16 April 2023: Articles 
Acute Immune Reconstitution Inflammatory Syndrome-HBV Flare in an HIV/HBV Coinfected Patient After Antiretroviral Therapy Initiation: Case Report and Literature Review
Unknown etiology, Challenging differential diagnosis
Iqra Arshad1AEF*, Mukti Gandhi1EF, Marcia Gossai1EF, Addi Feinstein2EFDOI: 10.12659/AJCR.939210
Am J Case Rep 2023; 24:e939210
Abstract
BACKGROUND: Immune reconstitution inflammatory syndrome (IRIS) is a well-recognized complication after antiretroviral therapy (ART) initiation among patients with HIV. Acute HBV flares after starting antiretroviral therapy have been reported in 20% to 25% of coinfected patients, among whom only 1% to 5% develop clinical hepatitis. Liver biopsy and serological evaluation help in diagnosis.
CASE REPORT: A 24-year-old man with history of HIV diagnosed in 2018 developed severe IRIS-related HBV flare after initiation of ART. He was taking ART since 2018 until his immigration to the United States in 2021. He came to establish care and was started on bictegravir/emtricitabine/tenofovir alafenamide (BIC/F/TAF). Three weeks later, he presented to the Emergency Department with polyarthralgia and loose stools; transaminases showed an increasing trend on follow-up. He was admitted for closer monitoring. Workup was remarkable for reactive HBsAg, HBeAg, and HBcIgM antibodies, with HBV viral load of 295 304 copies/mL. Abdominal imaging was unremarkable. ART was switched to rilpivirine/emtricitabine/tenofovir alafenamide (RPV/FTC/TAF), considering the hypothetical risk of hepatotoxicity from BIC/F/TAF. Despite therapy, transaminases were up-trending. He underwent computerized tomography-guided liver biopsy, showing moderate to severe acute hepatitis, compatible with IRIS. He received steroids, and ART was continued. Transaminases resolved, HBV load reduced significantly, HIV load became undetectable at 9 weeks, and he developed HBeAb (seroconversion) at 4 months after initiating ART.
CONCLUSIONS: Our case highlights the importance of early recognition and management of IRIS-HBV flares after initiation of ART among coinfected patients. Liver biopsy is indicated for definitive diagnosis. ART directed against both viruses should be continued.
Keywords: Antiretroviral Therapy, Highly Active, Hepatitis B e Antigens, HIV, Iris, Male, Humans, young adult, Adult, HIV Infections, Hepatitis B virus, coinfection, Immune Reconstitution Inflammatory Syndrome, Emtricitabine, Transaminases
Background
In about 10% to 20% of patients with HIV, immune restoration after initiation of antiretroviral therapy (ART), causes an exaggerated increase in CD4+ helper T cells and CD8+ suppressor T cells and dysregulation of regulatory T cells, resulting in a robust pathological inflammatory response directed against opportunistic infections, which is referred to as immune reconstitution inflammatory syndrome (IRIS) [1,2]. About 10% of patients with HIV have a coinfection with hepatitis B virus (HBV) and higher baseline viral loads with a subsequent increased risk of developing complications and progression to chronic HBV infection. HBV flares have been reported in 20% to 25% of coinfected patients within 2 to 8 weeks of starting ART, of whom, it is estimated that 1% to 5% progress to clinical hepatitis [7]. The diagnosis can be supported by eliminating hepatotoxicity from ART itself, viral serological testing, and by liver biopsy that shows predominant T-cell infiltration. An IRIS-HBV flare is mostly self-limiting, has a low likelihood of progressing to fulminant hepatic failure, and has been shown to rapidly lower the HBV-DNA and hepatitis B surface antigen (HBsAg) levels. Since the cessation of ART could cause worsening of a flare, ART with activity against both HBV and HIV should be continued, with corticosteroids being reserved for severe IRIS cases.
Case Report
A 24-year-old man, originally from Jamaica, with a past medical history of HIV infection, diagnosed in 2018, presented to our HIV clinic to establish care. He was a non-smoker and non-drinker. The patient had been taking ART since 2018 until his immigration to the United States in 2021, when he stopped. The duration for which patient was not on ART was unclear because he did not remember the name of the medication and the last time he took the pill prior to his presentation in our clinic. Initial laboratory assessment was notable for an HIV-1 viral load of 168 577 copies/mL, CD4 count of 494, reactive HBsAg, nonreactive HB core (c) and HB surface (s) antibodies, and non-detected HBV-DNA (Table 1). An HIV genomic study showed subtype B with pan-sensitivity to ART, and bictegravir/emtricitabine/tenofovir alafenamide (BIC/F/TAF) was started. Three weeks later, the patient presented to the Emergency Department (ED) with polyarthralgia, non-bloody diarrhea, and fatigue. The clinical examination was unremarkable; however, repeated laboratory studies showed elevated transaminases (aspartate aminotransferase [AST] 646 U/L and alanine aminotransferase [ALT] 1098 U/L, compared with AST 28 U/L and ALT 20 U/L on first clinic visit), with normal bilirubin and alkaline phosphatase levels suggestive of hepatocellular injury pattern; (Table 1). The patient received symptomatic treatment and was discharged from the ED with an appointment for the clinic. Upon follow-up at the clinic, the patient reported compliance to ART and denied interim use of herbal supplements, nonsteroidal anti-inflammatory drugs and over-the-counter medications, alcohol, and recreational drugs. IRIS vs ART/drug-induced acute liver injury in the setting of recent ART initiation was suspected; however, other causes, such as acute viral hepatitis and autoimmune hepatitis, were also considered, and further workup including all viral hepatitis serology tests were sent from the clinic. On follow-up laboratory tests, an increasing trend of transaminases (AST >800 U/L, ALT 1370 U/L) with interval development of mild direct hyperbilirubinemia (direct bilirubin 1.5 mg/dL) and elevated alkaline phosphatase levels (131 U/L), which was suggestive of a mixed hepatocellular and cholestatic pattern, were noted, and the patient was admitted to the hospital for closer monitoring. This increasing trend was observed 3 weeks after ART commencement (Table 1). Considering the rapidly rising trend of transaminases, initially, a trail of N-acetylcysteine infusion was given for 48 h, with no improvement. Further workup revealed reactive HBsAg, hepatitis B e antigen (HBeAg), and hepatitis B core IgM antibodies HBV viral load of 295 304 copies/mL, repeat HIV-1 viral load of 36 copies/mL, and elevated serum ferritin levels (Table 1). Other viral (HAV/HEV/HCV/HDV) serology tests, toxicology screening, and autoimmune panel all tested negative. Furthermore, abdominal imaging was unremarkable.
ART was continued, as sudden cessation could cause worsening of the flare, but was switched from BIC/F/TAF to rilpivirine/emtricitabine/tenofovir alafenamide (RPV/FTC/TAF) because of a hypothetical risk of hepatotoxicity from BIC/F/TAF. The patient’s liver enzyme levels continued to increase, with AST >1000 U/L and ALT >2000 U/L, but he did not meet the criteria for acute fulminant hepatic failure or liver transplant, as per the King’s College criteria. We decided to start the patient on prednisone 1–2 mg/kg per day for severe IRIS and get a computerized tomography (CT)-guided liver biopsy to differentiate between an acute HBV flare and idiosyncratic drug-induced liver injury, as per Gastroenterology service recommendations. Later, the liver biopsy showed moderate to severe acute hepatitis with T-cell predominance, consistent with an IRIS-HBV flare (Figure 1). Thus, the diagnosis was confirmed. Transaminases levels began to improve after steroid initiation, which was later tapered off with the normalization of liver enzymes after 9 weeks of restarting ART, with the resolution of the flare (Table 1). The patient had a significant reduction in HBV load to 110 946 copies/mL and an undetectable HIV load at 4 weeks and developed hepatitis B e antibodies (HBeAb), suggestive of seroconversion in 4 months of restarting ART, on follow-up (Table 1). At the time of this report, he was doing well clinically and was maintained on ART.
Discussion
Pathogens commonly implicated in IRIS are mycobacterium, herpes simplex virus, cryptococcus, cytomegalovirus, and HBV [3,4]. Symptoms associated with IRIS due to HBV infection are fatigue, polyarthralgia, anorexia, tender hepatomegaly, and jaundice; however, these symptoms can vary depending on the severity of the IRIS. Several studies have demonstrated that a low CD4 count at ART initiation increases the risk of IRIS [5,6]. The diagnostic criteria of HBV-related IRIS include worsening liver enzymes after initiating ART, low initial CD4 count, positive virological and immunological markers, absence of drug-induced hepatotoxicity, and medication noncompliance [7]. The molecular mechanism in cases of HBV-related IRIS remains broadly speculative despite multiple assumptions and observations. However, it is proposed to most likely result from monocytes and CD8+ T-cell recruitment to the liver from exaggerated lymphocyte activation and elevated chemokines [7]. In the absence of ART, coinfected patients have higher levels of HBV DNA polymerase, less spontaneous clearance of HBeAg, and lower levels of ALT, compared with patients infected with HBV alone [8]. This suggests that HIV-mediated immune suppression allows for increased HBV replication and decreased clearance by the immune system.
In a case report by Michael et al [7], an acute HBV-IRIS flare occurred in a patient coinfected with HBV/HIV after ART with emtricitabine, tenofovir, and raltegravir and sulfamethoxazole-trim-ethoprim prophylaxis for 2 months. Initially, drug-induced liver injury was suspected and prophylaxis was held, but ART was continued with minimal improvement, and later the HBV-IRIS diagnosis was confirmed by liver biopsy on hospital day 7. The patient’s AST and ALT levels normalized, with an undetectable viral load 3 months after discharge. Fujiko et al [6] described 2 case reports on HBV-related IRIS in HBV/HIV coinfected patients after commencement of ART; diagnosis was confirmed on liver biopsy in both cases, revealing lymphocytes predominance, which was diffusely positive for CD8 and CD3 on immunohistochemical staining. The patients had gradual clinical and laboratory improvement with the continuation of ART. We observed a similar trend in our case, although our patient’s clinical presentation differed from the cases described above.
In many other studies, a transient increase in HBV-DNA levels has been reported in conjunction with clinical hepatitis following the initiation of ART [9–12]. However, continuation of ART during flares is associated with the development of a variety of hepatitis B antibodies, with the clearance of HBsAg and resolution of the hepatitis. Therefore, this further signifies the importance of ART continuation during IRIS flares.
In the present case, the patient developed acute liver injury after 3 weeks of commencement of ART and had all positive virological and immunological markers, suggestive of an acute HBV-IRIS flare; however, the possibility of hepatotoxicity from ART could not be ruled out completely until the liver biopsy was performed and diagnosis was confirmed. The patient had initial laboratory improvement with steroidal treatment and later had complete normalization of liver enzymes at 9 weeks of restarting ART, with a significant reduction in HBV load (110 946 copies/mL) and an undetectable HIV load at 4 weeks and development of HBeAb, suggestive of seroconversion at 4 months after discharge and ART initiation.
The elevated liver enzymes with a high HBV viral load at presentation could have been a reflection of natural alterations in hepatitis disease activity, direct hepatotoxicity from ART and/ or immune restoration (IRIS), or a combination of all. Drug-induced hepatotoxicity and hepatic failure are serious complications that can occur in patients taking ART, and HBV coinfection further increases the risk of hepatotoxicity due to severe hepatic cytolysis by the virus [13].
Chronic HBV infection is associated with significant morbidity and mortality, and coexisting HIV infection can lead to early progression of the disease [14]. The presence of HBeAg, considered as a marker of active viral replication, significantly increases the risk of development of progressive disease, cirrhosis, and, subsequently, hepatocellular carcinoma. Seroconversion, meaning the absence of HBeAg or development of HBeAb, indicates remission and is usually seen after initiating antiviral treatment [15]. Our patient had positive HBeAg during the active flare, and as ART directed against HBV was continued, he subsequently achieved seroconversion. Seroconversion indicates resolution of acute viral activity and improved outcomes in chronic HBV infection but has no clear association or role in diagnosing IRIS. The management of IRIS-related acute HBV flares usually involves the continuation of ART targeting both viruses, as discontinuation could worsen the flare and underlying opportunistic infections and increase the risk of drug resistance. Despite the fact that HIV/HBV coinfected patients can develop HBV-IRIS flares after ART initiation, these cases are often under-reported, and many times an invasive approach, namely liver biopsy for definitive diagnosis, is warranted, as was observed in the present case.
Conclusions
There is limited data on HBV-IRIS flares as a potential cause of acute liver injury after starting ART in coinfected patients. Early recognition and close monitoring during the flare are required to assess for progression and resolution of flares. The significant elevation of liver enzymes necessitates an invasive approach, namely liver biopsy, to differentiate IRIS from an idiosyncratic drug reaction. Management usually entails the continuation of ART targeting both viruses, and steroids are reserved for severe cases of IRIS. HBeAg-positive patients should be evaluated for seroconversion after a flare is resolved.
References:
1.. French MA, HIV/AIDS: Immune reconstitution inflammatory syndrome: A reappraisal: Clin Infect Dis, 2009; 48; 101-7
2.. Sereti I, Rodger AJ, French MA, Biomarkers in immune reconstitution inflammatory syndrome: Signals from pathogenesis: Curr Opin HIV AIDS, 2010; 5; 504-10
3.. Murdoch DM, Venter WD, Feldman C, Van Rie A, Incidence and risk factors for the immune reconstitution inflammatory syndrome in HIV patients in South Africa: A prospective study: AIDS, 2008; 22(5); 601-10
4.. Shelburne SA, Hamill RJ, Rodriguez-Barradas MC, Immune reconstitution inflammatory syndrome: Emergence of a unique syndrome during highly active antiretroviral therapy: Medicine (Baltimore), 2002; 81(3); 213-27
5.. Lawn SD, Myer L, Bekker LG, Wood R, Tuberculosis-associated immune reconstitution disease: Incidence, risk factors and impact in an antiretroviral treatment service in South Africa: AIDS, 2007; 21(3); 335-41
6.. Mitsumoto F, Murata M, Kato Y, Hepatitis B virus-related immune reconstitution inflammatory syndrome in two patients coinfected with human immunodeficiency virus diagnosed with a liver biopsy: Intern Med, 2014; 53(18); 2165-70
7.. Rowley MW, Patel A, Zhou W, Immune reconstitution syndrome with initiation of treatment of HBV/HIV co-infection: Activity flare associated with E antigen seroconversion: Ann Hepatol, 2019; 18(1); 220-24
8.. Gilson RJC, Hawkins AE, Beecham MR, Interactions between HIV and hepatitis B virus in homosexual men: Effects on the natural history of infection: AIDS, 1997; 11; 597-606
9.. Mangold C, Hannoun G, Wywiol A, Reactivation of hepatitis B virus replication accompanied by acute hepatitis in patients receiving highly active antiretroviral therapy: Clin Infect Dis, 2001; 32; 144-48
10.. Mastroianni CM, Trinchieri V, Santopadre P, Acute clinical hepatitis B in an HIV-seropositive hepatitis B carrier receiving protease inhibitor therapy: AIDS, 1998; 12; 1939-40
11.. Proia LA, Ngui SL, Kaur S, Reactivation of hepatitis B in patients with human immunodeficiency virus infection treated with combination anti-retroviral therapy: Am J Med, 2000; 108; 249-51
12.. Velasco M, Moran A, Tellez MJ, Resolution of chronic hepatitis B after ritonavir treatment in an HIV-infected patient [letter]: N Engl J Med, 1999; 340; 1765-66
13.. Eholié SP, Lacombe K, Serfaty L, Acute hepatic cytolysis in an HIV-infected patient taking atazanavir: AIDS, 2004; 18(11); 1610-11
14.. , Hepatitis B fact sheet June 24, 2022 Available from: https://www.who.int/news-room/fact-sheets/detail/hepatitis-b
15.. Liaw YF, HBeAg seroconversion as an important end point in the treatment of chronic hepatitis B: Hepatol Int, 2009; 3(3); 425-33
Tables
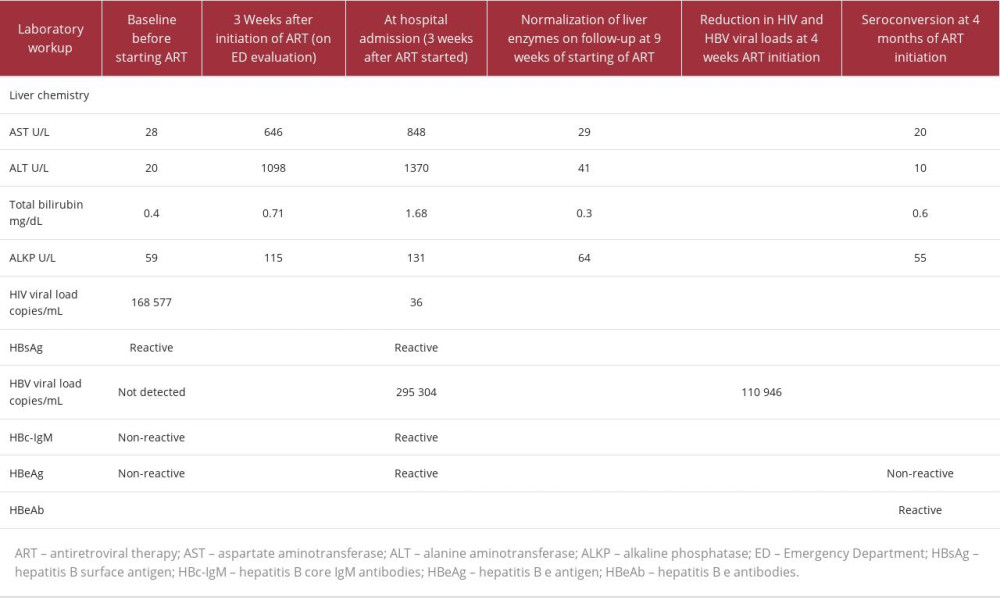 Table 1.. Timeline of development and resolution of acute immune reconstitution inflammatory syndrome-hepatitis B virus flare in a coinfected patient after antiretroviral therapy initiation.
Table 1.. Timeline of development and resolution of acute immune reconstitution inflammatory syndrome-hepatitis B virus flare in a coinfected patient after antiretroviral therapy initiation. Table 1.. Timeline of development and resolution of acute immune reconstitution inflammatory syndrome-hepatitis B virus flare in a coinfected patient after antiretroviral therapy initiation.
Table 1.. Timeline of development and resolution of acute immune reconstitution inflammatory syndrome-hepatitis B virus flare in a coinfected patient after antiretroviral therapy initiation. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








