26 September 2020: Articles 
SARS-CoV-2 Infection and COVID-19 in 5 Patients in Ecuador After Prior Treatment with Hydroxychloroquine for Systemic Lupus Erythematosus
Unusual clinical course, Diagnostic / therapeutic accidents, Unusual setting of medical care, Adverse events of drug therapy, Educational Purpose (only if useful for a systematic review or synthesis)
María Fernanda Zurita1BDEF, Adriana Iglesias Arreaga1BEF*, Adriana A Luzuriaga Chavez1F, Luis Zurita2ABDFDOI: 10.12659/AJCR.927304
Am J Case Rep 2020; 21:e927304
Abstract
BACKGROUND: This case series describes 5 patients with SARS-CoV-2 infection and COVID-19 in Ecuador who had been treated with hydroxychloroquine for systemic lupus erythematosus (SLE) prior to their COVID-19 illness.
CASE REPORT: Case #1 reports a 29-year-old woman who had been treated with 200 mg of hydroxychloroquine per day for 1 year and presented with flu-like symptoms, chest pain, fever, odynophagia, asthenia, dry cough, and chills. Case #2 was a 34-year-old woman whose treatment for SLE included 200 mg of hydroxychloroquine per day since 2017. She arrived at the clinic with a dry cough, asthenia, and myalgias. Case #3 was a 24-year-old woman who had been using 200 mg of hydroxychloroquine per day since 2010. She presented with asthenia, myalgias, headaches, hypogeusia, and anosmia. Case #4 was a 39-year-old woman taking 200 mg of hydroxychloroquine every day for SLE who presented with dyspnea, chest pain, odynophagia, hypogeusia, anosmia, diarrhea, and fever. Case #5 was a 46-year-old woman who had been taking 200 mg of hydroxychloroquine since 2019. She came to our hospital complaining of chest pain, fever, and dyspnea. In all 5 patients, SARS-CoV-2 infection was confirmed with a nasopharyngeal SARS-CoV-2 reverse transcription-polymerase chain reaction (RT-PCR) test using the Cepheid/GeneXpert system.
CONCLUSIONS: All 5 of our patients with SLE who were taking hydroxychloroquine presented with SARS-CoV-2 infection and symptoms of COVID-19. This case series provides support for a lack of prevention of COVID-19 by hydroxychloroquine.
Keywords: COVID-19, hydroxychloroquine, Lupus Erythematosus, Systemic, SARS Virus, COVID-19, COVID-19 Testing, Clinical Laboratory Techniques, Coronavirus Infections, Dyspnea, Ecuador, Emergency Service, Hospital, Fever, Pandemics, Pneumonia, Viral, RNA, Viral, Real-Time Polymerase Chain Reaction, Risk Assessment, Sampling Studies, Treatment Failure, young adult
Background
In December 2019, an outbreak of the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), started in the city of Wuhan, China, rapidly spread throughout the world, and was officially declared a pandemic by the World Health Organization (WHO) on March 11, 2020 [1]. In Latin America, Ecuador is among the countries most affected by SARS-CoV-2, with more than 62 380 confirmed cases as of July 6, 2020, 26% of which were located in the coastal province of Guayas [2].
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease that can affect multiple organs and tissues. For this reason, diagnosis can be complex, relying on clinical and immunological findings and often using the 2019 European League Against Rheumatism (EULAR)/ACR SLE classification criteria [3]. These require the presence of positive antinuclear antibodies, along with other criteria. These additional criteria include 7 clinical (constitutional, hematologic, neuropsychiatric, mucocutaneous, serosal, musculoskeletal, and renal) and 3 immunologic (antiphospholipid antibodies, complement proteins, and SLE-specific antibodies) criteria, with each criterion ranked from 2 to 10. Patients with 10 or more points are classified as having SLE.
Currently, there are no vaccines available against SARS-CoV-2 and many drug strategies have been taken into consideration. One of the drugs that has been in the spotlight is the antimalarial drug hydroxychloroquine, which is commonly used in the treatment of autoimmune diseases. Hydroxychloroquine was first used to treat cutaneous lupus in 1834 due to its anti-inflammatory properties, and since then has been considered among the mainstays of SLE treatment [4]. Hydroxychloroquine has a recommended initial dosage of 400 mg to be taken orally once or twice per day, and a maintenance dose between 200 and 400 mg per day [5].
Due to studies suggesting that hydroxychloroquine has antiviral activity against SARS-CoV-2, the drug was included in treatment strategies against COVID-19 starting July 1, 2020 [6,7]. However, the FDA and others have suggested that it is ineffective and that, in fact, it carries the risk of adverse effects, the most predominant of which is cardiotoxicity (QT prolongation, ventricular arrhythmias, torsades des pointes, and conduction disorders) [8]. Other studies found that hydroxychloroquine did not reduce mortality or improve outcomes in patients hospitalized with COVID-19 and did not help to prevent infection when it was used as post-exposure prophylaxis [9,10].
In recent months, the use of hydroxychloroquine has generated differing opinions and has elicited many inquiries in the medical community. One of these is whether or not patients who have been using hydroxychloroquine for some time, such as SLE patients, are protected against SARS-CoV-2 [11–13]. In this case series, we describe 5 patients from Guayaquil, Ecuador, who were seen for SARS-CoV-2 infection and COVID-19 after prior treatment with hydroxychloroquine for SLE.
Case Report
CASE 1:
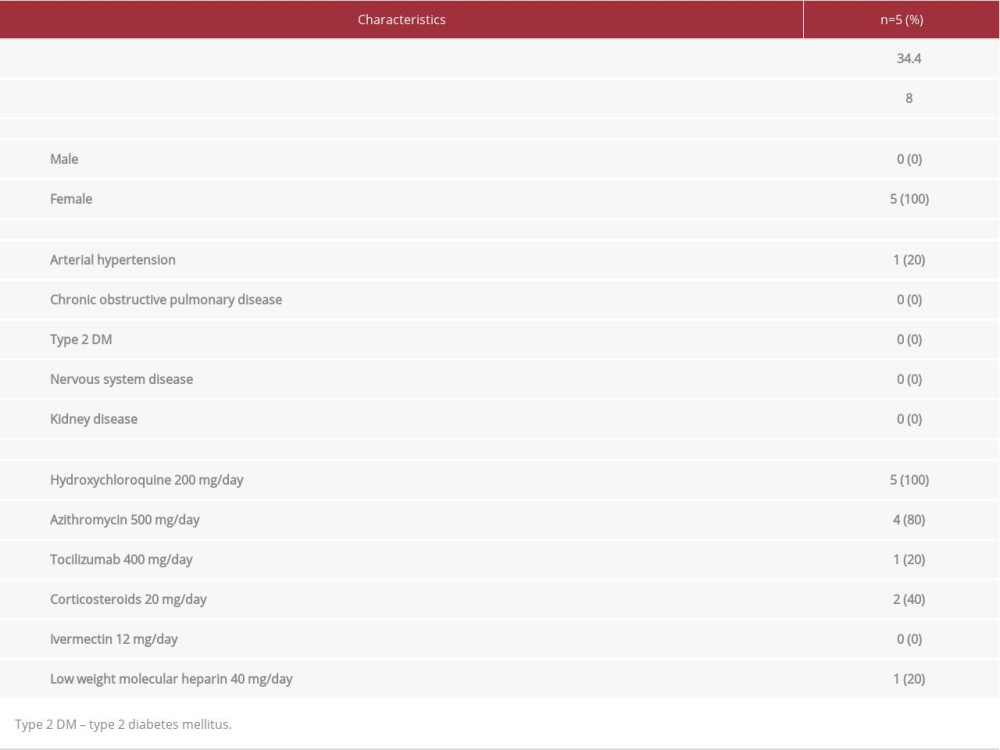
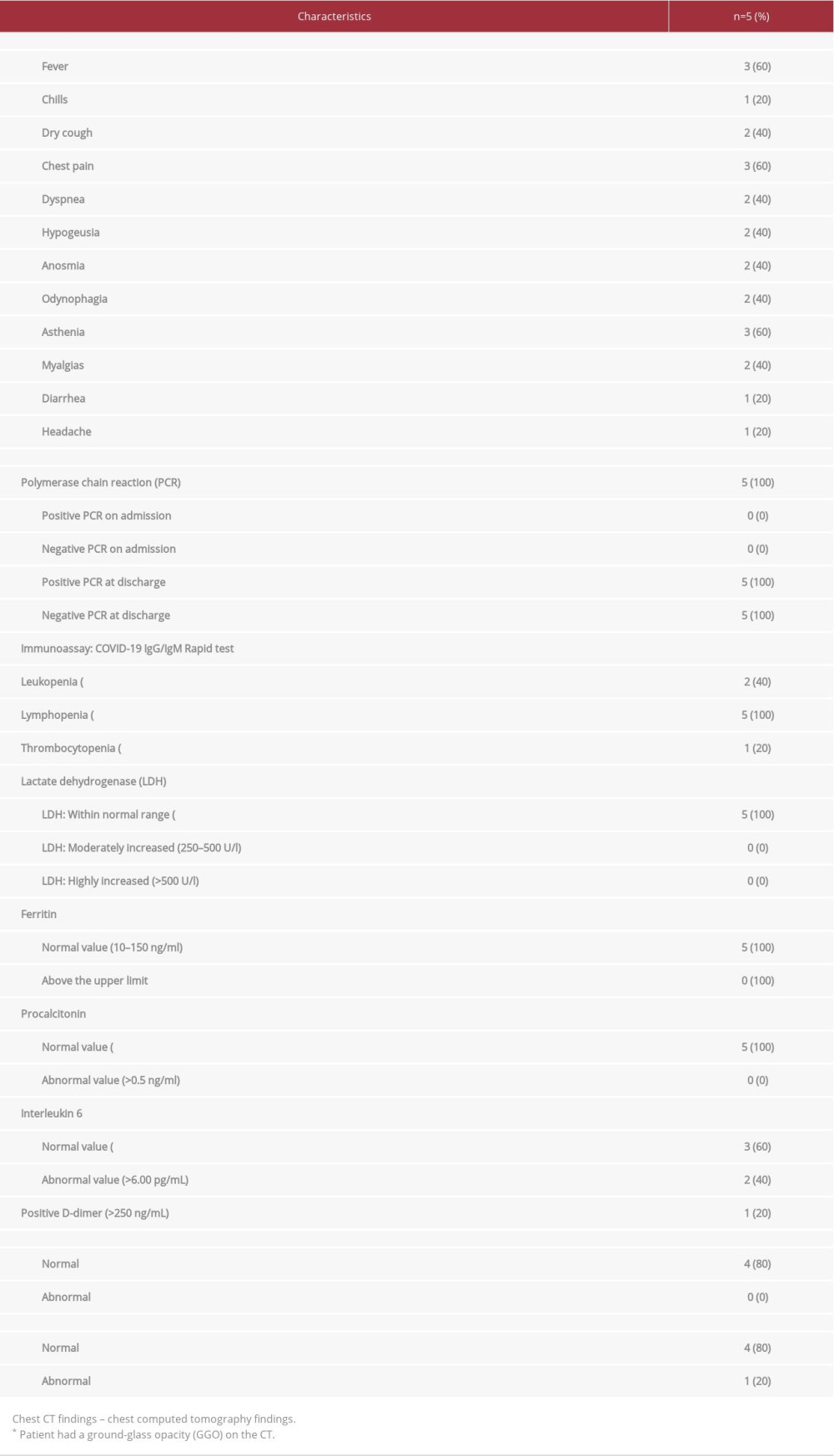
A 29-year-old woman, with a 5-year history of SLE, had been treated with a daily dose of 200 mg of hydroxychloroquine since January, 2019. She presented to the clinic on March 3, 2020 after 4 days of flu-like symptoms, chest pain, fever, odynophagia, asthenia, dry cough, and chills. Her oxygen saturation was 99%. Physical examination was unremarkable and her vitals were within normal limits. Her laboratory findings revealed lymphopenia as the only abnormality. There were no abnormalities on chest X-ray. Testing for SARS-CoV-2 via nasopharyngeal sample and reverse transcription-polymerase chain reaction (RT-PCR) using the Cepheid/GeneXpert system showed positive results. The patient was administered 500 mg per day of azithromycin for 5 days and continued taking hydroxychloroquine, which was already being used to treat her lupus. On March 25, 2020, her RT-PCR test came back negative for SARS-CoV-2.
CASE 2:
A 34-year-old woman with a 12-year history of SLE who had been treated with 200 mg of hydroxychloroquine per day since June 2017 presented to our clinic on April 20, 2020 after 1 week of having a dry cough, asthenia, and myalgias. The patient had been in close contact with a family member who had been infected with SARS-CoV-2. Her oxygen saturation was 99% on room air and clinical examination revealed normal organ findings. Her vital signs and her chest X-ray were normal. Laboratory results showed lymphopenia and thrombocytopenia. SARS-CoV-2 infection was confirmed by RT-PCR testing from nasopharyngeal swabs using the Cepheid/GeneXpert system. This patient did not receive any additional medication to treat the infection besides hydroxychloroquine, which was part of her SLE treatment. Another test against SARS-CoV-2 was performed 2 weeks later and the results were negative.
CASE 3:
A 24-year-old woman with a 4-year history of SLE, who had been taking 200 mg of hydroxychloroquine per day as part of her SLE treatment since June 2010, presented to our clinic on April 5, 2020 after 2 days of asthenia, myalgias, headaches, hypogeusia, and anosmia. Her oxygen saturation was 97% and the physical exam was normal. Vitals were within normal limits and no abnormalities were found on chest X-ray. Laboratory testing was significant for leukopenia and lymphopenia. Testing for SARS-CoV-2 via RT-PCR of a nasopharyngeal sample, using the Cepheid/GeneXpert system, came back positive on April 6, 2020. She received 500 mg per day of azithromycin for 5 days, along with hydroxychloroquine, which was already part of her lupus management. Four weeks after being diagnosed with the infection, her RT-PCR came back negative.
CASE 4:
A 39-year-old woman with a 13-year history of SLE, who had been taking hydroxychloroquine as part of her lupus regimen since July 2015, presented to our clinic on March 29, 2020 with a 3-day history of chest pain, odynophagia, hypogeusia, anosmia, diarrhea, and fever. Upon arrival, the patient was dyspneic and her oxygen saturation was 96%. Her heart rate was 115 beats/min. Given her clinical presentation, a CT scan was performed, which showed ground-glass opacities and consolidations that had recently developed since she had no other pathological images on previous scans. Routine blood test results revealed some abnormalities including lymphopenia, elevated values of D-dimer, and interleukin 6. Testing for SARS-CoV-2 via RT-PCR of a nasopharyngeal sample using the Cepheid/ GeneXpert system showed positive results. Since this patient had a different clinical presentation from the cases mentioned above, in addition to the accepted therapy at the time (500 mg per day of azithromycin for 5 days and continuation of the hydroxychloroquine she was already taking for SLE), she received a daily dose of 40 mg of low molecular weight heparin (LMWH). The LMWH was administered subcutaneously for 10 days as a prophylactic measure due to her elevated levels of D-dimer. She also received 20 mg of prednisone for 10 days and an early dose of 400 mg per day of tocilizumab for 2 days.
It is important to point out that interleukin 6 is believed to be a marker of poor prognosis in COVID-19. It plays a vital role in the development of cytokine storm and in potentially causing acute respiratory distress syndrome (ARDS) [14], which might have contributed to some of our patient’s clinical findings, such as dyspnea. Interestingly, however, she showed immediate improvement and it was not necessary to administer oxygen therapy or ventilator support. She tested negative for SARS-CoV-2 on April 13, 2020.
CASE 5:
A 46-year-old woman with a past medical history of hypertension controlled with medication and a 6-year history of SLE had been taking 200 mg of hydroxychloroquine as a part of her SLE management since January 2019. She came to our clinic on April 28, 2020 with a 2-day history of chest pain, fever, and dyspnea. She had been in close contact with an individual who had been infected with SARS-CoV-2. Her oxygen saturation was 98% and blood pressure was 138/88 mmHg. Her chest X-ray did not show any alterations. The initial laboratory evaluation showed leucopenia, lymphopenia, and mildly elevated levels of interleukin 6. Given her clinical presentation and exposure to an infected person, a nasopharyngeal SARS-CoV-2 RT-PCR test (using the Cepheid/GeneXpert system) was performed, which was positive. To treat the COVID-19, she received 500 mg per day of azithromycin for 5 days, 200 mg of prednisone for 10 days, which was helpful to control her dyspnea and chest tightness, and her regular dose of hydroxychloroquine, which she had already been taking as part of her treatment for SLE. Her laboratory results came back negative for SARS-CoV-2 on May 21, 2020.
It is also important to note that none of the patients required oxygen therapy, nor were any of them admitted to the Intensive Care Unit (ICU). None of the patients developed any serious complications such as ARDS or shock.
Discussion
Hydroxychloroquine is an antimalarial drug that was originally approved by the Food and Drug Administration in April 1955 and has become part of current treatment guidelines for many rheumatic diseases, including lupus, due to its anti-inflammatory properties [15]. The main mechanisms of action of hydroxychloroquine on the immune system include interference with lysosomal acidification and inhibition of chemo-taxis, phagocytosis, and antigen presentation. It also inhibits several inflammatory cytokines such as Il-1, IFNα, and TNF and interferes with Toll-like receptor signaling pathways [16]. The mechanism of action of hydroxychloroquine against SARSCoV-2 is not clearly understood, but it has been found that it changes the pH of endosomes and is believed to prevent viral entry, transport, and post-entry events [17].
There have been many inquiries in the medical community regarding the use of Hydroxychloroquine as a prophylactic drug, with the Indian council of medical research empirically recommending the use of it as prophylaxis for asymptomatic health-care workers and household contacts of confirmed cases in a dose of 400 mg twice per day on day 1 followed by 400 mg once per week for 7 weeks [18,19]. On the other hand, it is also important to point out that some studies have not found hydroxychloroquine to be useful as post-exposure prophylaxis for COVID-19. A randomized, double-blind, placebo-controlled trial that enrolled 821 participants found that the incidence of COVID-19 did not differ significantly between those taking hydroxychloroquine within 4 days after exposure and those who received a placebo [20]. Another fundamental fact that must be considered is that the FDA issued a statement against hydroxychloroquine and chloroquine for COVID-19 use outside the hospital setting due to safety concerns, especially related to heart problems. This did not include FDA-approved uses for malaria and autoimmune diseases [21].
Our patients had already been taking hydroxychloroquine for treatment of SLE. We did not find that this drug prevented them from contracting COVID-19. This finding is consistent with that of a study conducted in France, in which data were collected from 17 patients with lupus who had been taking hydroxychloroquine for a median duration of 7.5 years; 76% of these patients developed COVID-19 [22]. Another study, conducted by the Global Rheumatology Alliance, looked at 110 patients with rheumatic disease and showed that 17% of the patients who developed COVID-19 also had lupus [23].
One caveat worth noting is that none of our patients developed severe COVID-19 complications; this may be due to the early administration of hydroxychloroquine in combination with azithromycin. Million et al. reported similar findings in a study of 1061 patients with COVID-19 who received early administration of hydroxychloroquine+azithromycin: only 4.4% of their population had a poor clinical outcome [24].
Conclusions
Based on our observations, we can conclude that our 5 patients with SLE who were taking hydroxychloroquine presented with SARS-CoV-2 infection and symptoms of COVID-19. This case series supports other evidence that hydroxychloroquine is not able to prevent COVID-19. However, it should be noted that 4 of our patients managed this disease at a time when the viral load affecting the population was very high, but presented with only mild-moderate symptoms.
Did hydroxychloroquine influence this outcome? It is still unclear. Further studies will be needed to get a better understanding of the clinical course of COVID-19 in patients with SLE treated with hydroxychloroquine.
References:
1.. Saqrane S, El Mhammedi MA, Review on the global epidemiological situation and the efficacy of chloroquine and hydroxychloroquine for the treatment of COVID-19: New Microbes New Infect, 2020; 35; 100680
2.. , Actualización de casos de coronavirus en Ecuador 2020 [Available from: ] [in Spanish]https://www.salud.gob.ec/actualizacion-de-casos-de-coronavirus-en-ecuador/
3.. Aringer M, Costenbader K, Daikh D, 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus: Arthritis Rheumatol, 2019; 71(9); 1400-12
4.. Chang AY, Piette EW, Foering KP, Response to antimalarial agents in cutaneous lupus erythematosus: A prospective analysis: Arch Dermatol, 2011; 147(11); 1261-67
5.. Ponticelli C, Moroni G, Hydroxychloroquine in systemic lupus erythematosus (SLE): Expert Opin Drug Saf, 2017; 16(3); 411-19
6.. Liu J, Cao R, Xu M: Cell Discov, 2020; 6; 16
7.. Yao X, Ye F, Zhang M: Clin Infect Dis, 2020; 71(15); 732-39
8.. , FDA cautions against use of hydroxychloroquine or chloroquine for COVID-19 outside of the hospital setting or a clinical trial due to risk of heart rhythm problems 2020, July 1 https://wwwfda.gov/drugs/drug-safety-and-availability/fda-cautions-against-use-hydroxychloroquine-or-chloroquine-covid-19-outside-hospital-setting-or
9.. Mahase E, Covid-19: Hydroxychloroquine was ineffective as postexposure prophylaxis, study finds: Br Med J, 2020; 369; m2242
10.. Torjesen I, Covid-19: Hydroxychloroquine does not benefit hospitalised patients, UK trial finds: Br Med J, 2020; 369; m2263
11.. Patrì A, Fabbrocini G, Hydroxychloroquine and ivermectin: A synergistic combination for COVID-19 chemoprophylaxis and treatment?: J Am Acad Dermatol, 2020; 82(6); e221
12.. Joob B, Wiwanitkit V, SLE, hydroxychloroquine and no SLE patients with COVID-19: A comment: Ann Rheum Dis, 2020; 79(6); e61
13.. Sawalha AH, Patients with lupus are not protected from COVID-19: Ann Rheum Dis, 2020 [Epub ahead of print]
14.. Buonaguro FM, Puzanov I, Ascierto PA, Anti-IL6R role in treatment of COVID-19-related ARDS: J Transl Med, 2020; 18(1); 165
15.. Sinha N, Balayla G, Hydroxychloroquine and covid-19: Postgrad Med J, 2020; 96(1139); 550-55
16.. Ben-Zvi I, Kivity S, Langevitz P, Shoenfeld Y, Hydroxychloroquine: From malaria to autoimmunity: Clin Rev Allergy Immunol, 2012; 42(2); 145-53
17.. Singh AK, Singh A, Shaikh A, Chloroquine and hydroxychloroquine in the treatment of COVID-19 with or without diabetes: A systematic search and a narrative review with a special reference to India and other developing countries: Diabetes Metab Syndr, 2020; 14(3); 241-46
18.. Tahiri Joutei Hassani R, Bennis A, Hydroxychloroquine as antiviral prophylaxis for exposed caregivers to Covid-19: An urgent appraisal is needed: J Infect Public Health, 2020; 13(6); 865-67
19.. Sachdeva M, Shah M, Ziv M, Risks of hydroxychloroquine use for COVID-19 prophylaxis: J Am Acad Dermatol, 2020; 83(1); e73-74
20.. Boulware DR, Pullen MF, Bangdiwala AS, A randomized trial of hydroxychloroquine as postexposure prophylaxis for Covid-19: N Engl J Med, 2020; 383(6); 517-25
21.. Singh AP, Tousif S, Umbarkar P, Lal H, A pharmacovigilance study of hydroxychloroquine cardiac safety profile: Potential implication in COVID-19 mitigation: J Clin Med, 2020; 9(6); 1867
22.. Mathian A, Mahevas M, Rohmer J, Clinical course of coronavirus disease 2019 (COVID-19) in a series of 17 patients with systemic lupus erythematosus under long-term treatment with hydroxychloroquine: Ann Rheum Dis, 2020; 79(6); 837-39
23.. Gianfrancesco MA, Hyrich KL, Gossec L, Rheumatic disease and COVID-19: Initial data from the COVID-19 Global Rheumatology Alliance provider registries: Lancet Rheumatol, 2020; 2(5); e250-53
24.. Million M, Lagier JC, Gautret P, Early treatment of COVID-19 patients with hydroxychloroquine and azithromycin: A retrospective analysis of 1061 cases in Marseille, France: Travel Med Infect Dis, 2020; 35; 101738
In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133