24 February 2023: Articles 
Optic Neuritis as a Consequence of Long-Term Medical Treatment Using Anastrozole: A Case Report
Challenging differential diagnosis, Unusual or unexpected effect of treatment, Adverse events of drug therapy, Rare coexistence of disease or pathology
Gabriel Velez Oquendo1BE*DOI: 10.12659/AJCR.939026
Am J Case Rep 2023; 24:e939026
Abstract
BACKGROUND: Vision loss secondary to optic neuritis is an uncommon adverse effect of nonsteroidal aromatase inhibitors. There have been few reports in the literature on visual disturbance in patients on long-term treatment with Anastrozole for breast cancer prevention; but none had symptoms worse than blurry vision and/or xerostomia. The present patient had acute onset of right-sided vision loss without other neurologic deficits while using the aromatase inhibitor Anastrozole for breast cancer treatment.
CASE REPORT: A 69-year-old woman presented to the Emergency Department with approximately 1 month of worsening right eye vision loss that was not associated with any other neurologic deficits or any acute symptoms. The symptom was constant and without alleviating or aggravating factors. After extensive workup with ophthalmologic evaluation, Infectious Disease evaluation, autoimmune tests, brain imaging, lumbar puncture with CSF analysis, and temporal artery biopsy reporting unremarkable results, it was determined that the patient was in an inflammatory state induced by long-term use of Anastrozole, an aromatase inhibitor.
CONCLUSIONS: The patient’s long-term use of Anastrozole likely played a large part in developing right visual disturbance secondary to optic neuritis, as a patient of this age has little risk of developing conditions such as optic neuritis, unilateral loss of vision, and/or autoimmune conditions.
Keywords: Anastrozole, Aromatase Inhibitors, optic neuritis, Vision, Low, Female, Humans, Aged, Vision Disorders, Breast Neoplasms
Background
Breast cancer is the most common non-skin cancer among women in the United States and the second leading cause of cancer-related death [1]. Early work on therapeutic prevention of breast cancer focused on selective estrogen receptor modulators (SERMs), such as tamoxifen and raloxifene, which show anti-estrogenic effects on the breast, as well as agonist and antagonistic effects on other organs. In recent years, aromatase inhibitors (AIs) such as Anastrozole and letrozole have been increasingly prescribed to postmenopausal breast cancer patients as adjuvant therapy after tamoxifen treatment [2]. The short-term ocular adverse effects of AIs often seem to be mild; some studies suggest that Anastrozole can cause small retinal hemorrhages in some patients, which may be the result of excessive traction on the retina caused by estrogen depletion [2]. Other possible adverse effects of AIs include photopsia and increased incidence of floaters. A prospective observational study showed that AI therapy causes deteriorations in many of the ocular surface parameters and corneal structural changes, which are associated with the duration of treatment [3]. In a systematic review of trials conducted for the United States Preventive Services Task Force, aromatase inhibitors such as tamoxifen and raloxifene were found to reduce the incidence of invasive breast cancer [1], and these absolute benefits are likely even higher for women with a predicted breast cancer risk of 3% or greater. The adverse effects of aromatase inhibitors include vasomotor symptoms, gastrointestinal symptoms, and musculoskeletal pain. Data on long-term adverse effects of aromatase inhibitors for reduction of breast cancer are limited [1]. This case report presents a patient with no known history of multiple sclerosis, optic neuritis, or autoimmune diseases, who was currently using an aroma-tase inhibitor, which resulted in significant visual disturbance.
Case Report
A 69-year-old woman with a past medical history of sleep apnea on CPAP, hypothyroidism, fibromyalgia, depression, right knee osteoarthritis, and history of left breast cancer on Anastrozole medical treatment presented to the Emergency Department with worsening right eye vision loss. She reported that she started having vision loss approximately 1 month before admission. Consequently, she visited her optometrist, who referred her to a neuro-ophthalmologist, who discovered swelling of the right optic nerve. Results of an extensive work-up including MRI of the brain and temporal artery biopsy were unremarkable. She was prescribed a 1-week course of prednisone 80 mg PO daily. Her visual loss continued to progress, involving the bottom half of the right eye visual field. Due to worsening of her visual disturbance, she presented with progressive right eye blindness, having a “dark stripe covering over ¾ of the visual field”. The patient described it as if she was “looking through a screen with little light passing through”. Associated symptoms included severe headache behind the right eye radiating to the occipital area of her head, and mild bilateral leg weakness and fatigue, which were relieved with rest. She denied other associated symptoms such as high-grade fever, nuchal rigidity, temporal tenderness, jaw claudication, dizziness, dysphagia, chest pain, palpitations, shortness of breath, bowel changes, nausea, vomiting, urinary incontinence, musculoskeletal pain, unintentional weight loss, or loss of consciousness. She also denied any family history of auto-immune diseases, specifically multiple sclerosis.
In the Emergency Department she was hemodynamically stable. A laboratory workup including CBC and CMP was unre-markable. Urinalysis showed a WBC count of 11, RBC count of 7, but negative for nitrites, as well as ESR of 33 and CRP <0.40. A CTA of the head and neck showed no acute intracranial processes. Afterwards, the patient received methylprednisolone 500 mg in the Emergency Department, Neurology was consulted for further workup and recommendations, and Hospital Medicine was consulted for admission of the patient and further management.
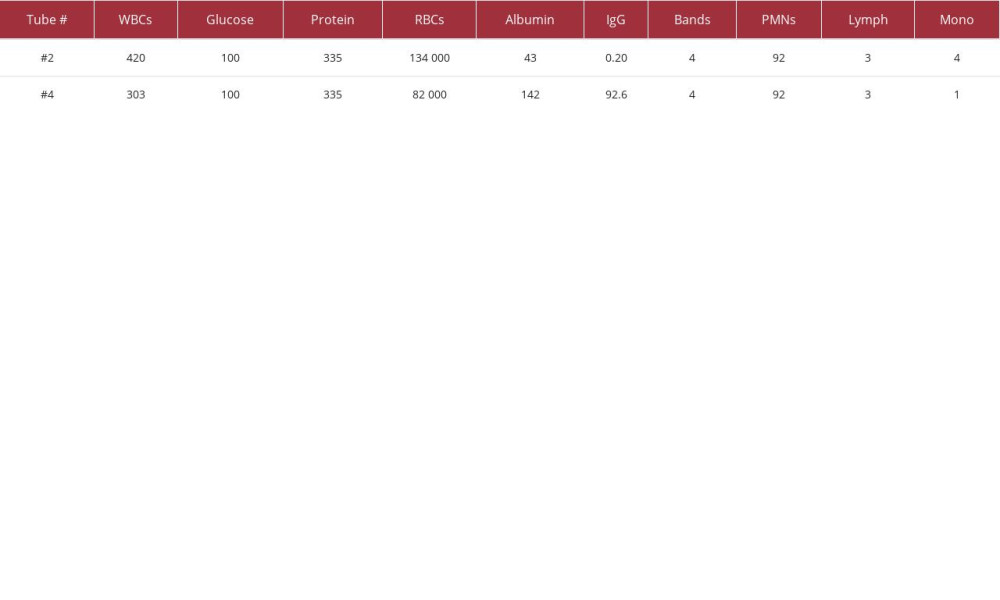
The patient’s MRI of the brain and orbits with and without contrast did not show any abnormality within either optic nerve, which were symmetric in appearance. There was no abnormal enhancement or mass lesions. The right temporal biopsy during hospitalization was negative for active or chronic arteritis. Lumbar puncture attempts at L3–L4 and L5–S1 were unsuccessful. The patient was prepped and draped in the maximal sterile fashion again, with successful advancement into the thecal sac. There was return of approximately 14 ml of blood-tinged cerebrospinal fluid collected in 4 tubes and sent for lab evaluation. Only cerebrospinal fluid (CSF) analysis of tubes #2 and #4 reported results with a cloudy appearance with xanthochromia. CSF non-gynecologic cytology was negative in all tube specimens. Gram stain was negative. The Meningitis and Encephalitis PCR panel was negative (Table 1).
The Multiple Sclerosis panel reported increased levels of immunoglobulin G at 543, CSF immunoglobulin G at 29.1, albumin at 3276, CSF albumin 142, albumin index 43.3, IgG index 1.24, CSF IgG/Albumin ratio 0.20, CSF oligoclonal bands were negative, and CSF IgG synthesis rate of 92.6. Isoelectric focusing/immunofixation revealed matching or identical oligoclonal bands in the CSF and the serum. This result is consistent with a systemic, not an intrathecal immune reaction, and is considered to be a negative result for oligoclonal bands. Approximately 5% of patients with clinically definitive multiple sclerosis will have a negative result. Increased concentrations of IgG in the CSF are an important indicator for MS but may also be associated with increased permeability of the blood-CSF barrier, or increased local production of IgG, or both. Increased IgG production is demonstrated by an increased CSF IgG/Albumin ratio, IgG Index, and IgG synthesis rate. An elevated albumin index indicates damage to the blood-CSF barrier or contamination of CSF with blood during sample collection. An index value less than 9 is considered consistent with an intact barrier. Values of 9 to 14 are interpreted as slight impairment, of 14 to 30 as moderate impairment, and of 30 to 100 as severe impairment. Values exceeding 100 indicate complete breakdown of the barrier.
During the second day of steroid treatment, she indicated that her visual disturbance started improving, and no longer had the symptoms she first presented with. At this point, Neurology did not recommend further workup, and the patient was subsequently discharged with indications to follow-up with Outpatient Neurology and Ophthalmology if further worsening of symptoms occurred or new concerns arose.
Discussion
This case report suggests the possibility of a high-risk long-term medication such as Anastrozole, a nonsteroidal aroma-tase inhibitor used to treat hormone receptor-positive breast cancer. It is also used as a first-line treatment of hormone receptor-positive or hormone receptor-unknown advanced and/or metastatic breast cancer, as well as being used for breast cancer that has failed tamoxifen treatment. Importantly, this medical treatment can only be used in postmenopausal females.
The incidence of certain adverse effects, especially visual disturbance, related to AI use is quite variable. Papilledema and macular edema secondary to use of Anastrozole secondary to possible decrease in estrogen levels leading to optic nerve swelling has been reported before [4]. In that case, the patient experienced complete resolution of swelling and visual impairment in the affected eye after a course of steroid treatment, similar to the presented case. Anastrozole use appears to be associated with an increased prevalence of retinal hemorrhages, which may mark systemic vascular compromise resulting from estrogen depletion, and/or they may be consequences of vitreoretinal traction resulting from estrogen depletion. They may also depend on the use of medications for controlling common Anastrozole-induced adverse effects [6]. Although visual disturbances are possible adverse effects of aromatase inhibitor use, the incidence is quite variable. The literature demonstrates evidence of retinopathy secondary to Anastrozole therapy and other AIs. The management includes stopping the causing medication and providing steroidal treatment [5].
Ocular complications secondary to aromatase inhibitor use are documented, but continue to be reported less when compared to tamoxifen use. Prompt ophthalmologic evaluation and high index of suspicion is recommended when assessing patients with visual disturbances currently under treatment with AIs. Larger prospective studies are needed to analyze the severity of ocular complications [5].
Conclusions
This case report demonstrates a patient with minimal to no risk for optic disc swelling/inflammation other than the use of Anastrozole, an aromatase inhibitor, coming to the hospital with worsening vision and signs of optic neuritis. Additional awareness should be prompted by patients presenting with similar symptoms, and emergency department physicians should suspect optic neuritis as a likely culprit in patients being treated with aromatase inhibitors, especially Anastrozole, for breast cancer prevention. This case report should encourage questioning patients about specific use of medical treatment in the presentation of symptoms similar to autoimmune presentations. There are multiple studies noting the general safety and low risk profile of aromatase inhibitor therapy. There is most likely a need for increased awareness about aromatase inhibitors for breast cancer preventions and how they can contribute to development of an inflammatory state and vision loss.
References:
1.. , Medication use to reduce risk of breast cancer: Recommendation statement: Am Fam Physician, 2020; 101(6) Online
2.. Boughton B, Watch for ocular effects of breast cancer drugs: EyeNet Magazine (American Academy of Ophthalmology) March, 2013 Available at: (Accessed: November 4, 2022)https://www.aao.org/eyenet/article/watch-ocular-effects-of-breast-cancer-drugs?march-2013
3.. Ağın A, Kocabeyoglu S, Yucel Gencoglu A, The effects of systemic aromatase inhibitors on meibomian glands and corneal structure: Eye (Lond), 2022; 36(6); 1185-93
4.. Coppes OJ, Lukas RV, Fleming GF, Bilateral optic disc swelling following anastrozole therapy: Neuroophthalmology, 2014; 38(5); 268-71
5.. Almafreji I, Smith C, Peck F, Review of the literature on ocular complications associated with aromatase inhibitor use: Cureus, 2021; 13(8); e17565
6.. Eisner A, Falardeau J, Toomey MD, Vetto JT, Retinal hemorrhages in anastrozole users: Optom Vis Sci, 2008; 85(5); 301-8
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133