18 August 2020: Articles 
A 31-Year-Old Man with COVID-19-Associated Empyema and Lupus Anticoagulant
Unknown etiology, Unusual clinical course, Challenging differential diagnosis, Diagnostic / therapeutic accidents, Unusual setting of medical care
Keerthi Yarlagadda1ABCDEF*, Kaihong Mi2ACDEF, Selin Sendil1EF, Connie L. Koons3E, Saketram Komanduri1EF, John T. Cinicola1ADOI: 10.12659/AJCR.926623
Am J Case Rep 2020; 21:e926623
Abstract
BACKGROUND: COVID-19 was declared a pandemic in March 2020 in the United States. It has been associated with high mortality and morbidity all over the world. COVID-19 can cause a significant inflammatory response leading to coagulopathy and this hypercoagulable state has been associated with worse clinical outcomes in these patients. The published data regarding the presence of lupus anticoagulant in critically ill COVID-19-positive patients is limited and indicates varying conclusions so far.
CASE REPORT: Here, we present a case of a 31-year-old man who was admitted to the hospital with COVID-19 pneumonia, complicated with superadded bacterial empyema and required video-assisted thoracoscopic surgery with decortication. This patient also had prolonged prothrombin time on preoperative labs, which was not corrected with mixing study. Further workup detected positive lupus anticoagulant and anti-cardiolipin IgM along with alteration in other coagulation factor levels. The patient was treated with fresh frozen plasma and vitamin K before surgical intervention. He had an uneventful surgical course. He received prophylactic-dose low molecular weight heparin for venous thromboembolism prophylaxis and did not experience any thrombotic events while hospitalized.
CONCLUSIONS: COVID-19 infection creates a prothrombotic state in affected patients. The formation of micro-thrombotic emboli results in significantly increased mortality and morbidity. Routine anticoagulation with low molecular weight heparin can prevent thrombotic events and thus can improve patient outcomes. In patients with elevated prothrombin time, lupus anticoagulant/anti-cardiolipin antibody-positivity should be suspected, and anticoagulation prophylaxis should be continued perioperatively for better outcomes.
Keywords: Anticoagulants, COVID-19, Lupus Coagulation Inhibitor, Antifibrinolytic Agents, Betacoronavirus, COVID-19, Cardiolipins, Chest Tubes, Coronavirus Infections, Empyema, Pleural, Immunoglobulin M, International Normalized Ratio, Pandemics, Partial Thromboplastin Time, Plasma, Pneumonia, Viral, Prothrombin Time, SARS-CoV-2, Tomography, X-Ray Computed, venous thromboembolism, Vitamin K
Background
Corona Virus Disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus-2 (SARS-COV-2), is a global problem, with the number of cases increasing exponentially since the outbreak in December 2019 [1]. Although the initial cases were detected in the city of Wuhan, Hubei Province, Central China, COVID-19 was declared as a pandemic globally in March 2020. Uncertain pathogenesis along with a lack of vaccination and targeted medications have made the control of disease challenging and caused many deaths all over the world [2]. Treatment thus far as per the Chinese literature with minimal data has been supportive of oxygen therapy as needed, antiviral medications, steroids, empiric antibiotics, continuous renal replacement therapy (CRRT), and intravenous immunoglobulins (IVIG) [3]. Various drugs including Remdesivir have been studied and approved by the FDA in the USA for severe COVID-19. Favilavir has been approved in Japan, while chloroquine and hydroxychloroquine are being studied in clinical trials [4,5].
COVID-19 is believed to activate various complement pathways which lead to microvascular injury associated with procoagulant state and result in thrombotic events [6]. Coagulation dys-function is thought to be one of the major causes of mortality in these patients [7], which varies ranging from COVID-19-associated coagulopathy to disseminated intravascular coagulation (DIC). Abnormal coagulation parameters, including elevated D-dimer and fibrin degradation products, are correlated with a poor prognosis [8]. However, there is not much published data regarding lupus anticoagulant (LA) in these critically ill patient populations, and the published data so far points to varying conclusions [8,9]. Here, we present a case of a 31-year-old man with positive lupus anticoagulant triggered by COVID-19 infection.
Case Report
A 31-year-old man with a past medical history of asthma and achalasia but no known personal or family history of coagulopathy or bleeding disorders presented to the Emergency Department (ED) with complaints of cough and chest pain for 3 days. His initial vital signs were significant for a heart rate of 145/minute and fever of 39.3°C but otherwise he had stable blood pressure (124/64 mmHg), respiratory rate (16/minute), and oxygen saturation of 92% on room air. A physical exam was significant for decreased breath sounds on the left hemithorax but was otherwise noncontributory. Initial bloodwork revealed elevated white blood cell (WBC) 19 900 m/mm3 with left shift, while platelet count and hemoglobin were within normal limits. Liver function tests were mildly deranged, with an elevated ALT (alanine aminotransferase) of 84 U/L, total bilirubin of 1.3 mg/dL, and albumin of 2.6 gm/dl. Given his exposure to multiple COVID-19-positive contacts, he was subsequently tested by reverse transcription-polymerase chain reaction (RT-PCR) for SARS-COV-2 from a nasopharyngeal swab, which later came back positive. We used the Cepheid GenXpert system to detect SARS-COV-2. The initial chest X-ray (Figure 1C) was significant for prominent opacity with mixed density involving the left hemithorax and associated large left-sided ef-fusion, concerning for left lower-lobe pneumonia with parapneumonic effusion
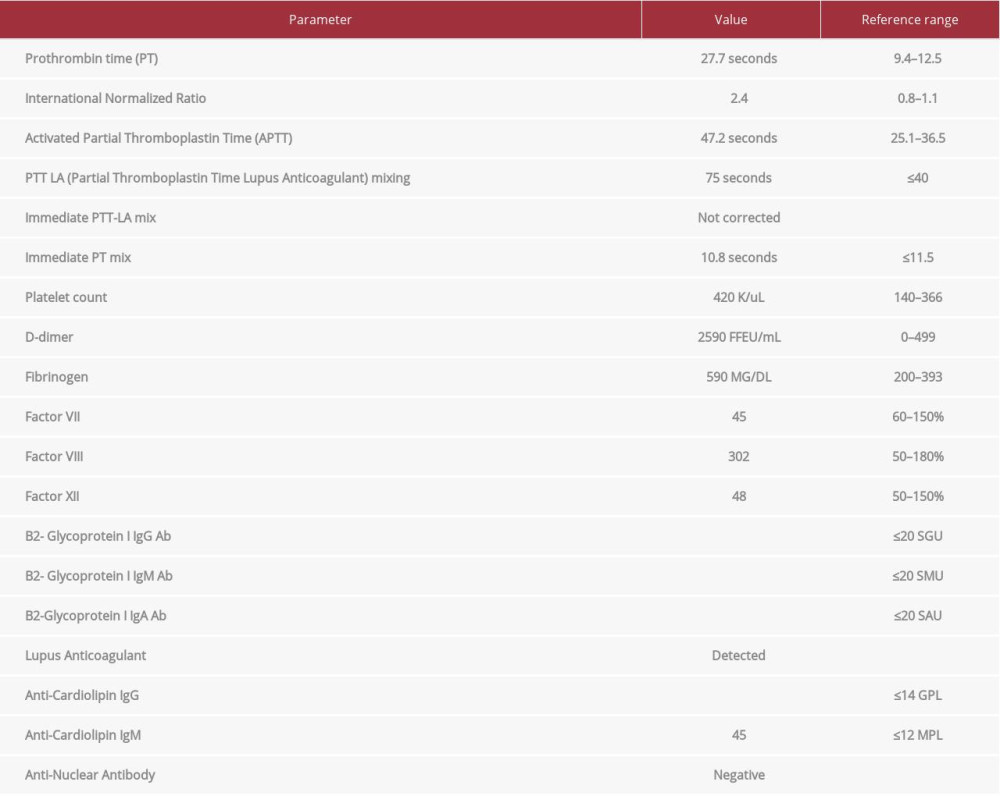
Cardiothoracic Surgery was consulted for potential intervention to treat the empyema. A pre-operative workup revealed that activated partial thromboplastin time (aPTT), prothrombin time (PT), and international normalized ratio (INR) were prolonged (Table 1). Mixing studies showed corrected PT, but not aPTT. His platelet count, D-dimer, and fibrinogen were elevated (Table 1). Hematology was consulted and recommended administration of low molecular weight heparin (LMWH) for venous thromboembolism (VTE) prophylaxis. Immediate aPTT was not corrected in mixing studies, suggesting the presence of immediate-acting inhibitors such as factor-specific inhibitors, LA, or anticoagulation therapies. Intrinsic coagulation factors (II, VIII, IX, X, XI), LA, anti-cardiolipin antibodies, and anti-glycoprotein antibodies were also obtained (Table 1). Fresh frozen plasma (FFP) and vitamin K were given due to elevated PT prior surgery. The patient underwent the procedure but also was started on LMWH in the immediate post-operative period, without any bleeding complications. An updated hematology workup a week later showed normal levels of Factor II, V, IX, and IX, and no evidence of factor-specific inhibitors.
The patient underwent multiple interventions without any hematologic complications. Two chest tubes were placed at the bedside, followed by video-assisted thoracoscopic surgery (VATS) with full decortication. Bronchoscopy and left lower-lobe lavage were also performed. Empyema was suspected to be secondary as a result of aspiration due to the patient’s history of achalasia. He also had esophagogastroduodenoscopy and PEG (percutaneous endoscopic gastrostomy) tube placement concomitantly. Discharge from the chest tube was sent for culture and came back positive for Streptococcus angiosus. He was successfully discharged on a 6-week course of intravenous ertapenem for empyema and recovered without further issues. A repeat chest X Ray was done post chest tube insertion showing resolution of empyema as seen in Figure 1D.
Discussion
Viral illnesses including Hepatitis-C, human immunodeficiency virus (HIV), Epstein–Barr virus (EBV), and Hepatitis-B as per meta-analyses have been associated with an increase in anti-phospholipid, anti-cardiolipin, and anti-β2-glycoprotein-1 antibodies and cause prothrombotic state resulting in thromboembolic events [10,11]. COVID-19 results in acute respiratory distress syndrome (ARDS) through a cytokine storm which thereby increases pulmonary vascular shunting through microvascular injury and thrombus formation [12]. COVID-19-associated coagulopathy (CAC) is characterized by elevated fibrinogen and D-dimer levels and is associated with thrombotic events [13]. There are cases with multiple infarcts in the different parts of the body reported in China with positivity for anti-cardiolipin IgA antibodies as well as anti-β2-glycoprotein I IgA and IgG antibodies [14]. In a study of the Chinese population, treatment with LMWH based on sepsis-induced coagulopathy score and elevated D-dimer levels improved outcomes [15].
Tang et al. reported statistically significant increased mortality correlating with higher D-dimer and fibrin degradation product levels, and longer PT and aPTT. They found that 71.4% of non-survivors and 0.6% of survivors met the criteria of DIC during their hospital stay [8]. In a separate letter to the editor, Tang reported that very few of their patients had positive LA results and concluded that the antiphospholipid antibody does not exist universally in COVID-19 patients [9]. In a recent letter to the editor, Harzallah et al. reported their experience with 56 COVID-19 patients in Mulhouse, France [16]. They found that 45% (n=25) of cases were lupus anticoagulant-positive, whereas only 10% (n=5; 3 also with LA) of the patients were positive for anti-cardiolipin or anti-β2-glycoprotein. The authors emphasized the importance of early anticoagulant therapy, although in acute infections, lupus anticoagulant elevation can be seen, often not requiring therapy. Another interesting report, by Bowles et al., reported LA results performed in 34 patients, and 31 (91%) were positive. When lupus anticoagulant-positive patients’ plasma is mixed with an equal amount of normal plasma, prolongation of aPTT was seen in these specimens. Although heparin was detected in 28 of the 35 specimens that can cause false-positive detection of lupus anticoagulant, the authors concluded that the heparin effect was neutralized due to the assay containing heparinase. The authors also suggested that the use of anticoagulation for the prevention and treatment of VTE should not be limited in COVID-19 patients with prolonged aPTT [17].
As mentioned before, our patient had prolonged PT/INR and aPTT but had no clinically significant bleeding complications. The concern in our case was that the elevated fibrinogen and platelet counts may put the patient at risk of thrombosis. Therefore, hematology recommended LMWH for DVT prophylaxis despite the abnormal PT and aPTT noted with no personal/family history of bleeding disorder. His prolonged aPTT is due to positive lupus anticoagulant and anti-cardiolipin antibody, which will not cause bleeding but possibly increase the risk of thrombosis. Although observed in a small study, there has also been a potential beneficial effect of supplementing dipyridamole in severely ill COVID-19 patients [18]. This suggests that the pathophysiology of thrombosis associated with COVID-19 differs in comparison to non-COVID patients [19]. Further studies should determine the role of lupus anticoagulant in the pathogenesis of COVID-19-associated thrombosis.
Conclusions
In COVID-19 patients with prolonged aPTT but no history of coagulopathy or bleeding, it is reasonable to suspect positive LA, anti-cardiolipin antibody, or anti-β2-glycoprotein-1 antibody. Prolonged aPTT should not be a barrier to surgery. Instead, immediate VTE prophylaxis is recommended perioperatively. Earlier detection of this prothrombotic state and appropriate anticoagulation with LMWH or direct oral anticoagulant, although not studied, may prevent adverse thrombotic events in critically ill COVID-19 patients.
References:
1.. Tu H, Tu S, Gao S, The epidemiological and clinical features of COVID-19 and lessons from this global infectious public health event: Journal of Infection, 2020; 81(1)
2.. Li H, Liu SM, Yu XH, Coronavirus disease 2019 (COVID-19): Current status and future perspectives: Int J Antimicrob Agents, 2020; 55(5); 105951
3.. Ge H, Wang X, Yuan X, The epidemiology and clinical information about COVID-19: Eur J Clin Microbiol Infect Dis, 2020; 39(6); 1011-19
4.. Lai CC, Shih TP, Ko WC, Severe acute respiratory syndrome corona-virus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges: Int J Antimicrob Agents, 2020; 55(3); 105924
5.. Abd El-Aziz TM, Stockand JD, Recent progress and challenges in drug development against COVID-19 coronavirus (SARS-CoV-2) – an update on the status: Infect Genet Evol, 2020; 83; 104327
6.. Magro C, Mulvey JJ, Berlin D, Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: A report of five cases: Transl Res, 2020; 220; 1-13
7.. Song JC, Wang G, Zhang W, Chinese expert consensus on diagnosis and treatment of coagulation dysfunction in COVID-19: Mil Med Res, 2020; 7(1); 19
8.. Tang N, Li D, Wang X, Sun Z, Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia: J Thromb Haemost, 2020; 18(4); 844-47
9.. Tang N, Response to “Lupus anticoagulant is frequent in patients with Covid-19”: J Thromb Haemost, 2020 [Online ahead of print]
10.. Uthman IW, Gharavi AE, Viral infections and antiphospholipid antibodies: Semin Arthritis Rheum, 2002; 31(4); 256-63
11.. Abdel-Wahab N, Talathi S, Lopez-Olivo MA, Suarez-Almazor ME, Risk of developing antiphospholipid antibodies following viral infection: a systematic review and meta-analysis: Lupus, 2018; 27(4); 572-83
12.. Leisman DE, Deutschman CS, Legrand M, Facing COVID-19 in the ICU: Vascular dysfunction, thrombosis, and dysregulated inflammation: Intensive Care Med, 2020; 46(6); 1105-8
13.. Connors JM, Levy JH, COVID-19 and its implications for thrombosis and anticoagulation: Blood, 2020; 135(23); 2033-40
14.. Zhang Y, Xiao M, Zhang S, Coagulopathy and antiphospholipid antibodies in patients with Covid-19: N Engl J Med, 2020; 382(17); e38
15.. Tang N, Bai H, Chen X, Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy: J Thromb Haemost, 2020; 18(5); 1094-99
16.. Harzallah I, Debliquis A, Drénou B, Lupus anticoagulant is frequent in patients with Covid-19: J Thromb Haemost, 2020 [Online ahead of print]
17.. Bowles L, Platton S, Yartey N, Lupus anticoagulant and abnormal coagulation tests in patients with Covid-19: N Engl J Med, 2020; 383(3); 288-90
18.. Liu X, Li Z, Liu S, Potential therapeutic effects of dipyridamole in the severely ill patients with COVID-19: Acta Pharm Sin B, 2020 [Online ahead of print]
19.. Helms J, Tacquard C, Severac F, High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study: Intensive Care Medicine, 2020; 46(6); 1089-98
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133