety: Psychometric properties: J Consult Clin Psychol, 1988; 56(6); 893-97
14.. Buysse DJ, Reynolds CF, Monk TH, The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research: Psychiatry Res, 1989; 28(2); 193-213
15.. Lo AH, Humphreys M, Byrne GJ, Pachana NA, Test-retest reliability and practice effects of the Wechsler Memory Scale-III: J Neuropsychol, 2012; 6(2); 212-31
16.. Seelye AM, Howieson DB, Wild KV, Wechsler Memory Scale-III Faces test performance in patients with mild cognitive impairment and mild Alzheimer’s disease: J Clin Exp Neuropsychol, 2009; 31(6); 682-88
17.. Beam W, Borckardt JJ, Reeves ST, George MS, An efficient and accurate new method for locating the F3 position for prefrontal TMS applications: Brain Stim, 2009; 2(1); 50-54
18.. Li CT, Chen MH, Juan CH, Efficacy of prefrontal theta-burst stimulation in refractory depression: A randomized sham-controlled study: Brain, 2014; 137(Pt 7); 2088-98
19.. Chu HT, Cheng CM, Liang CS, Efficacy and tolerability of theta-burst stimulation for major depression: A systematic review and meta-analysis: Prog Neuropsychopharmacol Biol Psychiatry, 2021; 106; 110168
20.. Chen YC, Ton That V, Ugonna C, Diffusion MRI-guided theta burst stimulation enhances memory and functional connectivity along the inferior longitudinal fasciculus in mild cognitive impairment: Proc Natl Acad Sci USA, 2022; 119(21); e2113778119
21.. Pabst A, Proksch S, Mede B, A systematic review and meta-analysis of the efficacy of intermittent theta burst stimulation (iTBS) on cognitive enhancement: Neurosci Biobehav Rev, 2022; 135; 104587
22.. Crook H, Raza S, Nowell J, Long COVID-mechanisms, risk factors, and management: BMJ, 2021; 374; n1648
23.. Voigt JD, Leuchter AF, Carpenter LL, Theta burst stimulation for the acute treatment of major depressive disorder: A systematic review and meta-analysis: Transl Psychiatry, 2021; 11(1); 330
24.. Toffanin T, Folesani F, Ferrara M, Cognitive functioning as predictor and marker of response to repetitive transcranial magnetic stimulation in depressive disorders: A systematic review: Gen Hosp Psychiatry, 2022; 79; 19-32
25.. Zou C, Yu Q, Wang C, Association of depression with cognitive frailty: A systematic review and meta-analysis: J Affect Disord, 2022; 320; 133-39
Tables
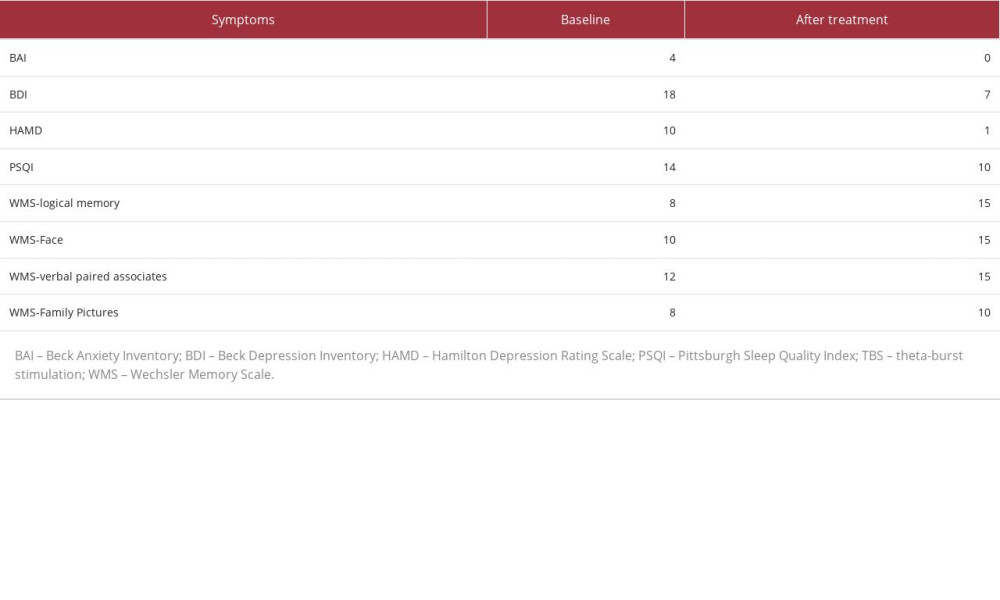 Table 1.. Clinical outcomes of accelerated TBS treatment. Anxiety, depression, insomnia, and mild cognitive impairment following COVID-19 responded to accelerated bilateral theta-burst transcranial magnetic stimulation over the prefrontal cortex.
Table 1.. Clinical outcomes of accelerated TBS treatment. Anxiety, depression, insomnia, and mild cognitive impairment following COVID-19 responded to accelerated bilateral theta-burst transcranial magnetic stimulation over the prefrontal cortex. Table 1.. Clinical outcomes of accelerated TBS treatment. Anxiety, depression, insomnia, and mild cognitive impairment following COVID-19 responded to accelerated bilateral theta-burst transcranial magnetic stimulation over the prefrontal cortex.
Table 1.. Clinical outcomes of accelerated TBS treatment. Anxiety, depression, insomnia, and mild cognitive impairment following COVID-19 responded to accelerated bilateral theta-burst transcranial magnetic stimulation over the prefrontal cortex. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








