17 January 2021: Articles 
Pediatric Fulminant Cerebellitis Is Still a Fatal Disease that We Know Little About! Two Case Reports and a Literature Review
Unusual clinical course, Challenging differential diagnosis, Rare disease
Hakem Alomani1ADEF*, Muhammad Arshad2EF, Mahmoud Elzonfly2B, Ali Ahmad Aldakhil2B, Abdullah H. Alharbi3BD, Abdulrahman Alasqah4BE, Bandar Rashed Alfheed5CD, Hesham Aldhalan6AEFDOI: 10.12659/AJCR.928370
Am J Case Rep 2021; 22:e928370
Abstract
BACKGROUND: Acute cerebellitis is a rare inflammatory disease with a variable clinical course that ranges from self-limiting illness with a benign outcome to a fulminant process (known as fulminant cerebellitis) associated with high risk of morbidity and mortality. It poses a unique challenge to the pediatric community due to its infrequent occurrence and variable presentation, which ultimately contribute to variation in treatment and variation in clinical outcome. This condition has received little attention, with scattered case reports and reviews in the literature and no clear consensus or guidelines for pediatricians to standardize the approach and management.
CASE REPORT: We report the cases of 2 previously healthy children diagnosed with fulminant cerebellitis. The clinical picture started with severe headache, irritability, and acute deterioration of consciousness level with the absence of cerebellar signs. There was radiological evidence of obstructive hydrocephalus and cerebellar tonsillar herniation in the context of the inflamed cerebellum. The 2 patients received timely standard medical and surgical management for acute cerebellitis in the form of neuroprotective measures, osmotherapy, broad-spectrum antibiotics, and CSF diversion. One patient received aggressive immunomodulatory therapy and had a better outcome compared with the other patient who received no immunomodulatory therapy and had a poor outcome.
CONCLUSIONS: A high index suspicion and early aggressive intervention are the best tools for achieving optimal outcomes in fulminant cerebellitis in children, given the lack of classic cerebellar signs. In addition to CSF diversion and broad-spectrum antibiotics and antivirals, some patients may benefit from early immunomodulator therapy and posterior fossa decompressive craniotomy.
Keywords: Cerebellar Ataxia, Cerebellar Diseases, Encephalocele, Adolescent, Encephalitis
Background
Acute cerebellitis is a rare inflammatory disease with a highly variable clinical course that ranges from benign self-limiting disease with minimal symptoms to a fulminant presentation (known as fulminant cerebellitis) associated with a high risk of morbidity and mortality. There is no consensus on the exact definition of acute cerebellitis. Various definitions/terminology are usually used loosely to describe the condition, which often contributes to the reader’s confusion about acute cerebellitis
Acute cerebellitis complications may be related to compression of posterior fossa structures, acute hydrocephalus, and/ or intracranial hypertension [2–4]. Fulminant cerebellitis is a severe form of acute cerebellitis that is a medical and surgical emergency, for which management should be started immediately. Medical management includes antibiotics, antiviral agents, steroids, and close observation in an acute-care setting for signs of high ICP. Surgical options include external ventricular drainage and, in some cases, posterior fossa decompression as a life-saving intervention [5].
Case Reports
CASE PRESENTATION 1:
A previously healthy 13-year-old Saudi boy presented to the ER complaining of severe headache and neck pain of 1-week duration. One day prior to the presentation, the headache increased in severity and was not relieved by analgesics. There was no history of trauma, projectile vomiting, abnormal gait, abnormal eye movement, febrile illness with rash, or exposure to toxins.
Clinically, his vital signs showed HR 80 bpm, BP 95/60 mmHg, normal peripheral perfusion (capillary refill time (CRT) <2 s), RR 22, SpO2 97% in room air, and pain score 10/10. He was alert and oriented to time and place, with a Glasgow Coma Scale (GCS) of 15/15. There was no neck stiffness.
The focused CNS exam revealed pupils 3-mm bilaterally equal and reactive, with no nystagmus, and all other cranial nerves were intact, with good cough and gag reflexes. The ophthalmology exam showed no papilledema. Power, tone, gait, and reflexes were normal. The rest of the exam was unremarkable.
Initial labs showed normal CBC, normal renal, and normal liver function tests, including coagulation profile. An urgent plain brain CT was done and showed a picture of acute obstructive hydrocephalus with dilated lateral and third ventricles, with no mass lesions (Figure 1).
He was initially admitted to the general pediatric ward with a working diagnosis of CNS infection. At that time, he was afebrile with occasional irritability. His GCS remained 15/15, but his heart rate fluctuated toward bradycardia (100–56 bpm), and his BP was around 100/90 mmHg. He received antibiotics in the form of vancomycin, ceftriaxone (started as meningitic doses), and acyclovir, as well as morphine for severe pain. Lumbar puncture (LP) was deferred because of the concern of high intracranial pressure.
A few hours later, the patient became more agitated and irritable (thrashing around and difficult to console). Eventually, he was transferred to the PICU for neuro-vitals monitoring. Six hours later, after admission to the PICU, the patient started to have abnormal behavior in the form of visual and auditory hallucinations, and his pupil’s exam revealed unequal pupils. He was electively intubated, and an urgent brain CT was repeated, which showed a progression of the previously seen hydrocephalus, along with herniation of cerebellar tonsils 9 mm into the foramina magnum. Urgent boluses of 3% NaCl (2 boluses given as 5 ml/kg), fluid resuscitation, and inotropic support were started along with neuroprotective measures targeting deep sedation in addition to phenytoin as an anti-epileptic drug (AED), although no convulsions were observed.
Based on clinical status and the repeated brain CT findings, the patient was taken to the OR for CSF diversion via external ventricular drainage. The surgery was completed within 11 h from his last deterioration, and the brain was under moderate pressure as per Neurosurgery notes, who drained 20 ml of clear CSF with the insertion of an EVD, which drained 52 ml on day 1, 72 ml on day 2, and, unfortunately, no documentation for EVD output in day 3 (the last day before death).
The CSF result showed colorless clear fluid with no WBC or RBCs, with CSF protein and CSF glucose in normal ranges. There was no bacterial growth after 5 days of CSF culture. No CSF virology was done due to lack of PCR services at our institution; however, we empirically started him on acyclovir from day 1. Although he had no history of risk of lead exposure, we tried to have his lead levels tested, but also this test was also not available.
Brain MRI was done the following day, with suboptimal images due to artifacts in the context of the presence of dental braces, but essentially showed progression of the inflamed cerebellum along with cerebellar tonsillar herniation (Figure 2). Although we noticed the clinical and radiological progression and we recognize that its pathophysiology was underpinned by increased pressure in the posterior fossa compartment and death was inevitable without urgent surgical intervention, posterior fossa decompression was not an option at our institution. The request for transfer to a higher-level medical center was denied.
On the fourth day of admission, he started to have fixed dilated pupils (3-mm) bilaterally. An urgent brain CT was done, which showed more progressive downward displacement of the cerebellar tonsils below the foramen magnum base compared to the previous images. There was effacement of cortical sulci, suggesting diffuse brain edema. There was no evidence of intracerebral or axial hemorrhage. Unfortunately, the patient’s condition rapidly worsened, with the loss of all brain stem functions, and brain death was confirmed after a formal 2-day brain death exam as per the Ministry of Health guidelines. The patient was declared dead, and life support was withdrawn.
CASE PRESENTATION 2:
A 14-year-old Saudi girl presented to the ER with a history of persistent headaches for 2 months. The headache was mainly in the frontal area and was moderate in intensity, which interfered with performing her usual daily activities and was not relieved by analgesics. One month before the admission, she noticed a progressive increase in the headache’s intensity, which was associated with fever; however, there was no history of neck stiffness, no photophobia, and no abnormal movements. There was no history of abnormal gait, abnormal eye movements, fever with rash, or toxins exposure.
On the day of admission, the patient complained of severe frontal headache associated with nausea and vomiting.
Clinically, her vital signs were stable: BP 108/65, RR 26, HR 60–70, and body temperature 37.5°C. She appeared to be ill and in pain, but was alert, with no signs of meningeal irritation. Her pupils were 2–3 mm, equal and reactive bilaterally, with no nystagmus, and other cranial nerves were intact, with good cough and gag reflexes. She had normal power, tone, and reflexes, as well as normal gait. Her ophthalmology exam revealed papilledema grade 1. The remainder of the exam was not contributary.
A brain CT was done and showed evidence of obstructive hydrocephalus along with diffuse cerebellar swelling causing compression of the fourth ventricle. The cerebellar tonsils were shown to be slightly displaced below the foramen magnum base with the compressed fourth ventricle and prominent third ventricle. There was no mass effect or midline shift (Figure 3).
Based on the clinical condition and brain CT findings, she was admitted to the PICU with a provisional diagnosis of acute hydrocephalus in the context of acute cerebellitis (post-infectious), and she was electively intubated and given 3% NaCl infusion and kept on full neuroprotective measures. Neurosurgery was involved and planned for CSF diversion with the insertion of a ventriculoperitoneal (VP) shunt. She went to the OR within 14 h of her intubation, and the brain was under moderate pressure during surgery as per Neurosurgery observation.
Her CBC, renal function test, and liver function tests were essentially normal, and her cerebrospinal fluid (CSF) results were also normal, with WBC nil and RBS 840 cells/µl. Her CSF protein and glucose were 38 mg/dl and 3.4 mmol/L, respectively. A CSF culture was negative, as was a serology for brucella. No CSF virology was done due to the lack of PCR services at our institution.
A follow-up MRI (Figure 4) performed 24 h after V-P shunt insertion showed regression of the hydrocephalus, with signs of cerebellar tonsil herniation and edematous cerebellar hemisphere. She was kept on acyclovir 20 mg/kg/dose TID, vancomycin 20 mg/kg/dose QID, and ceftriaxone 50 mg/kg/dose BID. Given the history of fever and the nonspecific symptoms, we started Tamiflu 60 mg/kg/dose BID pending H1N1 screening results. We also started her on azithromycin OD for 3 days for possible mycoplasma infection; however, no mycoplasma serology was done due to lack of this service at our institution. Eventually, H1N1 screening came back negative, and all blood/CSF cultures revealed negative results. Given the grave outcome of the previous patient with fulminant cerebellitis (the boy in case 1 was admitted a few weeks before this) and along with consultation with a senior neurologist, we started pulse steroid therapy 30 mg/kg daily for 3 days as well as IVIG 1 gm/kg for 2 days as immunomodulating agents in treating the fulminant cerebellitis. The patient responded well, with clear clinical and radiological improvement. She was extubated successfully after 6 days and started her rehabilitation course with optimization of her nutritional status as well as intensive physiotherapy. She was discharged from the PICU to continue her rehabilitation plan and complete the antibiotic course. Before discharge from the hospital (1 week after the previous MRI), a follow-up MRI confirmed resolution of the mass effect and normalization of cerebellar structures (Figure 5). In her 3-month follow-up, there was no evidence of major sequelae, with normal CNS exam and gait; however, she had slurred speech, which was not present before, and we think it is a residual of her cerebellum involvement unmasked after her recovery.
Discussion
Acute cerebellitis is a rare inflammatory condition with a variable course. It may have a benign, self-limited course, or it can rarely present as a fulminant disease (known as fulminant cerebellitis), resulting in significant morbidity and mortality [6]. Fulminant cerebellitis with cerebellar swelling usually leads to the creation of obstructive hydrocephalus due to obstruction of normal CSF flow by compression of the fourth ventricle, and the swelled cerebellum eventually compresses the brainstem structures. If these processes are not reversed with timely appropriate intervention, the inevitable increase in pressure inside the closed posterior fossa compartment will lead to more compression of the vital brainstem structures, as well as downward herniation of posterior fossa structures, which ultimately lead to patient death [7–9].
The classic cerebellitis presentation usually involves finding of cerebellar signs (e.g., ataxia, nystagmus, tremors, dysmetria, slurred speech); however, in fulminant cerebellitis, the presentation is usually acute and lacks cerebellar signs. The absence of cerebellar signs in fulminant cerebellitis may be explained by the acute decrease of LOC that precedes the evolution of any cerebellar signs and eventually conceals these signs in clinical examinations. Acute cerebellitis and its fulminant form have received little attention, with scattered case reports and literature reviews, with no clear consensus or guidelines to the pediatric community to standardize the approach and management. We summarized selected case reports that we found in our literature review for acute cerebellitis with a detailed report for presenting symptoms, interventions, and outcomes (Table 1).
Both of the present patients had clinical and radiological features that satisfy the definition of acute fulminant cerebellitis, and both were previously healthy children. Notably, the predominant clinical symptoms were severe headache and irritability with no cerebellar signs, and both had progressive cerebellar swelling that led to obstructive hydrocephalus and eventual brainstem compression, along with downward herniation of posterior fossa structures. The boy presented with a short history of acute-onset, rapidly progressive, very severe headache, while the girl presented with a 2-month history of protracted headache. Usually, the cerebellum’s inflammatory swelling tends to compress the brainstem and induce alterations of consciousness, which can mask the initial stage of cerebellar signs. This process might explain why neither of our cases presented with acute cerebellar signs. Fulminant cerebellitis can present with no antecedent or concurrent fever, and blood tests are often normal or nonspecific in fulminant cerebellitis, which was the case in our patients [2,10]. CSF studies were performed in both children and were essentially normal. We provided a radiographic correlation of this disease and documented changes in the cerebellum with magnetic resonance and computed tomography scans, which showed evidence of inflammatory swelling in the cerebellum. The initial radiological workup usually starts with a brain CT scan due to logistic reasons; however, brain MRI is diagnostic and very useful for follow-up later in the course. A brain CT scan was performed in both cases and showed a picture of acute obstructive hydrocephalus with dilated lateral and third ventricles with cerebellar tonsils herniation and no mass lesions.
Diagnosing acute cerebellitis was challenging in these cases without cerebellar signs and with normal CSF examination; however, the brain MRI could be an adequate imaging technique to demonstrate the cerebellar involvement, and it appears to be diagnostic in such cases. In earlier reports, the most characteristic and common MRI finding mentioned is diffuse cortical swelling of the cerebellar hemispheres, although some reports only mention unilateral cerebellar hemisphere involvement, often complicated by hydrocephalus and/or tonsillar herniation [11,12].
There is no accepted consensus regarding the treatment of fulminant cerebellitis in children; however, most management lines are tailored toward reversing the pathophysiology with CSF diversion in the presence of obstructive hydrocephalus and immunomodulating agents to suppress the ongoing swelling in the cerebellum, as well as broad-spectrum antibiotic/anti-viral drugs for possible concurrent CNS infection. It should be noted that CSF diversion alone will not address the underlying issue of the progressively enlarging cerebellum, and advanced neurosurgical intervention like posterior fossa decompression should be attempted in case of failure of the initial measures [5,13,14].
Both patients received a timely standard medical and surgical management for cerebellitis, in which the obstructive hydro-cephalus and high ICP were managed by osmotherapy, broad-spectrum antibiotics, CSF diversion, and neuroprotective measures. It should be noted that CSF diversion via EVD was carried out after around 10 h in both cases; it might have been done earlier if we had a dedicated in-house pediatric neurosurgery service. Immunomodulating therapy like high-dose steroid (pulse therapy) and/or IVIG can be life-saving, especially in hospitals where advanced neurosurgical interventions such as decompressive posterior fossa are not available. However, because of the lack of a large series of patients with acute cerebellitis, the exact role of steroids and IVIG is unclear [16]. One of our patients received an additional intervention in the form of aggressive immunomodulating therapy by pulse steroids and IVIG and we believe it contributed to the better clinical and radiological outcome. This patient survived and showed evidence of improved cerebellar changes on follow-up imaging, with no major neurological deficit, compared with the other patient, who did not receive immunomodulating agents and ended up with a poor outcome (Table 2).
Conclusions
Acute cerebellitis is a serious disease that can rapidly progress to fulminant cerebellitis, which is a rare but very serious phase/entity of acute cerebellitis. Diagnosis of fulminant cerebellitis can be very challenging due to lack of classical cerebellar signs as well as its nonspecific presenting symptoms that mimic a long list of CNS diseases; hence, a high index of suspicion is of paramount importance. However, these patients usually present with headache, irritability, and signs of high ICP due to the obstructive hydrocephalus, and LOC usually is affected. Surprisingly, cerebellar signs are absent despite cerebellum involvement. It is considered a medical/surgical emergency, and early aggressive interventions should be initiated promptly, which include: urgent brain imaging, a broad-spectrum antibiotic, antiviral agents, CSF diversion, and immunomodulating therapy like high-dose steroid and/or IVIG; however, in some cases, decompression craniotomy for the posterior fossa is needed.
Figures
References:
1.. Sawaishi Y, Takada G, Acute cerebellitis: Cerebellum, 2002; 1(3); 223-28
2.. Montenegro M, Santos S, Li L, Cendes F, Neuroimaging of acute cerebellitis: J Neuroimaging, 2002; 12; 72-74
3.. Wagel J, Gruszka J, Szewczyk P, Herniation to foramen magnum in the course of cerebellitis in a 4-year-old boy, as shown by CT and MRI – case report: Pol J Radiol, 2010; 75(3); 42-46
4.. De Mendonca J, Barbosa H, Viana S, Pseudotumoural hemicerebellitis: Imaging findings in two cases: Br J Radiol, 2005; 935(78); 1042-46
5.. De Ribaupierre S, Meagher-Villemure K, Villemure J, The role of posterior fossa decompression in acute cerebellitis: Childs Nerv Syst, 2005; 21; 970-74
6.. Kornreich L, Shkalim-Zemer V, Levinsky Y, Acute cerebellitis in children: A many-faceted disease: J Child Neurol, 2016; 31(8); 991-97
7.. Kamate M, Chetal V, Hattiholi V, Fulminant cerebellitis: A fatal, clinically isolated syndrome: Pediatr Neurol, 2009; 41(3); 220-22
8.. Levy EI, Harris AE, Omalu BI, Sudden death from fulminant acute cerebellitis: Pediatr Neurosurg, 2001; 35; 24-28
9.. Dogulu F, Onk A, Kaymaz M, Acute cerebellitis with hydrocephalus: Neurology, 2003; 60; 1717
10.. De Bruecker Y, Claus F, Demaerel P, MRI findings in acute cerebellitis: Eur Radiol, 2004; 14; 1478-83
11.. Ciardi M, Giacchetti G, Fedele C, Acute cerebellitis caused by herpes simplex virus type 1: Clin Infect Dis, 2003; 36; e50-54
12.. Sawaishi Y, Takada G, Acute cerebellitis: Cerebellum, 2002; 1; 223-28
13.. Yiş U, Kurul SH, Cakmakçi H, Dirik E, Acute cerebellitis with cerebellar swelling successfully treated with standard dexamethasone treatment: Cerebellum, 2008; 7(3); 430-32
14.. Hamada H, Kurimoto M, Masuoka T, A case of surgically treated acute cerebellitis with hydrocephalus: Childs Nerv Syst, 2001; 17(8); 500-2
15.. Emelifeonwu JA, Shetty J, Kapiaperumal C, Acute cerebellitis in children: A variable clinical entity: J Child Neurol, 2018; 33(10); 675-84
16.. Amador N, Scheithauer BW, Giannini C, Raffel C, Acute cerebellitis presenting as tumor: J Neurosurg, 2007; 107(Suppl. 1); 57-61
Figures
Tables
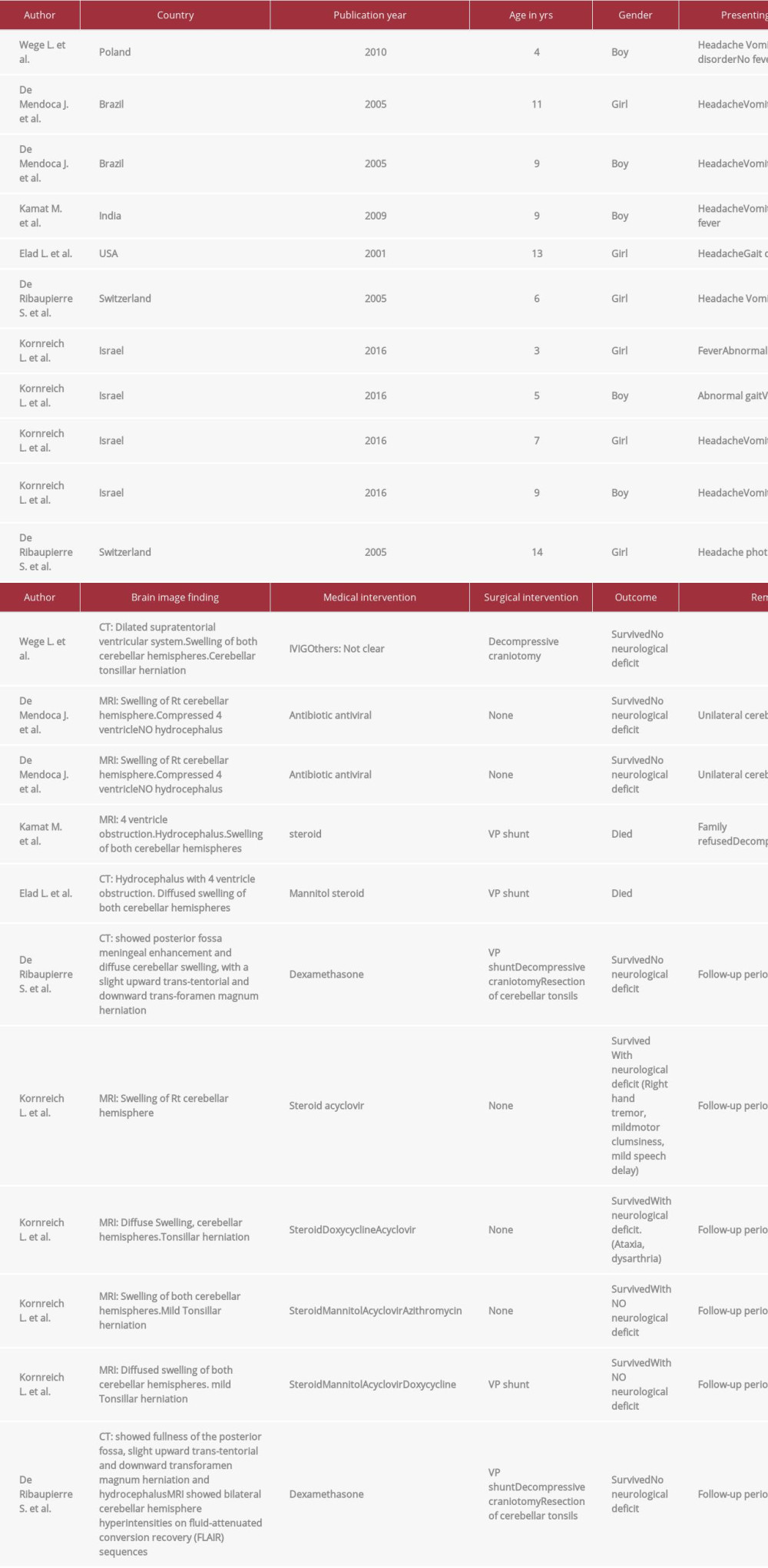 Table 1.. Summary and findings of case reports in the literature review of fulminant cerebellitis (total 11 cases).
Table 1.. Summary and findings of case reports in the literature review of fulminant cerebellitis (total 11 cases).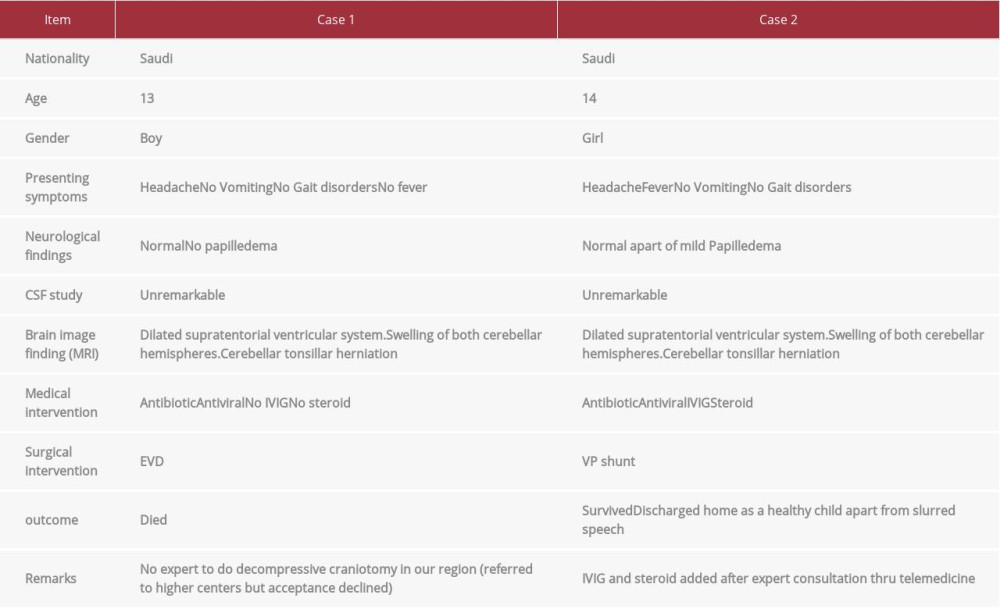 Table 2.. Summary and finding of our cases of fulminant cerebellitis (total 2 cases).
Table 2.. Summary and finding of our cases of fulminant cerebellitis (total 2 cases). Table 1.. Summary and findings of case reports in the literature review of fulminant cerebellitis (total 11 cases).
Table 1.. Summary and findings of case reports in the literature review of fulminant cerebellitis (total 11 cases). Table 2.. Summary and finding of our cases of fulminant cerebellitis (total 2 cases).
Table 2.. Summary and finding of our cases of fulminant cerebellitis (total 2 cases). In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








