27 May 2021: Articles 
An Unusual Case of Hashimoto’s Thyroiditis Presenting as Impending Cardiac Tamponade in a Patient with Acquired Immune Deficiency Syndrome (AIDS)
Challenging differential diagnosis, Management of emergency care, Educational Purpose (only if useful for a systematic review or synthesis), Rare coexistence of disease or pathology
Ryan B. Sinit1AEF, Janet H. Leung23BD, Wayne S. Hwang4BDEF, J. Susie Woo4BD, David M. Aboulafia15ABDEF*DOI: 10.12659/AJCR.929249
Am J Case Rep 2021; 22:e929249
Abstract
BACKGROUND: Cardiac tamponade is a life-threatening pericardial compressive disorder that is a common downstream manifestation of infections, malignancy, and metabolic disorders. Hypothyroidism is a rare cause of tamponade that is attributed to the slow accumulation of effusive fluid into the intrapericardial space. In individuals living with HIV/AIDS, tamponade is commonly associated with infection or malignancy. To our knowledge, this is the first reported case of a patient with HIV/AIDS to have been identified with tamponade secondary to hypothyroidism.
CASE REPORT: Herein, we describe the case of a 52-year-old male patient with a history of AIDS, who presented with nausea, vomiting, diarrhea, and episodic gastrointestinal discomfort for the past several weeks, in conjunction with progressive fatigue. At initial presentation, he had no hemodynamic or clinical signs of tamponade, but pericardial effusion was incidentally found on imaging. Further investigations revealed an undiagnosed Hashimoto’s thyroiditis as a function of restored immunocompetency, which ultimately led to the impending tamponade in this patient. We describe his clinical course through diagnosis of autoimmune hypothyroidism, review cardiac tamponade and hypothyroidism in the context of people living with HIV/AIDS, and discuss this rare manifestation of restored immunocompetency.
CONCLUSIONS: Hypothyroidism should be ruled out in all patients presenting with pericardial effusions or cardiac tamponade, even in people living with HIV/AIDS or those with a history of immune deficiencies.
Keywords: Cardiac Tamponade, Hashimoto disease, HIV, Hypothyroidism, Pericardial Effusion, Acquired Immunodeficiency Syndrome
Background
Cardiac tamponade is a life-threatening pericardial compressive disorder that results from pericardial effusion, with or without pericarditis. Cardiac tamponade occurs when these forces increase the intrapericardial pressure to a level above the limit of pericardial stretch and force the heart to compensate for the reduced intrapericardial volume. In turn, all cardiac chambers are compressed, cardiac filling is impeded, and cardiac function is compromised [1].
Although pericardial effusions may present either asymptomatically or symptomatically, patients with acute cardiac tamponade usually present with chest pain, hypotension, tachypnea, and dyspnea. Beck’s triad (hypotension with a narrowed pulse, jugular venous distension, and muffled heart sounds) and
Determining the etiology of pericardial effusion or pericarditis leading to cardiac tamponade can be difficult as there is a wide gamut of causes, including infectious, autoimmune, malignant, iatrogenic, and traumatic etiologies [3]. This assessment is complicated further in patients with multiple comorbidities, including those with acquired or iatrogenic immunosuppression [4].
Herein, we describe a patient with a long history of HIV infection, but with immune reconstitution and a non-detectable HIV viral load, who presented to medical attention reporting fatigue, confusion, and non-specific gastrointestinal symptoms. He was ultimately diagnosed with a large pericardial effusion and tamponade likely due to severe Hashimoto’s hypothyroidism, as a complete workup found no other etiology. An immune reconstitution syndrome (IRIS) may rarely complicate immune restoration following initiation of antiretroviral therapy (ART) in HIV-infected patients [5]. We review etiologies of pericardial effusions and cardiac tamponade and then focus on clinical experience with this rare cause of cardiovascular collapse in the setting of HIV infection.
Case Report
A 52-year-old gay man presented to a local emergency room complaining of nausea, vomiting, diarrhea, and episodic gastrointestinal discomfort for the past several weeks, in conjunction with progressive fatigue. His symptoms were punctuated by mild confusion and acute epigastric pain of several hours’ duration. He denied cold intolerance or weight changes. His relevant past medical history included HIV infection of 20 years duration absent of AIDS-defining complications, spontaneous clearance of acute hepatitis C genotype 1a infection a decade earlier, and several hospitalizations for methicillin-resistant
At initial evaluation, he was afebrile with a blood pressure of 150/96 mmHg, a heart rate of 80 beats/minute, and a respiratory rate of 14/minute. He was lethargic, with significant psychomotor retardation, but his neurologic exam was otherwise non-focal. His neck veins were nominally distended, his lungs were clear to auscultation, and his cardiac exam revealed a regular rhythm and a soft 1/6 systolic ejection murmur. There was no extremity edema or skin rash.
Laboratory studies, including a complete blood count (with normal differential), complete metabolic panel, immunology panel, HIV viral load testing, and cardiac enzyme results, are reviewed in Table 1. Results were notable for new hyponatremia, renal insufficiency, mild transaminitis, and elevated C-reactive protein. The toxicology screen demonstrated recent use of amphetamines and opiates.
Initial imaging studies included a normal computed tomography (CT) scan of the head. A CT scan of the abdomen and pelvis, to better assess the patient’s non-specific gastrointestinal symptoms, was also unremarkable except for upper abdominal cuts showing a large pericardial effusion, trace left pleural effusion, and small abdominal ascites. These CT findings prompted an electrocardiogram (ECG) tracing that showed low voltage in the limb leads (Figure 1) and transthoracic echocardiogram (TTE) that showed a small left ventricle with a normal left ventricular ejection fraction (LVEF) of 63% (normal, 50–70%), enlarged aortic root, and a large pericardial effusion with signs of hemo-dynamic compromise and tamponade physiology (40% respiratory variation in mitral valve inflow velocities and diastolic inversion of the right ventricle) (Figure 2). Around this time, the patient became dyspneic, his cardiovascular exam was notable for diminished heart sounds, and his hemodynamics were labile.
A cardiologist initially drained 750 mL of a cloudy yellow fluid via radiographic-assisted pericardiocentesis. This procedure normalized the central venous pressure from 8 mmHg to 3 mmHg. The pericardial drain was left in place as per protocol. Pericardial fluid was mildly alkaline (pH of 7.70, normal 6.80–7.60) and exudative in character (protein of 6.4 g/dL, white blood cell [WBC] count of 306×106 cells/L, red blood cell count [RBC] of <10 000×106 cells/L, and lactate dehydrogenase [LDH] of 427 U/L [normal, <243 U/L]). The fluid was negative for rheumatoid factor and adenosine deaminase, and smears and culture showed no bacterial or fungal pathogens, while cytopathology and flow cytometry showed no neoplastic cells.
Although the patient received sedation at the time of pericardiocentesis, he remained obtunded for several days. His failure to return to baseline mental status prompted a brain magnetic resonance imaging (MRI) scan, which was unremarkable. Lumbar puncture showed a normal opening pressure, cerebro-spinal fluid (CSF) protein of 165 mg/dL (normal, 15–45 mg/dL), no RBCs, WBC of 1.0×106 cells/L (normal, 1.0–5.0×106 cells/L), and a non-reactive viral disease research laboratory (VDRL) test for syphilis. To elucidate the etiology of cardiac tamponade, a wider panel of laboratory studies was ordered (Table 2).
The patient was diagnosed with Hashimoto’s thyroiditis. Clinically, he had borderline myxedema coma as he had depressed mental status and hyponatremia but without hypothermia or hypercapnia. Using a proposed diagnostic scoring system for myxedema, he would have met criteria with a score of >60 (15 points for obtundation, 5 for non-specific gastrointestinal symptoms, 10 for pericardial effusion, 10 for pulmonary edema, 10 for hyponatremia, and 10 for decreased glomerular filtration rate). However, 20 of the 30 points were related to the presence of effusion [6]. Due to the persistent obtundation but hemodynamic stability, a cosyntropin stimulation test was performed, and adrenal insufficiency was ruled out prior to starting a mini-load of intravenous levothyroxine at 150 mcg for 2 daily doses, followed by 3 additional days of 200 mcg oral levothyroxine daily. His mental status gradually returned to baseline and his gastrointestinal symptoms resolved. We recorded pericardial fluid output daily for 5 days of 750 mL, 60 mL, 165 mL, 73 mL, and 42 mL, respectively. On the fifth day, a follow-up TTE showed mild concentric left ventricular hypertrophy, normal LVEF, a mildly enlarged ascending aorta, bilateral pleural effusions, and resolution of the pericardial effusion. The pericardial drain was removed, and at time of discharge his free T4 was 0.56 ng/dL. He was prescribed 100 mcg of levothyroxine to take daily (1.6 mcg/kg body weight).
Follow-up laboratory studies 6 weeks after discharge were notable for an elevated creatinine of 1.58 mg/dL, elevated TSH of 56.47 mIU/mL, total T4 of 4.6 (range 4.5–12 mcg/dL), and increased level of TPO antibody of 689.6 IU/mL.
Discussion
The etiology of cardiac tamponade is protean and can be difficult to elucidate even with exhaustive investigations (Table 3) [7,8]. In a retrospective study of 322 patients with pericardial effusions confirmed by TTE, the most common etiologies of cardiac tamponade were malignancy-related (22% of all cases of tamponade, 60% having a known, active malignancy), iatrogenic (18%), and infectious (8%). Twenty-nine percent of cases were considered idiopathic and 4% due to hypothyroidism [8]. Despite the lack of a formal diagnosis by initial pericardial fluid cytological analysis and cultures, many cases deemed idiopathic ultimately prove to be related to malignancy or infection [3,9–11].
The most common cardiovascular complications of hypothyroidism are diastolic hypertension, sinus bradycardia, and failure of the sinus node to respond appropriately to stress [12].
The incidence of any-sized pericardial effusion may be as low as 3% at mild/early stage hypothyroidism and as high as 80% when myxedema is present [13]. Hypothyroidism-associated cardiac tamponade is, however, a particularly rare event, attributed to the slow rate of fluid accumulation. Fewer than 60 cases are reported in the literature, 10 of which were cases of previously unrecognized hypothyroidism [13–17]. In a review of 8 patients with cardiac tamponade secondary to hypothyroidism, patients reportedly had significantly lower heart rates than in other cases of tamponade (80.75±13.45 beats/minute compared to 112.75±12.87 beats/minute). Thus, tamponade without sinus tachycardia may be a clue to hypothyroidism [18]. Because of the gradual accumulation of pericardial fluid, hypothyroid patients may be less likely to present with significant hemodynamic changes [14,17,19].
In the setting of HIV infection, pericardial effusions and tamponade were much more frequent in the pre-HAART (highly active antiretroviral therapy) era (reviews have reported a range of 5–45% of patients with HIV having pericardial effusions) as compared to the HAART era [4,20–22]. In a recent prospective study involving 802 people living with HIV (PLWH), only 2 were identified with effusions [23]. Effusions and cardiac tamponade are most prevalent in the setting of AIDS and can be a marker of advanced HIV infection, as its occurrence is most often associated with a low CD4+ cell count [24]. Causes of cardiac tamponade in PLWH are predominantly infectious or malignancy-related; in a review of 66 cases, tamponade was caused by Mycobacterium tuberculosis (26%), bacteria (17%), lymphoma and Kaposi sarcoma (10%),
Our patient initially presented with non-specific gastrointestinal symptoms and altered mental status that were presumed to be a function of recent ethanol consumption and illicit drug use. His initial physical examination was largely unremarkable, but in a few hours’ time, was notable for attenuated cardiac sounds and fluctuating blood pressures. As in several other cases in which hypothyroidism was the cause of tamponade, there were no overt signs of hemodynamic compromise and no tachycardia, and the drained fluid showed no signs of infection or malignancy [8,17,20,22,24]. As initial investigations into the etiology of his pericardial effusion proved non-diagnostic and he remained lethargic, additional assessments were pursued, including a full panel of thyroid tests and a lumbar puncture. Profoundly low thyroid hormone levels confirmed a new diagnosis of hypothyroidism, and high TPO antibodies confirmed Hashimoto’s as the etiology.
Hashimoto’s thyroiditis is a unique cause of tamponade in the setting of AIDS. We undertook a PubMed literature search using the various combinations of “cardiac tamponade,” “Hashimoto’s disease,” “hypothyroidism,” “pericardial effusions,” and “HIV/ AIDS.” We found no other cases of Hashimoto’s thyroiditis presenting as cardiac tamponade in PLWH.
Although seemingly paradoxical, the coexistence of immuno-deficiency and autoimmune disease is a clinical scenario that is becoming more common, especially as HAART restores immunocompetency in PLWH. Autoimmune disease can develop at the time of immunologic recovery, also known as immune restoration inflammatory syndrome, or in the context of normalized CD4+ T cell counts [26].
The most common autoimmune diseases in PLWH are vasculitis, autoimmune cytopenias, and rheumatic diseases. Thyroid dys-function is less common; hypothyroidism affects 3% of women and 0.2% of men living with HIV and accounts for 88% of all thyroid dysfunction after HIV/HAART [27]. Graves’ disease is less common, estimated to affect 2.4% of women and 0.2% of men living with HIV/AIDS. Of the 12% of thyroid autoimmune disease in HIV/HAART patients, only half are positive for thyroid autoantibodies, in contrast with the general population, where 90% of thyroid autoantibodies are due to Hashimoto’s [28–31]. In a retrospective review of 52 people infected with HIV and diagnosed with autoimmune disease, 6 cases of autoimmune thyroid disease were identified (5 patients with Graves’ disease and 1 with Hashimoto’s disease which presented as painful thyroiditis) [32]. The median time from HAART initiation to autoimmune thyroid disease onset was 17 months. In contrast, our patient’s HIV disease had been well controlled for over a decade before the occurrence of cardiac tamponade due to severe hypothyroidism.
After initial emergent management of tamponade via a pericardial drain, pericardial effusions secondary to hypothyroidism are responsive to thyroid hormone replacement and regress slowly over a period of several months [15,16,33]. If escalation to immunosuppressive therapy is required in PLWH, the same treatment utilized in non-HIV-infected individuals can be used with good tolerance and efficacy [32].
Conclusions
Large pericardial effusions and cardiac tamponade in PLWH are typically secondary to malignancy or infection, or are iatrogenic. Our experience of insidious onset of cardiac tamponade secondary to Hashimoto’s thyroiditis in a person with HIV and with a normal CD4+ cell count and a non-detectable HIV viral load provides a cautionary case study. Tamponade in the setting of hypothyroidism can be absent of typical findings such as tachycardia or hemodynamic compromise. Although Hashimoto’s hypothyroidism appears to be rarer in patients with HIV/AIDS than in the general population, hypothyroidism should be ruled out in all patients presenting with pericardial effusions or cardiac tamponade, even in PLWHA or those with a history of immune deficiencies.
Figures
References:
1.. Spodick DH, Acute cardiac tamponade: N Engl J Med, 2003; 349(7); 684-90
2.. Bodson L, Bouferrache K, Vieillard-Baron A, Cardiac tamponade: Curr Opin Crit Care, 2011; 17(5); 416-24
3.. Strobbe A, Adriaenssens T, Bennett J, Etiology and long-term outcome of patients undergoing pericardiocentesis: J Am Heart Assoc, 2017; 6(12); 1-8
4.. Prendergast BD, HIV and cardiovascular medicine: Heart, 2003; 89(7); 793-800
5.. Visser R, de Mast Q, Netea-Maier RT, van der Ven AJAM, Hashimoto’s thyroiditis presenting as acute painful thyroiditis and as a manifestation of an immune reconstitution inflammatory syndrome in a human immuno-deficiency virus-seropositive patient: Thyroid, 2012; 22(8); 853-55
6.. Popoveniuc G, Chandra T, Sud A, A diagnostic scoring system for myxedema coma: Endocr Pract, 2014; 20(8); 808-17
7.. Sagristà-Sauleda J, Angel J, Permanyer-Miralda G, Soler-Soler J, Long-term follow-up of idiopathic chronic pericardial effusion: N Engl J Med, 1999; 341(27); 2054-59
8.. Sagristà-Sauleda J, Mercé J, Permanyer-Miralda G, Soler-Soler J, Clinical clues to the causes of large pericardial effusions: Am J Med, 2000; 109(2); 95-101
9.. Ben-Horin S, Bank I, Guetta V, Livneh A, Large symptomatic pericardial ef-fusion as the presentation of unrecognized cancer: A study in 173 consecutive patients undergoing pericardiocentesis: Medicine (Baltimore), 2006; 85(1); 49-53
10.. Levy PY, Corey R, Berger P, Etiologic diagnosis of 204 pericardial effusions: Medicine (Baltimore), 2003; 82(6); 385-91
11.. Ma W, Liu J, Zeng Y, Causes of moderate to large pericardial ef-fusion requiring pericardiocentesis in 140 Han Chinese patients: Herz, 2012; 37(2); 183-87
12.. Grais IM, Sowers JR, Thyroid and the heart: Am J Med, 2014; 127(8); 691-98
13.. Kabadi UM, Kumar SP, Pericardial effusion in primary hypothyroidism: Am Heart J, 1990; 120(6); 1393-95
14.. Chou SL, Chern CH, How CK, A rare case of massive pericardial effusion secondary to hypothyroidism: J Emerg Med, 2005; 28(3); 293-96
15.. Ekka M, Ali I, Aggarwal P, Jamshed N, Cardiac tamponade as initial presenting feature of primary hypothyroidism in the ED: Am J Emerg Med, 2014; 32(6); 683 e1–.e3
16.. Chahine J, Ala CK, Gentry JL, Pantalone KM, Klein AL, Pericardial diseases in patients with hypothyroidism: Heart, 2019; 105(13); 1027-33
17.. Manolis AS, Varriale P, Ostrowski RM, Hypothyroid cardiac tamponade: Arch Intern Med, 1987; 147(6); 1167
18.. Wang J-L, Hsieh M-J, Lee C-H, Hypothyroid cardiac tamponade: Clinical features, electrocardiography, pericardial fluid and management: Am J Med Sci, 2010; 340(4); 276-81
19.. Zimmerman J, Yahalom J, Bar-On H, Clinical spectrum of pericardial effusion as the presenting feature of hypothyroidism: Am Heart J, 1983; 106(4 Pt 1); 770-71
20.. Rerkpattanapipat P, Wongpraparut N, Jacobs LE, Kotler MN, Cardiac manifestations of acquired immunodeficiency syndrome: Arch Intern Med, 2000; 160(5); 602-8
21.. Heidenreich PA, Eisenberg MJ, Kee LL, Pericardial effusion in AIDS: Incidence and survival. Circulation, 1995; 92(11); 3229-34
22.. Eisenberg MJ, Gordon AS, Schiller NB, HIV-associated pericardial effusions: Chest, 1992; 102(3); 956-58
23.. Lind A, Reinsch N, Neuhaus K, Pericardial effusion of HIV-infected patients – results of a prospective multicenter cohort study in the era of antiretroviral therapy: Eur J Med Res, 2011; 16(11); 480-83
24.. Katz AS, Sadaniantz A, Echocardiography in HIV cardiac disease: Prog Cardiovasc Dis, 2003; 45(4); 285-92
25.. Estok L, Wallach F, Cardiac tamponade in a patient with AIDS: A review of pericardial disease in patients with HIV infection: Mt Sinai J Med, 1998; 65(1); 33-39
26.. Rogers JS, Zakaria S, Thom KA, Immune reconstitution inflammatory syndrome and human immunodeficiency virus-associated myocarditis: Mayo Clin Proc, 2008; 83(11); 1275-79
27.. Muller I, Moran C, Lecumberri B, 2019 European Thyroid Association guidelines on the management of thyroid dysfunction following immune reconstitution therapy: Eur Thyroid J, 2019; 8(4); 173-85
28.. Roszkiewicz J, Smolewska E, Kaleidoscope of autoimmune diseases in HIV infection: Rheumatol Int, 2016; 36(11); 1481-91
29.. Zandman-Goddard G, Shoenfeld Y, HIV and autoimmunity: Autoimmun Rev, 2002; 1(6); 329-37
30.. Chen F, Day SL, Metcalfe RA, Characteristics of autoimmune thyroid disease occurring as a late complication of immune reconstitution in patients with advanced human immunodeficiency virus (HIV) disease: Medicine (Baltimore), 2005; 84(2); 98-106
31.. Ragusa F, Fallahi P, Elia G, Hashimotos’ thyroiditis: Epidemiology, pathogenesis, clinic and therapy: Best Pract Res Clin Endocrinol Metab, 2019; 33(6); 101367
32.. Iordache L, Launay O, Bouchaud O, Autoimmune diseases in HIV-infected patients: 52 cases and literature review: Autoimmun Rev, 2014; 13(8); 850-57
33.. Omura Y, Ugi S, Sugimoto T, Nishio Y, Massive pericardial effusion secondary to Hashimoto’s disease: Eur J Intern Med, 2007; 18(5); 438-40
Figures
Tables
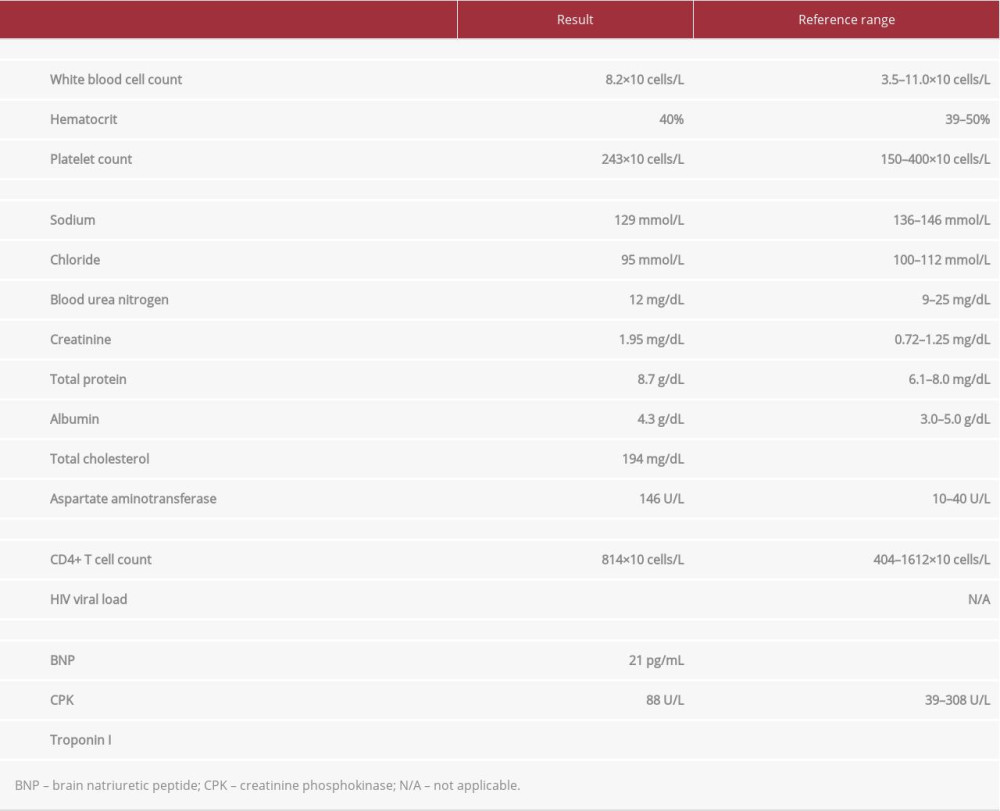 Table 1.. Select results from the patient’s initial laboratory evaluation.
Table 1.. Select results from the patient’s initial laboratory evaluation.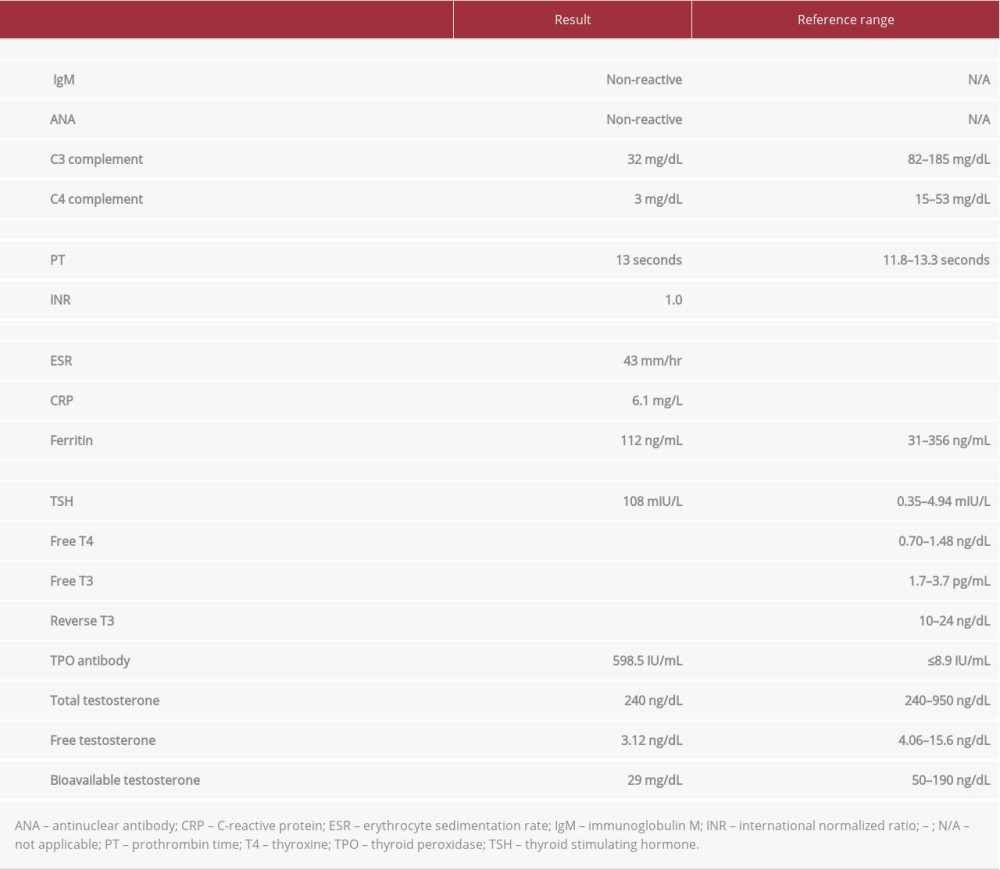 Table 2.. Laboratory tests used to determine etiology of impending cardiac tamponade.
Table 2.. Laboratory tests used to determine etiology of impending cardiac tamponade.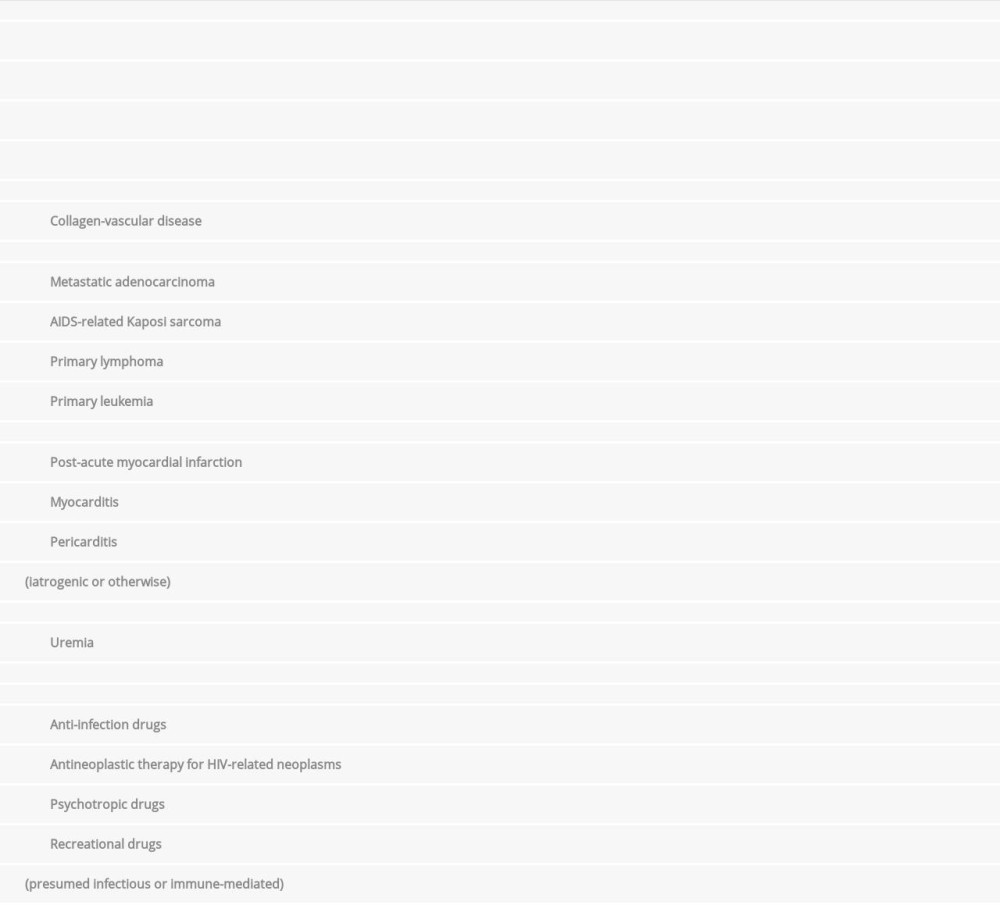 Table 3.. Etiologies of pericardial effusions/cardiac tamponade with a focus on causes related to patients living with HIV/AIDS.
Table 3.. Etiologies of pericardial effusions/cardiac tamponade with a focus on causes related to patients living with HIV/AIDS. Table 1.. Select results from the patient’s initial laboratory evaluation.
Table 1.. Select results from the patient’s initial laboratory evaluation. Table 2.. Laboratory tests used to determine etiology of impending cardiac tamponade.
Table 2.. Laboratory tests used to determine etiology of impending cardiac tamponade. Table 3.. Etiologies of pericardial effusions/cardiac tamponade with a focus on causes related to patients living with HIV/AIDS.
Table 3.. Etiologies of pericardial effusions/cardiac tamponade with a focus on causes related to patients living with HIV/AIDS. In Press
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.949976
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950290
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950607
Case report 
Am J Case Rep In Press; DOI: 10.12659/AJCR.950985
Most Viewed Current Articles
07 Dec 2021 : Case report  17,691,734
17,691,734
DOI :10.12659/AJCR.934347
Am J Case Rep 2021; 22:e934347
06 Dec 2021 : Case report  164,491
164,491
DOI :10.12659/AJCR.934406
Am J Case Rep 2021; 22:e934406
21 Jun 2024 : Case report  113,090
113,090
DOI :10.12659/AJCR.944371
Am J Case Rep 2024; 25:e944371
07 Mar 2024 : Case report  59,175
59,175
DOI :10.12659/AJCR.943133
Am J Case Rep 2024; 25:e943133








