24 May 2024: Articles 
Relationship Between Brugada Syndrome and Ischemic Stroke: A Case Report and Comprehensive Literature Review
Rare coexistence of disease or pathology
María Camila Arango-Granados12ABCDEF*, Valentina Muñoz-Patiño3CDEF, Juan Camilo Morales-Perez2ABCDEF, María Juliana Reyes-Cardona4CDEF, Julián Alejandro RivillasDOI: 10.12659/AJCR.943506
Am J Case Rep 2024; 25:e943506
Abstract
BACKGROUND: Brugada syndrome is characterized by specific electrocardiographic changes predisposing individuals to ventricular arrhythmias and sudden cardiac death. Cases of coexisting Brugada syndrome and ischemic stroke are seldom documented, and an underlying pathophysiological link is yet unknown. This article presents a case in which a patient exhibited both Brugada syndrome patterns and an ischemic stroke, prompting a comprehensive literature review to explore the potential association between Brugada syndrome and ischemic stroke.
CASE REPORT: A 49-year-old man, previously healthy, was admitted to the hospital after being discovered unconscious at his workplace. Physical exam showed low oxygen saturation, fever, and abnormal neurological findings. Head computed tomography revealed a significant posterior circulation ischemic stroke. An electrocardiogram revealed Brugada syndrome type II initially, progressing to type III pattern. Despite efforts, the patient’s condition rapidly deteriorated, leading to death within 24 hours. As far as we’re aware, Brugada patterns following a posterior circulation ischemic stroke have only been documented in 1 other instance, in which the patient was also diagnosed with atrial fibrillation.
CONCLUSIONS: Both our literature review and the presented case indicate that Brugada patterns may coexist with and even be associated with ischemic stroke. More extensive research is required to shed light on this potential association. The question of whether Brugada syndrome is a precursor to or a result of ischemic stroke remains unanswered. We propose that patients with ischemic stroke should undergo an evaluation for electrocardiographic signs indicative of Brugada syndrome, particularly if no clear causes, like cardioembolism, are evident.
Keywords: Brugada syndrome, Electrocardiography, ischemic stroke, Stroke, Humans, Male, Middle Aged, Fatal Outcome
Introduction
Brugada syndrome is a clinical condition characterized by specific electrocardiographic changes that make individuals susceptible to ventricular arrhythmias, potentially leading to syncope and sudden cardiac death [1]. This condition arises from disruptions in cardiac depolarization and repolarization, without an identifiable structural cause. Mutations within the cardiac sodium channel gene, specifically within the coding region of the SCN5A gene, have been documented as a risk factor [2]. Brugada syndrome primarily affects men, with a clinical prevalence 8 to 10 times higher than in women, typically manifesting in the third to fourth decade of life [3].
Brugada syndrome patterns often remain latent; hence, provocation tests using sodium channel blockers such as ajmaline, procainamide, or flecainide can be employed in such instances to unmask the characteristic pattern [4]. Moreover, patterns can be unmasked by various factors, including fever and metabolic disorders [5–7].
Reports have emerged documenting cases in which patients exhibit both Brugada syndrome and neurological manifestations [8–14]. Particularly, the coexistence of Brugada patterns and ischemic stroke has seldom been documented, and a specific association or underlying pathophysiological connection between the two has not been conclusively established.
We describe a case of a man with Brugada electrocardiogram (ECG) patterns following an extensive posterior circulation ischemic stroke. Considering this, we undertook a comprehensive literature review to explore the potential relationship between Brugada syndrome and ischemic stroke.
Case Report
A 49-year-old male resident of Timba, Cauca in Colombia, a dedicated farmer with no prior medical history, was admitted to the hospital after being found unconscious by his coworkers. The day before, he had complained of back pain without any other known additional symptoms. Upon admission, his vital signs were as follows: blood pressure 112/48 mmHg, heart rate 89 bpm, respiratory rate 18 rpm, oxygen saturation 89%, and temperature 39.9°C. Physical examination revealed a Glasgow Coma Scale score of 5/15, non-reactive dilated pupils (5 mm), clonus in lower limbs, no neck stiffness, profuse secretion mobilization during breathing, and prolonged capillary refill time. Airway protection was maintained through orotracheal intubation. Arterial blood gases indicated mild mixed acidosis (pH: 7.3, pCO2: 42 mmHg, pO2: 83 mmHg, HCO3: 20.7 mmol/L, BE: −5.5 mmol/L), hyperlactatemia (2.4 mmol/L), and mild hypokalemia (3.3 mmol/L). Blood cultures were obtained, and empirical antibiotic coverage with piperacillin-tazobactam was initiated. A plain head computed tomography (CT) scan (Figure 1) was performed, revealing an extensive posterior fossa ischemic stroke without signs of hemorrhagic transformation and strong suggestion of basilar artery obstruction.
Additional tests revealed a systemic inflammatory response, acute kidney injury (Cr 1.36 mg/dL, BUN 22 mg/dL), and rhabdomyolysis (CPK 5137 U/L). Laboratory testing of the orotracheal secretion for microbial pathogens revealed Gram-positive cocci in pairs and scant Gram-negative bacilli. A molecular panel detected
While investigating potential causes of the ischemic stroke, a 12-lead ECG initially revealed a Brugada syndrome type II pattern (Figure 2). Later, a follow-up ECG was performed with pre-cordial leads repositioned to the upper intercostal spaces, and displayed a Brugada syndrome type III pattern (Figure 3). The patient’s condition rapidly deteriorated and his neurological prognosis was ominous; he died 24 hours after admission. Given the high-risk electrocardiographic findings for sudden cardiac death, family members were advised to undergo electrocardiographic screening and assessments by electrophysiologists.
Discussion
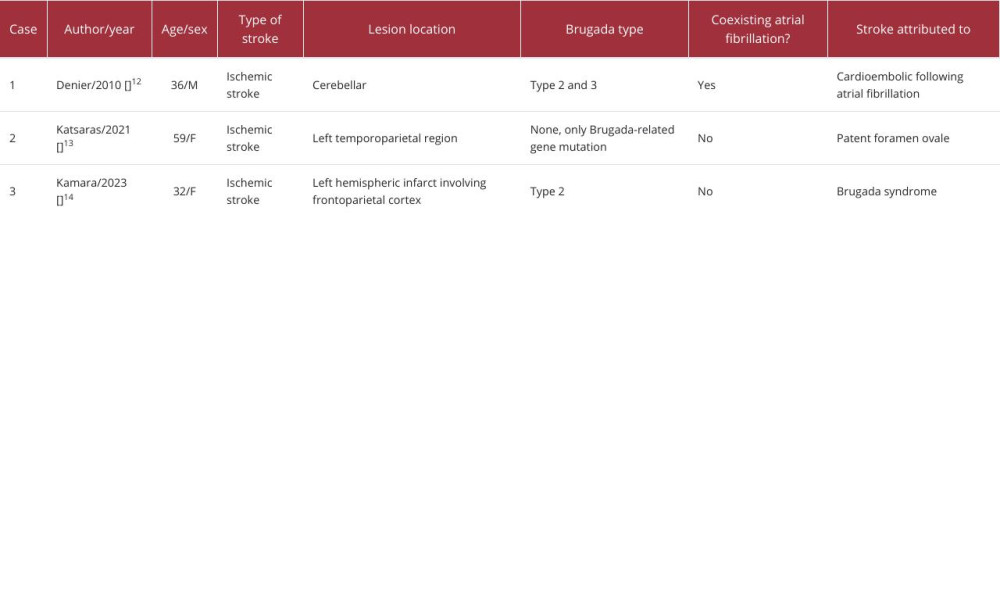
Brugada syndrome is a clinical disorder characterized by distinct electrocardiographic changes predisposing individuals to ventricular arrhythmias [1]. While many individuals with Brugada syndrome are asymptomatic, leading to underdiagnosis, symptomatic patients often experience syncopal episodes or cardiac arrests due to ventricular arrhythmias [15]. Remarkably, 23% of sudden cardiac death cases are preceded by syncopal episodes [3]. There have been reports of patients presenting with both Brugada syndrome and neurological symptoms [8–14]; among these, some of the patients experienced an ischemic stroke (Table 1).
Brugada syndrome presents 3 distinct electrocardiographic patterns: type I features a descending ST-segment elevation of ≥2 mm with negative T waves in multiple right precordial leads. Type II shows similar ST-segment elevation with a positive or biphasic T wave, creating a saddleback appearance. Type III displays ST-segment elevation <1 mm, either descending or saddleback. Although all 3 patterns can be observed in Brugada syndrome, even in the same patient at different times, only type I is considered diagnostic of the disease [16].
Electrocardiographic patterns in Brugada syndrome can often remain latent, necessitating provocative drug tests with sodium channel blockers, such as ajmaline, procainamide, or flecainide, to reveal them [4]. Given the limitations of standard electrocardiograms, new diagnostic strategies have been proposed. Placing right precordial leads in upper intercostal spaces (third or even second intercostal space) has demonstrated increased sensitivity in both baseline electrocardiograms and after sodium channel blocker administration [17].
In this scenario, our case highlights the patient’s shift from a Brugada ECG type II pattern to a type III pattern following the repositioning of the precordial leads. According to the current definition, the diagnostic criteria for Brugada syndrome are confined to type I morphology alone, thereby excluding types II and III ECG patterns as definitive Brugada syndrome diagnoses. Given the patient’s critical condition, we refrained from conducting a provocative test with antiarrhythmic drugs or pursuing genetic analysis. Unfortunately, genetic confirmation of the diagnosis was not feasible for the patient or his family members. Nonetheless, this electrocardiographic observation raised our awareness of a potential link between Brugada syndrome and ischemic stroke.
Several clinical conditions can unveil a Brugada pattern on the ECG. For example, fever can diminish sodium influx due to increased temperatures, potentially triggering such patterns [5]. Likewise, metabolic disorders, including hypokalemia, hyperkalemia, and metabolic acidosis, have been linked to the emergence of Brugada syndrome patterns on the ECG [6,7]. Notably, when Brugada patterns were detected in our patient, both fever and electrolyte imbalances had already been addressed, casting doubt on their roles as contributing factors.
Extensive neurological alterations have also been cited as potential triggers for revealing Brugada syndrome [18,19]. Brugada pattern type I elevation increases with parasympathetic stimulation due to the slowed ventricular repolarization typical of these states but decreases or disappears with sympathetic stimulation. This variability indicates the autonomic nervous system’s possible role in Brugada syndrome pathogenesis and arrhythmogenesis [18,19].
Post-stroke, around 39% of patients with no prior cardiac history exhibit arrhythmias. These predominantly include supraventricular tachyarrhythmias and atrial fibrillation (AF) during the acute phase, underscoring the significance of cardiac monitoring following a stroke [20,21]. In our literature review, we identified 3 documented cases that established an association between Brugada syndrome and stroke, among which 1 specifically documented the identification of Brugada syndrome post-stroke. In this case, a 36-year-old man initially presented with vertigo and horizontal nystagmus. MRI findings revealed a recent cerebellar infarction, and the ECG displayed both AF and type II and III Brugada patterns. Subsequent examinations excluded other cardioembolic sources [12]. While it is plausible that the ischemic stroke triggered the Brugada pattern, it is imperative not to dismiss the possibility that it might have been the Brugada pattern that served as the precursor to the stroke.
Brugada syndrome has been associated with an elevated risk of AF, with reported incidences ranging between 9–39%. Abnormal atrial conduction may serve as an electrophysiological basis for the onset of AF in individuals with Brugada syndrome [22]. Brugada syndrome patients with AF carry a heightened risk of thromboembolic events [23]. A study involving 671 patients with Brugada syndrome found that the incidence of cardioembolic stroke in those with both Brugada syndrome and AF was 13.9%. Of these events, 54.5% were classified as strokes, while 45.5% were transient ischemic attacks [23].
Unlike the aforementioned case, our patient did not exhibit concurrent AF upon admission. While we cannot completely dismiss the potential for paroxysmal AF, this observation compels us to consider whether a substantial link between Brugada syndrome and stroke truly exists.
The second case involves a Brugada-related gene mutation in a patient with congenital absence of the right coronary artery and a patent foramen ovale. The patient underwent genetic evaluation due to family history of Brugada syndrome, with one of her sons dying during sleep at the age of 35 years, while the other son had an implantable cardioverter-defibrillator. The patient suffered a stroke, and while Brugada syndrome is associated with AF and increased stroke risk, the absence of documented AF led to the conclusion that the stroke was likely related to the patent foramen ovale [13]. In contrast, the third case features a 32-year-old woman exhibiting an atypical manifestation of type II Brugada syndrome, characterized by seizures and stroke. In this instance, the occurrence of the latter 2 events was attributed to a potential complication of Brugada syndrome [14].
These atypical presentations underscore the importance of recognizing and comprehending variation in Brugada syndrome manifestations, emphasizing the essential role of a multidisciplinary approach. Given these findings, we recommend evaluating patients with ischemic stroke for electrocardiographic indications of Brugada syndrome, especially when no evident causes such as cardioembolism are present. It is worth noting that the association between the Brugada ECG pattern and ischemic stroke may also be coincidental, as there is no evidence of a clear correlation between these 2 clinical features.
Lastly, in an effort to determine a potential relationship between Brugada syndrome and other neurological conditions, notable pathophysiological links were found in the literature. Studies have highlighted the concurrent presence of idiopathic epilepsy in Brugada syndrome patients. This coexistence is bolstered by genomic research pinpointing genetic mutations, especially those in voltage-dependent ion channels that are expressed in both cardiac and neural tissues [8]. Moreover, hidden Brugada syndrome patterns in epileptic patients with a genetic inclination toward Brugada syndrome might be revealed when they are treated with specific antiepileptic drugs, such as carbamazepine and lamotrigine, which impact voltage-dependent channels [8,9]. To date, 2 cases have documented type I Brugada syndrome patterns in patients with multiple sclerosis. The authors of these studies propose that autonomic nervous system dysfunction, a known occurrence in multiple sclerosis patients, might be the sole anomaly linked to this ECG observation [10,11].
Conclusions
In conclusion, both our literature review and the case we presented suggest that Brugada patterns can coexist with, and perhaps even be related to, ischemic stroke. However, more robust research is essential to further elucidate this potential relationship and its underlying mechanisms. The question of whether Brugada syndrome is a precursor to, or a result of, ischemic stroke remains open. Given the inherent variability of Brugada syndrome patterns within individual patients and the heightened risk of sudden cardiac death, the importance of post-stroke ECG monitoring becomes evident, as it can detect this and other cardiac sources of ischemic stroke. Furthermore, we propose that patients with ischemic stroke should undergo an evaluation for electrocardiographic signs indicative of Brugada syndrome, particularly if no clear causes, like cardioembolism, are evident. Lastly, considering the heightened risk to family members, early screening and timely intervention are crucial when Brugada syndrome is detected in any patient.
Figures
References:
1.. Brugada P, Brugada J, Right bundle branch block, persistent ST segment elevation and sudden cardiac death: A distinct clinical and electrocardiographic syndrome. A multicenter report: J Am Coll Cardiol, 1992; 20(6); 1391-96
2.. Chen Q, Kirsch GE, Zhang D, Genetic basis and molecular mechanism for idiopathic ventricular fibrillation: Nature, 1998; 392(6673); 293-96
3.. Priori SG, Napolitano C, Gasparini M, Natural history of Brugada syndrome: Insights for risk stratification and management: Circulation, 2002; 105(11); 1342-47
4.. Postema PG, Wolpert C, Amin AS, Drugs and Brugada syndrome patients: Review of the literature, recommendations, and an up-to-date web-site: Heart Rhythm, 2009; 6(9); 1335-41
5.. Antzelevitch C, Brugada R, Fever and Brugada syndrome: Pacing Clin Electrophysiol, 2003; 25(11); 1537-39
6.. Postema PG, Vlaar APJ, DeVries JH, Tan HL, Familial Brugada syndrome uncovered by hyperkalaemic diabetic ketoacidosis: Europace, 2011; 13(10); 1509-10
7.. Swe T, Dogar MH, Type 1 Brugada pattern electrocardiogram induced by hypokalemia: J Family Med Prim Care, 2016; 5(3); 709-11
8.. D’imperio S, Monasky MM, Micaglio E, Brugada syndrome: Warning of a systemic condition?: Front Cardiovasc Med, 2021; 8; 1-16
9.. Omer H, Omer MH, Alyousef AR, Unmasking of Brugada syndrome by lamotrigine in a patient with pre-existing epilepsy: A case report with review of the literature: Front Cardiovasc Med, 2022; 9; 1-14
10.. Gialafos E, Andreadou E, Kokotis P, Brugada sign in a multiple sclerosis patient: Relation to autonomic nervous system dysfunction and therapeutic dilemmas: Int J Cardiol, 2016; 202; 652-53
11.. Ibrahim M, Saint-Croix G, Colombo R, Brugada syndrome caused by autonomic dysfunction in multiple sclerosis: Case Rep Cardiol, 2019; 2019; 3937248
12.. Denier C, Guedj T, Dinanian S, Adams D, Brugada syndrome revealed by vertigo caused by cerebellar infarction: Eur J Neurol, 2010; 17(5); e35-36
13.. Katsaras D, Sanjeev-Kumar BT, Patel B, A 59-year-old woman with familial Brugada syndrome and the c.664C>T variant of the sodium voltage-gated channel alpha subunit 5 (SCN5A) gene, accompanied by congenital absence of the right coronary artery, patent foramen ovale, and ischemic stroke: Am J Case Rep, 2021; 22; e931535
14.. Kamara J, Ponnusamy S, Licenik R, An unusual first presentation of stroke and seizure in a 32-year-old patient with Brugada syndrome type 2 electrocardiogram pattern: Cureus, 2023; 15(9); e44630
15.. Brugada J, Brugada R, Antzelevitch C, Towbin J, Long-term follow-up of individuals with the electrocardiographic pattern of right bundle-branch block and ST-Segment elevation in precordial leads V1 to V3: Circulation, 2002; 105(1); 73-78
16.. Benito B, Brugada J, Brugada R, Brugada P, Brugada syndrome: Rev Esp Cardiol, 2009; 62(11); 1297-315
17.. Sangwatanaroj S, Prechawat S, Sunsaneewitayakul B, New electrocardiographic leads and the procainamide test for the detection of the Brugada sign in sudden unexplained death syndrome survivors and their relatives: Eur Heart J, 2001; 22(24); 2290-96
18.. Shen MJ, Zipes DP, Role of the autonomic nervous system in modulating cardiac arrhythmias: Circ Res, 2014; 114(6); 1004-21
19.. Miyazaki T, Mitamura H, Miyoshi S, Autonomic and antiarrhythmic drug modulation of ST-Segment elevation in patients with Brugada syndrome: J Am Coll Cardiol, 1996; 27(5); 1061-70
20.. Lavy S, Yaar I, Melamed E, Stern S, The effect of acute stroke on cardiac functions as observed in an Intensive Stroke Care Unit: Stroke, 1974; 5(6); 775-80
21.. Kallmünzer B, Schwab S, Cardiac arrhythmia following stroke: Neurol Int Open, 2017; 1; E8-17
22.. Morita H, Kusano-Fukushima K, Nagase S, Atrial fibrillation and atrial vulnerability in patients with Brugada syndrome: J Am Coll Cardiol, 2002; 40(8); 1437-44
23.. de Asmundis C, Mugnai G, Chierchia GB, Abnormally high risk of stroke in Brugada syndrome: J Cardiovasc Med (Hagerstown), 2019; 20(2); 59-65
Figures
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133