21 May 2023: Articles 
Renovascular Hypertension with Progressive Atherosclerotic Renal Artery Stenosis in an Elderly Patient with Known Fibromuscular Dysplasia: A Case Report
Challenging differential diagnosis, Rare coexistence of disease or pathology
Jacob AlexDOI: 10.12659/AJCR.939881
Am J Case Rep 2023; 24:e939881
Abstract
BACKGROUND: Atherosclerotic renal artery stenosis (ARAS) and renovascular fibromuscular dysplasia (FMD) are 2 of the most common etiologies of renovascular hypertension. They have different pathophysiologies, risk factors, presentations, and treatment options. However, as our population ages, it can become increasingly common to see patients who previously had FMD who develop ARAS at an advanced age, indicated by recurrent renovascular hypertension.
CASE REPORT: We present a case of a 66-year-old female patient who, in 2007, had presented with uncontrolled hypertension. She underwent magnetic resonance angiography and was found to have bilateral FMD, for which she received balloon angioplasty to a severe lesion on the mid-right renal artery and subsequently had normalization of blood pressures and resolution of symptoms. In 2021 she returned with uncontrolled hypertension while being treated with 3 antihypertensives. Bilateral renal arteriography revealed new severe ostial stenosis of the left renal artery and a patent right renal artery in which balloon angioplasty was performed 14 years ago. Based on the angiographic pattern of this new left RAS, we concluded this lesion was caused by atherosclerosis. The left ostial lesion was treated with a bare-metal stent and the patient was continued on antihypertensive medication and statin; at follow-up 4 months later, her blood pressures had normalized.
CONCLUSIONS: This case features a patient who developed severe ARAS with underlying history of bilateral renal artery FMD. Clinicians need to be aware that in patients with FMD, worsening renovascular hypertension at an advanced age could indicate the development of new hemodynamically significant ARAS. These patients need repeat diagnostic testing and treatment with medial optimization with or without endovascular revascularization in the appropriate clinical setting.
Keywords: Angioplasty, atherosclerosis, Fibromuscular Dysplasia, Hypertension Resistant to Conventional Therapy, Renal Artery Obstruction, Female, Humans, Aged, Hypertension, Renovascular, Renal Artery, Blood Pressure, Hypertension, Angioplasty, Balloon, Antihypertensive Agents
Background
Hemodynamically significant renal artery stenosis (RAS) is generally defined as the narrowing of the renal arteries by >60% [1]. Once there is a reduction in arterial blood flow to the kidney, RAS triggers a cascade of events, starting with activation of the renin-angiotensin-aldosterone-system and culminating in ischemic nephropathy [1]. During this process, patients may develop renovascular hypertension, which remains the most common type of secondary hypertension in elderly patients [2]. Ninety percent of all RAS cases can be attributed to atherosclerosis, with the remaining 10% due to fibromuscular dysplasia (FMD) [3]. The prevalence of atherosclerotic renal artery stenosis (ARAS) increases with age, male sex, smoking, diabetes mellitus, and peripheral arterial disease [4]. FMD, on the other hand, is more prevalent in younger women [5]. Their separate pathophysiologies require different approaches to treatment, involving life-style modifications, medical therapy, and endovascular interventions.
Here, we present a unique case of a female patient with recurrent, severe renovascular hypertension; arteriography revealed severe ostial ARAS of the left renal artery, which was treated with stenting. Fourteen years before the current presentation, she was diagnosed with renovascular hypertension secondary to FMD, which was treated with balloon angioplasty. Clinicians must be aware that as our population ages, severe ARAS can present as recurrent renovascular hypertension in patients with previously-treated renal artery stenosis from FMD.
Case Report
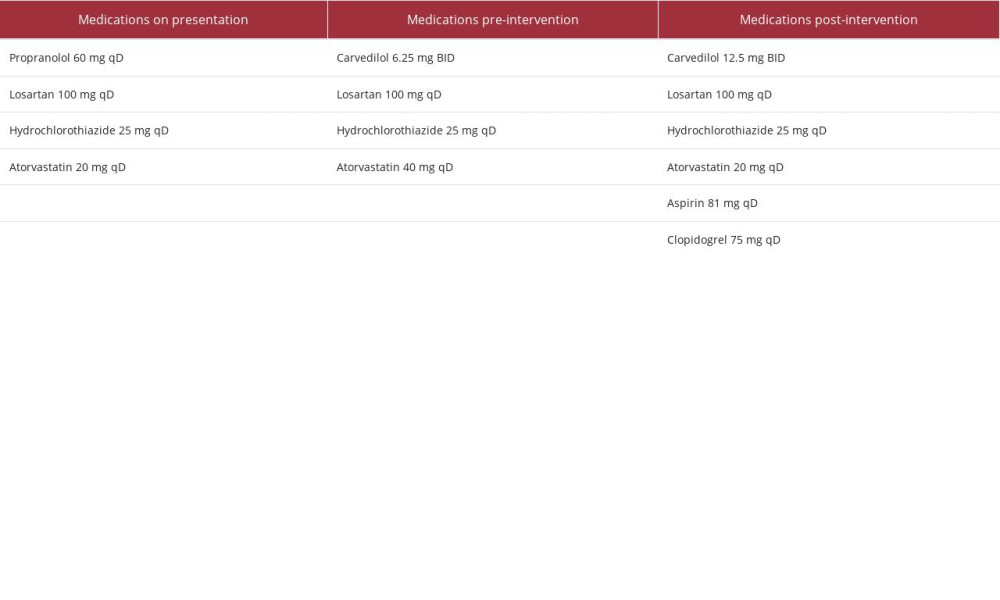
Our patient was a 66-year-old woman with a past medical history of hypertension, hyperlipidemia, and bilateral FMD treated with balloon angioplasty in 2007 (Figure 1). She presented to the cardiology clinic in 2021 with recurrence of hypertension. Review of systems was positive for frontal headaches, and she denied any illicit substance abuse, tobacco smoking, anginal symptoms, or claudication. Assessment of vital signs showed a blood pressure of 162/78 mmHg on the left arm and 161/80 on the right arm, heart rate of 75 beats per minute, and body mass index (BMI) 27 kg/m2. She had normal respiratory exam results, heart sounds were normal with a nondisplaced point of maximal impulse, and no abdominal bruits were noted. At the time of presentation in 2021, she was on a 3-drug antihypertensive regimen of propranolol 60 mg extended-release qD, losartan 100 mg po qD, and hydrochlorothiazide 25 mg po qD. She followed the Dietary Approaches to Stop Hypertension (DASH) diet for lowering blood pressure [6]. She was also on atorvastatin 40 mg for dyslipidemia. Her laboratory results included: creatinine 0.9 mg/dL (0.7–1.2), BUN 20 mg/dL (8–20), K 3.4 mEq/L (3.5–5.4), glucose 101 mg/dL (70–200), total cholesterol 132 mg/dL(140–199), HDL 32 mg/dL (40–90), triglycerides 155 mg/dL (30–149), and LDL 69 mg/dL (60–99). A bilateral renal ultrasound in 2015 showed a slightly atrophic left kidney with cortical thinning and increased cortical echogenicity, while her right kidney parameters were normal. She had normal electrocardiogram and transthoracic echocardiogram results. Bilateral renal ultrasound was repeated in 2021, which showed a left kidney with increased cortical echogenicity with diffuse cortical thinning, and normal right kidney parameters. Her antihypertensive regimen was suboptimal, so propranolol was discontinued in favor of carvedilol 6.25 mg BID. Losartan 100 mg po qD, and hydrochlorothiazide 25 mg po qD were continued. The progression of cortical thinning in the left kidney and return of uncontrolled hypertension made clinicians suspicious of RAS. A screening duplex ultrasound was indicated at this point; however, this was not completed, which is a limitation of this case report. Given the patient had prior FMD with angioplasty, the index of suspicion for restenosis or a new stenotic lesion was high. The decision was made to proceed with bilateral renal angiography, as it remains the criterion standard for diagnosis of RAS and has the option of endovascular intervention.
Angiography revealed a patent site of the previous FMD where balloon angioplasty was performed 14 years ago (Figure 2A). The left renal artery showed severe ostial stenosis, diffuse proximal vessel disease with greater than 80% stenosis, and eccentric calcification, highly suggestive of atherosclerosis (Figure 2A). The previously known mid-distal segment of fibromuscular disease appeared similar, without any progression. The left ostial lesion was treated with balloon angioplasty followed by placement of a Herculink® bare-metal stent (Figure 2B, 2C). The intervention had good angiographic results, with 0% residual stenosis and no complications. Her antihypertensive regimen of carvedilol 6.25 mg BID, losartan 100 mg po qD, and hydrochlorothiazide 25 mg po qD was continued along with atorvastatin (Table 1). On follow-up 4 months after the procedure, her blood pressure remained significantly improved, measuring 146/78 mmHg on the left arm and 140/76 mmHg on the right arm, and she remained on the antihypertensives at the same doses indicated above. Laboratory investigations showed creatinine 0.87 mg/dL (0.7–1.2) and BUN 18 mg/dL (8–20).
Discussion
FMD is a noninflammatory, nonatherosclerotic disease of the fibrous and intimal layers of the arterial vessel, with proposed genetic and hormonal pathogenesis [5]. The renal arteries are involved in 60–75% of patients [5]. The majority of cases are incidentally found in asymptomatic young women aged 15–50 years [5]. Historically, FMD is classified based on the predominant arterial layer affected; intima, media, and adventitia found histopathologically. The definitive diagnosis of FMD is made on angiography; thus, there has been a shift to the radiographic classification of multifocal and unifocal based on the AHA 2014 scientific statement [5]. Multifocal describes the classic “string of beads” found in the media type, which accounts for the vast majority of cases. The typical findings of multiple areas of stenosis and post-stenotic dilations predominantly in the middle to distal portion of the renal artery can be seen in our patient’s right and left renal arteries (Figure 1A). ARAS is responsible for up to 90% of renal artery stenosis cases [3]. In contrast to FMD, the prevalence of ARAS increases with age (>50 years) and with the presence of additional cardiovascular risk factors [3]. The pathogenesis involves atherosclerotic plaque buildup leading to progressive stenosis of the renal arteries; this is a systemic process and usually occurs simultaneously in other arterial beds. Lesions of the ARAS commonly involve the ostium and proximal third of the main renal artery and perirenal aorta, as seen in our patient (Figure 2A) [3]. Another possible differential for the left renal artery lesion is a dissection. Arteriopathies such are FMD are prone to dissection, most commonly found in men at the extracranial carotid, renal, and intracranial arteries [7]. The exact prevalence of FMD-related dissection is unknown due to lack of population-based studies [8]. FMD can be investigated with duplex ultrasound, but stenosis severity cannot be determined as in ARAS, where it is used as the first-line method to screen for significant (>60%) stenosis [5,9]. Angiography has the highest special resolution (<0.1 mm), and as such remains the criterion standard for diagnosing renovascular FMD and ARAS, and remains the only way to detect branching vessel FMD [5,9]. Angiography also offers the possibility of measuring the pressure gradient across the lesion, which is more useful in determining hemodynamic significance in moderate ARAS [9]. CTA and MRA both have high sensitivity (64–100% and 94–97%, respectively) and specificity (92–98% and 85–93%, respectively) for diagnosing FMD and ARAS; CTA is more readily available and has a better special resolution (0.5 mm) than MRA (1–2 mm) [5,9]. Although there is currently no consensus on management, options include antiplatelet therapy and stent placement for flow-limiting lesions [8]. Our patient showed evidence of end-organ damage indicated by new cortical thinning of the left kidney, which further supported an endovascular intervention.
Given the differing pathogeneses of FMD and ARAS, it is unlikely that the preexisting bilateral FMD lesions were pathologically related to the development of ARAS in the left kidney. Our patient was lost to follow-up for more than a decade; we suspect progression of disease along with potential suboptimal management of cardiovascular risk factors led to the development of severe ARAS of the left renal artery. Our literature review revealed 2 cases with ipsilateral ARAS and renal artery FMD [10,11]. The first case was of a 71-year-old woman with resistant hypertension, found to have ARAS of the proximal right renal artery and distal right renal artery FMD. The second case was of a 77-year-old woman, also with resistant hypertension, who was found to have ARAS of the ostial right renal artery and a mid-lesion, consistent with FMD [11]. Both patients likely had undiagnosed FMD earlier in life and developed hemodynamically significant ARAS later in life due to atherosclerotic risk factors and aging. As our population ages, it may become more common to find atherosclerotic lesions in various vascular beds in patients with preexisting subclinical vascular conditions.
RAS from FMD and atherosclerosis have distinct treatment strategies. Clinical trials have almost exclusively focused on the treatment of ARAS; to date, no randomized clinical trials have compared medical therapy to revascularization for FMD. Current AHA guidelines recommend revascularization in patients with RAS from FMD in the following scenarios: resistant hypertension on a 3-drug regimen, new-onset hypertension, renal artery aneurysm or dissection, and preservation of renal function in severe renal artery stenosis [5]. Revascularization in FMD is recommended with balloon angioplasty, as lesions have minimal arterial recoil, reflecting a differing pathophysiology from ARAS [5]. Meta-analyses by Trinquart et al showed 40–50% cure rates of hypertension in RAS from FMD, with balloon angioplasty being the preferred intervention [12]. In our patient, the right renal artery FMD lesion remained patent and free of significant disease 10 years after balloon angioplasty (Figure 2A).
Revascularization for ARAS remains a more controversial topic. The most significant trials were revascularization vs medical therapy for renal artery stenosis (ASTRAL) in 2009 and Cardiovascular Outcomes in Renal Atherosclerotic Lesions (CORAL) in 2014; neither showed a benefit from revascularization for ARAS [4,13]. ASTRAL has received 2 major criticisms: (1) lack of enrollment of high-risk patients and 40% of patients had renal artery stenosis less than 70% [13], and (2) this trial allowed physicians to enroll patients that they were uncertain would have a clinical benefit from revascularization, effectively introducing recruitment bias to the study. CORAL tried to address the flaws in ASTRAL by recruiting patients with a systolic blood pressure of at least 155 mmHg and GFR less than 60 mL/min/1.72 m2 [4]. However the systolic blood pressure criteria were removed partway through the trial due to low enrollment, allowing patients without hypertension to be enrolled [4]. Despite attempting to recruit high-risk patients in CORAL, the average renal artery stenosis was 67%, and most patients were initially only on a 2-drug antihypertensive regimen [4]. Patient like the one in this case report, with resistant hypertension on a 3-drug antihypertensive regimen and severe ARAS lesion, were not adequately represented in this trial. The ACC/AHA and ESC recommend medical management as the first-line treatment in all RAS (Class Ia) [9,14]. ACC/AHA/SCAI 2018 appropriate use criteria evaluated stent placement in hemodynamically significant ARAS lesions [15]. The recommendations on appropriateness of stent placement were: appropriate for declining renal function, bilateral RAS, solitary kidney with RAS, and flash pulmonary edema, and the recommendation of “may be appropriate” for resistant hypertension [15].
Conclusions
This case features a patient who developed severe ARAS with an underlying history of bilateral renal artery FMD; it highlights the differing angiographic and pathophysiologic presentations of FMD and ARAS. Clinicians need to understand that in patients with FMD, worsening renovascular hypertension at an advanced age could indicate the development of hemodynamically significant ARAS. The first step in screening for ARAS is duplex ultrasound of the renal artery, which can identify significant (>60%) ARAS. In the case of FMD, CTA and MRA are better diagnostic modalities, and angiography remains the criterion standard for diagnosing renovascular FMD and ARAS. Renovascular FMD has clear indications for revascularization via balloon angioplasty in specific clinical settings. Prior landmark trials have not shown a benefit of revascularization in ARAS. However, the study populations in these trials did not adequately represent severe ARAS. Therefore, clinicians must pay close attention to identifying the patients most likely to benefit from revascularization. Future randomized trials are needed to provide more robust evidence for treating these patients.
Figures
References:
1.. Courand P-Y, Dinic M, Lorthioir A, Resistant hypertension and atherosclerotic renal artery stenosis: Effects of angioplasty on ambulatory blood pressure. A retrospective uncontrolled single-center study: Hypertension, 2019; 74; 1516-23
2.. Whelton PK, Carey RM, Aronow WS, 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines: Hypertension, 2018; 71(6); e13-115
3.. Safian RD, Textor SC, Renal-artery stenosis: N Engl J Med, 2001; 344; 431-42
4.. Cooper CJ, Murphy TP, Cutlip DE, Stenting and medical therapy for atherosclerotic renal-artery stenosis: N Engl J Med, 2014; 370; 13-22
5.. Olin JW, Gornik HL, Bacharach JM, Fibromuscular sysplasia: State of the science and critical unanswered questions: A scientific statement from the American Heart Association: Circulation, 2014; 129; 1048-78
6.. Appel LJ, Moore TJ, Obarzanek E, A clinical trial of the effects of dietary patterns on blood pressure: N Engl J Med, 1997; 336; 1117-24
7.. Kadian-Dodov D, Gornik HL, Gu X, Dissection and aneurysm in patients with fibromuscular dysplasia: J Am Coll Cardiol, 2016; 68; 176-85
8.. Gornik HL, Persu A, Adlam D, First International Consensus on the diagnosis and management of fibromuscular dysplasia: Vasc Med, 2019; 24; 164-89
9.. Aboyans V, Ricco J-B, Bartelink M-LEL, 2017 ESC Guidelines on the Diagnosis and Treatment of Peripheral Arterial Diseases, in collaboration with the European Society for Vascular Surgery (ESVS): Document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteriesEndorsed by: The European Stroke Organization (ESO)The Task Force for the Diagnosis and Treatment of Peripheral Arterial Diseases of the European Society of Cardiology (ESC) and of the European Society for Vascular Surgery (ESVS): Eur Heart J, 2018; 39; 763-816
10.. Jayawardene S, Reidy J, Scoble J, Ipsilateral atherosclerotic and fibromuscular renal artery stenosis: Lancet, 2000; 356; 2138
11.. Aqel R, Gupta R, Zoghbi G, Coexistent fibromuscular dysplasia and athero-sclerotic renal artery stenosis: J Invasive Cardiol, 2005; 17; 572-73
12.. Trinquart L, Mounier-Vehier C, Sapoval M, Efficacy of revascularization for renal artery stenosis caused by fibromuscular dysplasia: A systematic review and meta-analysis: Hypertension, 2010; 56; 525-32
13.. Wheatley K, Ives N, Gray R, Revascularization versus medical therapy for renal-artery stenosis: N Engl J Med, 2009; 361; 1953-62
14.. Anderson JL, Halperin JL, Albert NM, Management of patients with peripheral artery disease (compilation of 2005 and 2011 ACCF/AHA guideline recommendations): A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines: Circulation, 2013; 127; 1425-43
15.. Bailey SR, Beckman JA, Dao TD, ACC/AHA/SCAI/SIR/SVM 2018 appropriate use criteria for peripheral artery intervention: J Am Coll Cardiol, 2019; 73; 214-37
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133